Abstract
Increases in brain blood flow, evoked by neuronal activity, power neural computation and form the basis of BOLD (blood-oxygen-level-dependent) functional imaging. Whether blood flow is controlled solely by arteriole smooth muscle, or also by capillary pericytes, is controversial. We demonstrate that neuronal activity and the neurotransmitter glutamate evoke the release of messengers that dilate capillaries by actively relaxing pericytes. Dilation is mediated by prostaglandin E2, but requires nitric oxide release to suppress vasoconstricting 20-HETE synthesis. In vivo, when sensory input increases blood flow, capillaries dilate before arterioles and are estimated to produce 84% of the blood flow increase. In pathology, ischaemia evokes capillary constriction by pericytes. We show that this is followed by pericyte death in rigor, which may irreversibly constrict capillaries and damage the blood–brain barrier. Thus, pericytes are major regulators of cerebral blood flow and initiators of functional imaging signals. Prevention of pericyte constriction and death may reduce the long-lasting blood flow decrease that damages neurons after stroke.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Peppiatt, C. M., Howarth, C., Mobbs, P. & Attwell, D. Bidirectional control of CNS capillary diameter by pericytes. Nature 443, 700–704 (2006)
Fernández-Klett, F., Offenhauser, N., Dirnagl, U., Priller, J. & Lindauer, U. Pericytes in capillaries are contractile in vivo, but arterioles mediate functional hyperemia in the mouse brain. Proc. Natl. Acad. Sci. USA 107, 22290–22295 (2010)
Chan-Ling, T. et al. Desmin ensheathment ratio as an indicator of vessel stability: evidence in normal development and in retinopathy of prematurity. Am. J. Pathol. 165, 1301–1313 (2004)
Bell, R. D. et al. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron 68, 409–427 (2010)
Armulik, A. et al. Pericytes regulate the blood–brain barrier. Nature 468, 557–561 (2010)
Daneman, R., Zhou, L., Kebede, A. A. & Barres, B. A. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature 468, 562–566 (2010)
Göritz, C. et al. A pericyte origin of spinal cord scar tissue. Science 333, 238–242 (2011)
Dore-Duffy, P., Katychev, A., Wang, X. & Van Buren, E. CNS microvascular pericytes exhibit multipotential stem cell activity. J. Cereb. Blood Flow Metab. 26, 613–624 (2006)
Puro, D. G. Physiology and pathobiology of the pericyte-containing retinal microvasculature: new developments. Microcirculation 14, 1–10 (2007)
Villringer, A., Them, A., Lindauer, U., Einhäupl, K. & Dirnagl, U. Capillary perfusion of the rat brain cortex. An in vivo confocal microscopy study. Circ. Res. 75, 55–62 (1994)
Schulte, M. L., Wood, J. D. & Hudetz, A. G. Cortical electrical stimulation alters erythrocyte perfusion pattern in the cerebral capillary network of the rat. Brain Res. 963, 81–92 (2003)
Jespersen, S. N. & Østergaard, L. The roles of cerebral blood flow, capillary transit time heterogeneity, and oxygen tension in brain oxygenation and metabolism. J. Cereb. Blood Flow Metab. 32, 264–277 (2012)
Attwell, D. et al. Glial and neuronal control of blood flow. Nature 468, 232–243 (2010)
Yemisci, M. et al. Pericyte contraction induced by oxidative-nitrative stress impairs capillary reflow despite successful opening of an occluded cerebral artery. Nature Med. 15, 1031–1037 (2009)
Vates, G. E., Takano, T., Zlokovic, B. & Nedergaard, M. Pericyte constriction after stroke: the jury is still out. Nature Med. 16, 959 (2010)
Hall, C. N., Klein-Flügge, M. C., Howarth, C. & Attwell, D. Oxidative phosphorylation, not glycolysis, powers presynaptic and postsynaptic mechanisms underlying brain information processing. J. Neurosci. 32, 8940–8951 (2012)
Hall, C. N. & Attwell, D. Assessing the physiological concentration and targets of nitric oxide in brain tissue. J. Physiol. (Lond.) 586, 3597–3615 (2008)
Zhu, X., Bergles, D. E. & Nishiyama, A. NG2 cells generate both oligodendrocytes and gray matter astrocytes. Development 135, 145–157 (2008)
