Abstract
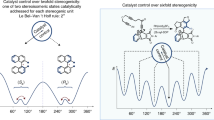
One of the most well-recognized stereogenic elements in a chiral molecule is an sp3-hybridized carbon atom that is connected to four different substituents. Axes of chirality can also exist about bonds with hindered barriers of rotation; molecules containing such axes are known as atropisomers1. Understanding the dynamics of these systems can be useful, for example, in the design of single-atropisomer drugs2 or molecular switches and motors3. For molecules that exhibit a single axis of chirality, rotation about that axis leads to racemization as the system reaches equilibrium. Here we report a two-axis system for which an enantioselective reaction produces four stereoisomers (two enantiomeric pairs): following a catalytic asymmetric transformation, we observe a kinetically controlled product distribution that is perturbed from the system’s equilibrium position. As the system undergoes isomerization, one of the diastereomeric pairs drifts spontaneously to a higher enantiomeric ratio. In a compensatory manner, the enantiomeric ratio of the other diastereomeric pair decreases. These observations are made for a class of unsymmetrical amides that exhibits two asymmetric axes—one axis is defined through a benzamide substructure, and the other axis is associated with differentially N,N-disubstituted amides. The stereodynamics of these substrates provides an opportunity to observe a curious interplay of kinetics and thermodynamics intrinsic to a system of stereoisomers that is constrained to a situation of partial equilibrium.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Data deposits
Crystallographic data are deposited with the Cambridge Crystallographic Data Centre under the accession number CCDC 969575 (for 5-(X-ray)).
References
Oki, M. Topics in Stereochemistry Vol. 1 Atropisomerism (Wiley Interscience, 1983)
Clayden, J., Moran, W. J., Edwards, P. J. & LaPlante, S. R. The challenge of atropisomerism in drug discovery. Angew. Chem. Int. Edn 48, 6398–6401 (2009)
Feringa, B. L. The art of building small: from molecular switches to molecular motors. J. Org. Chem. 72, 6635–6652 (2007)
Eliel, E. L. & Wilen, S. Stereochemistry of Organic Compounds 425 (Wiley Interscience, 1994)
Reist, M., Testa, B., Carrupt, P.-A., Jung, M. & Schurig, V. Racemization, diastereomerization, and epimerization: their meaning and pharmacological significance. Chirality 7, 396–400 (1995)
Reichert, S. & Breit, B. Development of an axial chirality switch. Org. Lett. 9, 899–902 (2007)
Wang, J. & Feringa, B. L. Dynamic control of chiral space in a catalytic asymmetric reaction using a molecular motor. Science 331, 1429–1432 (2011)
Kelly, T. R., De Silva, H. & Silva, R. A. Unidirectional rotary motion in a molecular system. Nature 401, 150–152 (1999)
LaPlante, S. R. et al. Assessing atropisomer axial chirality in drug discovery and development. J. Med. Chem. 54, 7005–7022 (2011)
Barrett, K. T. & Miller, S. J. Enantioselective synthesis of atropisomeric benzamides through peptide-catalyzed bromination. J. Am. Chem. Soc. 135, 2963–2966 (2013)
Keith, J. M., Larrow, J. F. & Jacobsen, E. N. Practical considerations in kinetic resolution reactions. Adv. Synth. Catal. 343, 5–26 (2001)
Cox, C. & Lectka, T. Synthetic catalysis of amide isomerization. Acc. Chem. Res. 33, 849–858 (2000)
Mannschreck, A., Mattheus, A. & Rissmann, G. Comparision of kinetic results obtained by NMR line shape and equilibration methods. J. Mol. Spectrosc. 23, 15–31 (1967)
Ahmed, A. et al. Barriers to rotation about the chiral axis of tertiary aromatic amides. Tetrahedron 54, 13277–13294 (1998)
Iwamura, H. & Mislow, K. Stereochemical consequences of dynamic gearing. Acc. Chem. Res. 21, 175–182 (1988)
Bringmann, G. et al. Atroposelective synthesis chiral biaryl compounds. Angew. Chem. Int. Edn 44, 5384–5427 (2005)
