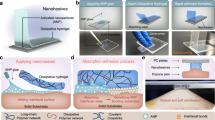
Abstract
Adhesives are made of polymers1 because, unlike other materials, polymers ensure good contact between surfaces by covering asperities, and retard the fracture of adhesive joints by dissipating energy under stress2,3. But using polymers to ‘glue’ together polymer gels is difficult, requiring chemical reactions, heating, pH changes, ultraviolet irradiation or an electric field4,5,6,7. Here we show that strong, rapid adhesion between two hydrogels can be achieved at room temperature by spreading a droplet of a nanoparticle solution on one gel’s surface and then bringing the other gel into contact with it. The method relies on the nanoparticles’ ability to adsorb onto polymer gels and to act as connectors between polymer chains, and on the ability of polymer chains to reorganize and dissipate energy under stress when adsorbed onto nanoparticles. We demonstrate this approach by pressing together pieces of hydrogels, for approximately 30 seconds, that have the same or different chemical properties or rigidities, using various solutions of silica nanoparticles, to achieve a strong bond. Furthermore, we show that carbon nanotubes and cellulose nanocrystals that do not bond hydrogels together become adhesive when their surface chemistry is modified. To illustrate the promise of the method for biological tissues, we also glued together two cut pieces of calf’s liver using a solution of silica nanoparticles. As a rapid, simple and efficient way to assemble gels or tissues, this method is desirable for many emerging technological and medical applications such as microfluidics, actuation, tissue engineering and surgery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kendall, K. Molecular Adhesion and Its Applications (Plenum, 2001)
Lake, G. J. & Thomas, A. G. The strength of highly elastic materials. Proc. R. Soc. 300, 108–119 (1967)
de Gennes, P. G. Soft adhesives. Langmuir 12, 4497–4500 (1996)
Sahlin, J. J. & Peppas, N. A. Enhanced hydrogel adhesion by polymer interdiffusion: use of linear poly(ethylene glycol) as an adhesion promoter. J. Biomater. Sci. Polym. Edn 8, 421–436 (1997)
Tamagawa, H. & Takahashi, Y. Adhesion force behavior between two gels attached with an electrolytic polymer liquid. Mater. Chem. Phys. 107, 164–170 (2008)
Saito, J. et al. Robust bonding and one-step facile synthesis of tough hydrogels with desirable shape by virtue of the double network structure. Polymer Chem. 2, 575–580 (2011)
Techawanitchai, P. et al. Photo-switchable control of pH-responsive actuators via pH jump reaction. Soft Matter 8, 2844–2851 (2012)
Gong, J. P. Friction and lubrication of hydrogels—its richness and complexity. Soft Matter 2, 544–552 (2006)
Reutenauer, P., Buhler, E., Boul, P. J., Candau, S. J. & Lehn, J.-M. Room temperature dynamic polymers based on Diels-Alder chemistry. Chem. Eur. J. 15, 1893–1900 (2009)
Nicolaÿ, R., Kamada, J., Van Wassen, A. & Matyjaszewski, K. Responsive gels based on a dynamic covalent trithiocarbonate cross-linker. Macromolecules 43, 4355–4361 (2010)
Harada, A., Kobayashi, R., Takashima, Y., Hashidzume, A. & Yamaguchi, H. Macroscopic self-assembly through molecular recognition. Nature Chem. 3, 34–37 (2011)
Cordier, P., Tournilhac, F., Soulié-Ziakovic, C. & Leibler, L. Self-healing and thermoreversible rubber from supramolecular assembly. Nature 451, 977–980 (2008)
Wang, Q. et al. High-water-content mouldable hydrogels by mixing clay and a dendritic molecular binder. Nature 463, 339–343 (2010)