Krueger, M. & Bechmann, I. CNS pericytes: concepts, misconceptions, and a way out. Glia 58, 1–10 (2010)
Hamilton, N. B., Attwell, D. & Hall, C. N. Pericyte-mediated regulation of capillary diameter: a component of neurovascular coupling in health and disease. Front. Neuroenergetics 2, 5 (2010)
Serebryakov, V., Zakharenko, S., Snetkov, V. & Takeda, K. Effects of prostaglandins E1 and E2 on cultured smooth muscle cells and strips of rat aorta. Prostaglandins 47, 353–365 (1994)
Crawford, C. et al. An intact kidney slice model to investigate vasa recta properties and function in situ. Nephron Physiol. 120, 17–31 (2012)
Kleinfeld, D., Mitra, P. P., Helmchen, F. & Denk, W. Fluctuations and stimulus-induced changes in blood flow observed in individual capillaries in layers 2 through 4 of rat neocortex. Proc. Natl. Acad. Sci. USA 95, 15741–15746 (1998)
Khatri, P. et al. Good clinical outcome after ischemic stroke with successful revascularization is time-dependent. Neurology 73, 1066–1072 (2009)
Gupta, E. et al. Higher volume endovascular stroke centers have faster times to treatment, higher reperfusion rates and higher rates of good clinical outcomes. J. Neurointerv. Surg. 5, 294–297 (2013)
Hauck, E. F., Apostel, S., Hoffmann, J. F., Heimann, A. & Kempski, O. Capillary flow and diameter changes during reperfusion after global cerebral ischemia studied by intravital video microscopy. J. Cereb. Blood Flow Metab. 24, 383–391 (2004)
Leffler, C. W., Beasley, D. G. & Busija, D. W. Cerebral ischemia alters cerebral microvascular reactivity in newborn pigs. Am. J. Physiol. 257, H266–H271 (1989)
Nelson, C. W., Wei, E. P., Povlishock, J. T., Kontos, H. A. & Moskowitz, M. A. Oxygen radicals in cerebral ischemia. Am. J. Physiol. 263, H1356–H1362 (1992)
Baird, A. E. et al. Reperfusion after thrombolytic therapy in ischemic stroke measured by single-photon emission computed tomography. Stroke 25, 79–85 (1994)
Attwell, D. & Iadecola, C. The neural basis of functional imaging signals. Trends Neurosci. 25, 621–625 (2002)
Lovick, T. A., Brown, L. A. & Kay, B. J. Neurovascular relationships in hippocampal slices: physiological and anatomical studies of mechanisms underlying flow-metabolism coupling in intraparenchymal microvessels. Neuroscience 92, 47–60 (1999)
Cohen, Z., Molinatti, G. & Hamel, E. Astroglial and vascular interactions of noradrenaline terminals in the rat cerebral cortex. J. Cereb. Blood Flow Metab. 17, 894–904 (1997)
Blinder, P. et al. The cortical angiome: a 3-D interconnected vascular network with noncolumnar patterns of blood flow. Nature Neurosci. 16, 889–897 (2013)
Mathiesen, C., Brazhe, A., Thomsen, K. & Lauritzen, M. Spontaneous calcium waves in Bergmann glia increase with age and hypoxia and may reduce tissue oxygen. J. Cereb. Blood Flow Metab. 33, 161–169 (2013)
Marcaggi, P. & Attwell, D. Endocannabinoid signaling depends on the spatial pattern of synapse activation. Nature Neurosci. 8, 776–781 (2005)
Peters, B. P. & Goldstein, I. J. The use of fluorescein-conjugated Bandeiraea simplicifolia B4-isolectin as a histochemical reagent for the detection of alpha-D-galactopyranosyl groups. Their occurrence in basement membranes. Exp. Cell Res. 120, 321–334 (1979)
Laitinen, L. Griffonia simplicifolia lectins bind specifically to endothelial cells and some epithelial cells in mouse tissues. Histochem. J. 19, 225–234 (1987)
Norup Nielsen, A. & Lauritzen, M. Coupling and uncoupling of activity-dependent increases of neuronal activity and blood flow in rat somatosensory cortex. J. Physiol. (Lond.) 533, 773–785 (2001)
Frostig, R. D., Lieke, E. E., Ts'o, D. Y. & Grinvald, A. Cortical functional architecture and local coupling between neuronal activity and the microcirculation revealed by in vivo high-resolution optical imaging of intrinsic signals. Proc. Natl. Acad. Sci. USA 87, 6082–6086 (1990)