Erol, S. & Dogan, I. Determination of barriers to rotation of axially chiral 5-methyl-2-(o-aryl)imino-3-(o-aryl)thiazolidine-4-ones. Chirality 24, 493–498 (2012)
Chupp, J. P. & Olin, J. F. Chemical and physical properties of some rotational isomers of α-haloacetanilides, a novel unreactive halogen system. J. Org. Chem. 32, 2297–2303 (1967)
Zhao, Y. & Truhlar, D. G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 120, 215–241 (2008)
Campomanes, P., Menendez, M. I. & Sordo, T. L. A theoretical analysis of enantiomerization in aromatic amides. J. Phys. Chem. 106, 2623–2628 (2002)
Bragg, R. A., Clayden, J., Morris, G. A. & Pink, J. H. Stereodynamics of bond rotation in tertiary aromatic amides. Chemistry 8, 1279–1289 (2002)
Pirkle, W. H., Welch, C. J. & Zych, A. J. Chromatographic investigation of the slowly interconverting atropisomers of hindered naphthamides. J. Chromatogr. A 648, 101–109 (1993)
Clayden, J. & Pink, J. H. Concerted rotation in a tertiary aromatic amide: toward a simple molecular gear. Angew. Chem. Int. Edn 37, 1937–1939 (1998)
Cuyegkeng, M. A. & Mannschreck, A. Chromatographic separation of enantiomers and barriers to enantiomerization of axially chiral aromatic carboxamides. Chem. Ber. 120, 803–809 (1987)
Lee, W. K., Park, Y. S. & Beak, P. Dynamic thermodynamic resolution: advantage by separation of equilibration and resolution. Acc. Chem. Res. 42, 224–234 (2009)
Hirsch, R. & Hoffmann, R. W. A test on the configurational stability of chiral organolithium compounds based on kinetic resolution; scope and limitations. Chem. Ber. 125, 975–982 (1992)
LaPlante, S. R., Edwards, P. J., Fader, L. D., Jakalian, A. & Hucke, O. Revealing atropisomer axial chirality in drug discovery. ChemMedChem 6, 505–513 (2011)
LaPlante, S. R. et al. Enantiomeric atropisomers inhibit HCV polymerase and/or HIV matrix: characterizing hindered bond rotations and target selectivity. J. Med. Chem. 57, 1944–1951 (2014)
Klussmann, M. et al. Thermodynamic control of asymmetric amplification in amino acid catalysis. Nature 441, 621–623 (2006)
Acknowledgements
We are grateful to the National Institute of General Medical Sciences of the NIH (GM-068649) for support. We also thank L. Guard for X-ray crystallography. A.J.M. was supported by the National Science Foundation Graduate Research Fellowship Program. All computational work was supported by the facilities and staff of the Yale University Faculty of Arts and Sciences High Performance Computing Center, and by the National Science Foundation under grant number CNS 08-21132, which partially funded acquisition of the facilities.
Author information
Authors and Affiliations
Contributions
S.J.M. and K.T.B. designed the project. K.T.B. performed the experiments. A.J.M. performed the theoretical calculations. All authors contributed to the analysis of data and composition of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data, Supplementary Figures, Supplementary Tables and additional references - see Contents for details. (PDF 7476 kb)
Rights and permissions
About this article
Cite this article
Barrett, K., Metrano, A., Rablen, P. et al. Spontaneous transfer of chirality in an atropisomerically enriched two-axis system. Nature 509, 71–75 (2014). https://doi.org/10.1038/nature13189
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13189
This article is cited by
-
Discovery and synthesis of atropisomerically chiral acyl-substituted stable vinyl sulfoxonium ylides
Nature Chemistry (2024)
-
Organocatalytic aromatization-promoted umpolung reaction of imines
Nature Chemistry (2024)
-
Photoinduced dual bond rotation of a nitrogen-containing system realized by chalcogen substitution
Nature Chemistry (2024)
-
Effective chirality discrimination via dissipation dynamics
Quantum Information Processing (2023)
-
Carbene-catalyzed atroposelective synthesis of axially chiral styrenes
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.