Haraguchi, K., Uyama, K. & Tanimoto, H. Self-healing in nanocomposite hydrogels. Macromol. Rapid Commun. 32, 1253–1258 (2011)
Carlsson, L., Rose, S., Hourdet, D. & Marcellan, A. Nano-hybrid self-crosslinked PDMA/silica hydrogels. Soft Matter 6, 3619–3631 (2010)
Gaharwar, A. K., Rivera, C. P., Wu, C.-J. & Schmidt, G. Transparent, elastomeric and tough hydrogels from poly(ethylene glycol) and silicate nanoparticles. Acta Biomater. 7, 4139–4148 (2011)
Duarte, A. P., Coelho, J. F., Bordado, J. C., Cidade, M. T. & Gil, M. H. Surgical adhesives: systematic review of the main types and development forecast. Prog. Polym. Sci. 37, 1031–1050 (2012)
Netz, R. R. & Andelman, D. Neutral and charged polymers at interfaces. Phys. Rep. 380, 1–95 (2003)
Santore, M. M. Dynamics in adsorbed homopolymer layers: understanding complexity from simple starting points. Curr. Opin. Colloid Interf. Sci. 10, 176–183 (2005)
Montarnal, D., Capelot, M., Tournilhac, F. & Leibler, L. Silica-like malleable materials from permanent organic networks. Science 334, 965–968 (2011)
Gent, A. N., Hamed, G. R. & Hung, W. J. Adhesion of elastomer layers to an interposed layer of filler particles. J. Adhes. 79, 905–913 (2003)
Nah, C., Jose, J., Ahn, J. H., Lee, Y. S. & Gent, A. N. Adhesion of carbon black to elastomers. Polym. Test. 31, 248–253 (2012)
Griot, O. & Kitchener, J. A. Role of surface silanol groups in the flocculation of silica suspensions by polyacrylamide. Part 1—Chemistry of the adsorption process. Trans. Faraday Soc. 61, 1026–1031 (1965)
Hourdet, D. & Petit, L. Hybrid hydrogels: macromolecular assemblies through inorganic cross-linkers. Macromol. Symp. 291–292, 144–158 (2010)
Kendall, K. Cracking of short lap joints. J. Adhes. 7, 137–140 (1975)
Johner, A. & Joanny, J.-F. Adsorption of polymeric brushes: bridging. J. Chem. Phys. 96, 6257 (1992)
Stöber, W., Fink, A. & Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interf. Sci. 26, 62–69 (1968)
Prevoteau, A., Soulié-Ziakovic, C. & Leibler, L. Universally dispersible carbon nanotubes. J. Am. Chem. Soc. 134, 19961–19964 (2012)
Bondeson, D., Mathew, A. & Oksman, K. Optimization of the isolation of nanocrystals from microcrystalline cellulose by acid hydrolysis. Cellulose 13, 171–180 (2006)
Greensmith, H. W. Rupture of rubber. X. The change in stored energy on making a small cut in a test piece held in simple extension. J. Appl. Polym. Sci. 7, 993–1002 (1963)
Acknowledgements
We thank D. Montero and L. Olanier for technical assistance with electron microscopy and tensile test equipment, respectively. We also thank L. Alison and L. Nègre for help with experiments and A. Legrand for synthesis of silica AL-30 particles. We thank A. Johner for discussions on adsorption and M. Cloitre, J. Lewiner, A. Maggs, R. Nicolaÿ and F. Tournilhac for encouragements and discussions. S.R. and A.P. acknowledge PhD fellowship funding from ED397, UPMC, Paris France. The financial support of the CNRS, the ESPCI and the UPMC is acknowledged.
Author information
Authors and Affiliations
Contributions
S.R. synthesized samples, designed and performed experiments, analysed data and discussed results, A.P. synthesized carbon nanotube and CNT-Thy samples and performed experiments, P.E. performed experiments and analysed data, D.H. advised on gel synthesis, A.M. conceived the project, designed and performed experiments, analysed and discussed results and wrote the manuscript, L.L. initiated and conceived the project, designed and performed experiments, interpreted results, and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Lap-shear and tensile tests of PDMA S0.1 gels.