Harrison, T. C., Sigler, A. & Murphy, T. H. Simple and cost-effective hardware and software for functional brain mapping using intrinsic optical signal imaging. J. Neurosci. Methods 182, 211–218 (2009)
Schaffer, C. B. et al. Two-photon imaging of cortical surface microvessels reveals a robust redistribution in blood flow after vascular occlusion. PLoS Biol. 4, e22 (2006)
Allen, N. J., Káradóttir, R. & Attwell, D. A preferential role for glycolysis in preventing the anoxic depolarization of rat hippocampal area CA1 pyramidal cells. J. Neurosci. 25, 848–859 (2005)
Dore-Duffy, P. et al. Pericyte migration from the vascular wall in response to traumatic brain injury. Microvasc. Res. 60, 55–69 (2000)
Gonul, E. et al. Early pericyte response to brain hypoxia in cats: an ultrastructural study. Microvasc. Res. 64, 116–119 (2002)
Nagel, S. et al. Neuroprotection by dimethyloxalylglycine following permanent and transient focal cerebral ischemia in rats. J. Cereb. Blood Flow Metab. 31, 132–143 (2011)
Garcia, J. H., Wagner, S., Liu, K. F. & Hu, X. J. Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats. Statistical validation. Stroke 26, 627–635 (1995)
Pardridge, W. M. Blood-brain barrier biology and methodology. J. Neurovirol. 5, 556–569 (1999)
Iadecola, C., Yang, G., Ebner, T. J. & Chen, G. Local and propagated responses evoked by focal synaptic activity in cerebellar cortex. J. Neurophysiol. 78, 651–659 (1997)
Blinder, P., Shih, A. Y., Rafie, C. & Kleinfeld, D. Topological basis for the robust distribution of blood to rodent neocortex. Proc. Natl. Acad. Sci. USA 107, 12670–12675 (2010)
Lindauer, U., Villringer, A. & Dirnagl, U. Characterization of CBF response to somatosensory stimulation: model and influence of anesthetics. Am. J. Physiol. 264, H1223–H1228 (1993)
Acknowledgements
We thank B. Clark, A. Gibb, A. Gourine, C. Howarth, R. Jolivet, C. Madry, P. Mobbs, B. Richardson and A. Silver for comments on the manuscript. This work was supported by the Fondation Leducq, European Research Council, Wellcome Trust, UK Medical Research Council, Rosetrees Trust, Nordea Foundation via the Center for Healthy Aging, the Lundbeck Foundation, NOVO-Nordisk Foundation and Danish Medical Research Council.
Author information
Authors and Affiliations
Contributions
All authors (C.N.H., C.R., B.G., N.B.H., A.M., B.A.S., F.M.O., A.M.B., M.L. and D.A.) contributed to designing the study, doing the experiments, analysing the results and writing the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
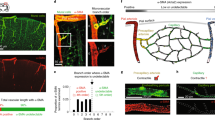
Extended Data Figure 1 Signalling to capillary pericytes in health and disease.
a, As vasodilators released from active neurons and their associated astrocytes (1) diffuse through the brain, they encounter pericytes before arteriole smooth muscle (2) because neurons are closer to capillaries than to arterioles31. This may partly explain why capillaries dilate before arterioles (Fig. 3). Pericyte dilation may spread to arteriole smooth muscle by current flow or [Ca2+]i changes passing through gap junctions (3). b, Oxygen-dependent signalling pathways regulating vessel diameter (adapted from ref. 13). Neuronal activity leads to the generation of nitric oxide (NO) and arachidonic acid. Arachidonic acid (AA) is converted into PGE2, which dilates vessels via EP4 receptors, but also into the vasoconstrictor 20-HETE. Production of 20-HETE is inhibited by NO. Together these pathways regulate capillary diameter (Fig. 1). Larger dilations to glutamate in low [O2] may reflect less production of 20-HETE from arachidonic acid. c, In ischaemia, the decrease of ATP concentration leads to a rise of [Ca2+]i in pericytes. This results in some of them contracting and constricting capillaries1, which will prevent the passage of white and red blood cells. Most pericytes then die (Figs 4 and 5). Death of pericytes in rigor will produce a long-lasting decrease of cerebral blood flow, and reduce the ability of the microvasculature to increase blood flow in response to neuronal activity.
Extended Data Figure 2 Drug effects on capillary baseline diameter and constriction to noradrenaline.
a, Rat capillaries are constricted by 100 µM L-NG-nitroarginine (L-NNA), suggesting that there is some tonic release of NO in the slice, with a greater constriction occurring at high [O2] (bars in a–d, f–n show percentage of initial drug-free diameter). b, However, L-NNA does not affect the diameter of vessels preconstricted with noradrenaline (NA; 2 μM, P = 0.81, ANOVA). c, The guanylyl cyclase blocker ODQ (10 µM) does not affect baseline capillary diameter, suggesting that the constricting effect of tonic NO release seen in a is not through the cGMP pathway but is via suppression of 20-HETE release. d, ODQ slightly enhances the constriction achieved with NA (P = 0.003, ANOVA). P values on the graph are from post hoc t-tests. e, As expected, ODQ blocks cGMP production by guanylyl cyclase, as assessed by radioimmunoassay. f, Inhibition of 20-HETE formation with 1 µM HET0016 (HET) does not affect baseline capillary diameter (white bars, at 20% O2, P = 0.78; at 95% O2, P = 0.49), presumably because the tonic NO release (in a) is sufficient to suppress tonic 20-HETE release, and there is no significant difference in baseline diameter in the presence of HET between the two O2 concentrations (P = 0.57, t-test). Unlike application of L-NNA alone (see a), application of L-NNA and HET together (black bars) does not significantly change capillary diameter (P = 0.59, ANOVA compared to HET alone). Indeed, comparing vessel diameters in HET plus L-NNA with those in L-NNA alone (black bars in f versus panel a) reveals that HET significantly relieves the constriction produced by L-NNA (P = 0.03, ANOVA). g, HET does not affect either the constriction to NA (white bars versus white bars in b, P = 0.51, ANOVA) or the diameter of vessels in L-NNA and NA (black bars versus black bars in b, P = 0.26, ANOVA). h, i, Blocking synthesis of epoxy-derivatives of arachidonic acid with MS-PPOH 10 µM does not affect the degree of constriction to NA (h, P = 0.92, ANOVA) or the dilation to glutamate (i; P = 0.92, ANOVA). j–l, Blocking EP4 receptors with 1 µM L161,982 had no effect on baseline diameter in cerebellum (j) or cortex (97.3 ± 1.4% of baseline in 20% O2, P = 0.07, n = 34) and also did not affect the constriction to NA in either area (cerebellum: k, P = 0.90, ANOVA over both [O2]; l, cortex in 20% O2). m, n, Applying DETA-NONOate (m, 100 μM) or prostaglandin E2 (n, 1 μM) dilated cerebellar capillaries preconstricted with noradrenaline (in 20% O2). Data shown as mean ± s.e.m.
Extended Data Figure 3 Pericyte current responses in NG2-DsRed mouse cerebellar slices.
Mean initial inward and later outward currents (as in Fig. 2a–d, 95% O2) evoked in pericytes by stimulation of the parallel fibres, and by superfusion of 500 μM glutamate or 100 μM NMDA. Numbers of cells apply to both black and white bars. Error bars show s.e.m.
Extended Data Figure 4 Smoothing of the in vivo diameter changes.
a, Specimen single frame from the image sequence for Fig. 3d (penetrating arteriole on the right, capillary on the left). The presence of red blood cells (RBC) leads to apparent holes in the image of the capillary diameter. b, RBC movement results in the holes being removed when averaging over 10 frames. c, For a step increase in vessel diameter (top), the effect on the measured diameter time course (bottom) of a running maximum intensity average being calculated over 10 frames starting at each time being considered: the maximum intensity summation results in the largest diameter at any time dominating the smaller diameters at other times, and so the diameter increase is brought forward by 10 frames. If this were not corrected for, the diameter would appear to increase 10 frames (1.72 s) before it actually does. To correct for this, the time axis needs to be advanced by 1.72 s. d, Correction of the 10-frame averaged time courses of the data used for Fig. 3d for the time shift introduced by the averaging. e, Effect of the five-point FFT procedure (which removes frequencies over 1.16 Hz) applied to the averaged time courses in d. The smooth dashed lines are the traces plotted in Fig. 3d.
Extended Data Figure 5 Responses of capillaries in somatosensory cortex in vivo.
a, b, Reproducibility of response to whisker-pad stimulation at 101 capillary locations in NG2-DsRed mice. Mean capillary response time courses are the same on repeated stimulation (a). Responses at 5 and 15 s into 15 s whisker-pad stimulation did not differ significantly between the first and second stimulation (b). c, Time to reach a certain percentage of the maximum dilation in j–1th order vessel minus that in jth order vessel (j is an integer ≥ 1) imaged simultaneously. The time to 10% and 20% of the peak is faster in first-order capillaries than in zero-order penetrating arterioles (see main text and Fig. 3f), although there are no significant differences between vessels of adjacent orders for any of the other bars shown (P = 0.33–1, t-tests; n for each comparison was as in Fig. 3f). d, Time course of responses in all responding (>5%) vessels of different order. Arterioles were significantly slower to reach 10% of their peak response than first- and second-order vessels (see main text). e, The response distributions of capillaries do not differ near pericyte somata or processes (P = 0.24, Kolmogorov–Smirnov test; 172 somata locations, 292 process locations). f, Comparison of time course of dilation of penetrating arterioles and first-order capillaries with that of the blood-flow increase in capillaries (n = 49, all orders averaged) assessed by line-scanning (normalized to the average value at the peak from 11.7 to 13.2 s). Data shown as mean ± s.e.m.
Extended Data Figure 6 Ischaemia-evoked pericyte death in cortical slices and in vivo.
a, Rat pericyte death is not affected by either of the free radical scavengers MnTBAP (150 µM) or PBN (100 µM; P = 0.78 and P = 1, respectively, ANOVA with Dunnett’s post hoc test versus no drug control). The amount of pericyte death did not differ between the two different scavengers (P = 0.88, ANOVA) so the data from the two scavengers were combined for the analysis in the main text (Fig. 4d). b, In addition to the drugs discussed in the main text, none of the following drugs affected pericyte death following OGD and reoxygenation (ANOVA with Dunnett’s post hoc test versus no drug control): an inhibitor of mitochondrial calcium uptake, Ru360 (50 µM, P = 1), the metabotropic glutamate receptor antagonist MCPG (500 µM, P = 0.93) or the 20-HETE synthesis blocker HET0016 (HET, 1 µM, P = 1). c, The percentage of dead cerebral cortical pericytes and endothelial cells after 24 h in the control and treated hemispheres of MCAO-treated rats, sham-operated animals where a filament was inserted into the ICA but was not advanced far enough to completely occlude the vessel (see Methods), sham animals without ICA occlusion, and naive animals that did not experience any surgery before being killed. These data were analysed together with the striatal data in the main Fig. 5f. There was no difference in cell death between cortex and striatum (P = 0.55, repeated measures ANOVA). A much greater proportion of pericytes died than endothelial cells (P = 2.1×10−7, repeated measures ANOVA), and pericyte death, but not endothelial cell death, was greater in the lesioned hemisphere (effect of hemisphere, P = 0.008, repeated measures ANOVA; interaction between hemisphere and cell type, P = 0.007). As expected, most pericyte death occurred in the MCAO treated animals, whereas sham-operated animals with ICA occlusion showed intermediate levels of death between MCAO and naive (or sham with no ICA occlusion) animals (MCAO versus naive animals, P = 0.004; MCAO versus sham without ICA occlusion, P = 0.01; MCAO versus sham with ICA occlusion, P = 0.20; sham with ICA occlusion versus naive, P = 0.13; Tukey post-hoc tests). Data shown as mean ± s.e.m.
Supplementary information
Noradrenaline constricts and glutamate dilates capillaries
Vessel in Figure 1d responding to noradrenaline and glutamate in a rat cerebellar slice. (MOV 9109 kb)
Glutamate release from neurons dilates capillaries
Vessel in Figure 2e-f responding to noradrenaline and parallel fibre stimulation in a rat cerebellar slice. (MOV 3479 kb)
Capillary and arteriole dilation in vivo
Penetrating arteriole and primary capillary in Figure 3d in mouse somatosensory cortex in vivo responding to whisker pad stimulation. The capillary dilates before the arteriole. Green is FITC-dextran; pericytes are labelled with DsRed. (MOV 2744 kb)
Rights and permissions
About this article
Cite this article
Hall, C., Reynell, C., Gesslein, B. et al. Capillary pericytes regulate cerebral blood flow in health and disease. Nature 508, 55–60 (2014). https://doi.org/10.1038/nature13165
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13165
This article is cited by
-
Sex, hormones and cerebrovascular function: from development to disorder
Fluids and Barriers of the CNS (2024)
-
Induced pluripotent stem cell derived pericytes respond to mediators of proliferation and contractility
Stem Cell Research & Therapy (2024)
-
Exosome-mediated repair of spinal cord injury: a promising therapeutic strategy
Stem Cell Research & Therapy (2024)
-
Profiling human brain vascular cells using single-cell transcriptomics and organoids
Nature Protocols (2024)
-
The role of cardiac pericytes in health and disease: therapeutic targets for myocardial infarction
Nature Reviews Cardiology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.