a, Lap-joint geometry. Displacement was measured by a video extensometer that followed two markers (white dots), which were placed at a distance of 5 mm from the edge of the lap joint. The total length of the assembled ribbons was 40 mm. w denotes the width and h the thickness of gel ribbons. l is the overlap length. b, Comparison of force–displacement curves for PDMA S0.1 ribbon (black line) and for the lap joint glued by spreading 15 μl of TM-50 silica solution (blue circles). Displacement was measured by optical extensometer from two markers, initially spaced by 20 mm and centred on the joint. The PDMA S0.1/S0.1 assemblies broke outside the joint (inset). c, Lap-shear adhesion test reproducibility. Force–displacement curves of PDMA S0.1/S0.1 lap joint illustrating lap-shear test reproducibility. All gel ribbons were cut from the same plate. Bulk failure outside the joint was systematically observed.
Extended Data Figure 2 Measurement of adhesion energy of PDMA S0.1 gels.
a, Lap-shear test geometry in which interfacial failure by peeling was observed for S0.1 gel ribbons glued by spreading 6 μl of TM-50 silica solution (l = 5 mm, w = 2 mm and h = 5 mm). b, Force–displacement curves for PDMA S0.1/S0.1 lap joints. Adhesive failure by interfacial peeling was observed. All gel ribbons were cut from the same gel plate and the tensile modulus was measured to be E ≈ 8.1 ± 1.0 kPa (error is s.d.). From the measured failure force, the adhesion energy can be estimated to be 6.2 ± 1.4 J m−2 (error is s.d.).
Extended Data Figure 3 Lap-shear test of PDMA S0.1 gels glued by various particle solutions.
In order of increasing deformation at the breaking point are shown the adhesive failure by interfacial peeling in CNT-Thy (grey), CNC1 (green), SM-30 (yellow) and HS-40 (purple). Fracture outside the junction occurred for TM-50 (blue) and AL-30 (red). Lap-joint dimensions were l = 5 mm, w = 5 mm and h = 2 mm. 6 μl of solution was spread to make the junction.
Extended Data Figure 4 Gluing gels of different stiffness or chemical nature.
a, Force–displacement curve for an assembly made of soft PDMA S0.1 and rigid PDMA S1.5 (red triangles) gels glued by TM-50 solution. For comparison the results obtained under identical conditions for the symmetric PDMA S0.1/S0 assembly are plotted (blue circles). Lap-joint dimensions were l = 10 mm, w = 5 mm and h = 2 mm. 15 μl of TM-50 solution was spread to make the junction. b, Glued at their preparation state, both PDMA S0.1 and PDMA S1.5 gels initially had the same size (diameter of about 10 mm). The picture shows gels after 5 h of swelling in deionized water. The highly crosslinked PDMA S1.5 gel (top piece) is less swollen than the PDMA S0.1 gel (bottom piece). Interfacial stresses induced by heterogeneous overswelling considerably exceed the shear stresses applied in the mechanical lap-shear test of a and lead to interfacial failure during immersion and overswelling in water. De-bonding was slow. c, Lap-shear force–displacement trace for the gelatine and S0.1 PDMA gel assembly (green stars) glued by spreading TM-50 silica solution. The failure occurred outside the lap joint and the fatal crack propagated in tension mode.
Extended Data Figure 5 Gluing overswollen gels and overswelling glued gels.
a, Adhesion energy of joints made of PDMA S0.1 hydrogels swollen before being glued with AL-30 silica solutions to various degrees of swelling Q (in blue) and adhesion energy of joints made of S0.1 hydrogels glued with TM-50 silica solutions at as-synthesized swelling degree (Q0 ≈ 8.5) and after being immersed in water and swollen to reach the maximum, equilibrium swelling degree, Qe ≈ 41 (in red) (error bars are s.d.). When S0.1 gels were glued with AL-30 particles at the as-synthesized state, bulk failure systematically occurred outside the junction even when the joints were very short, narrow and thick. To induce peeling (interfacial failure) making cuts at the interface was necessary. b, Lap-shear test for PDMA S0.1/S0.1 assembly glued by TM-50 silica at the preparation state, Q0 (blue circles) and after swelling in water for three days and attaining maximum equilibrium swelling Qe (red circles). c, Mechanical behaviour of a nanocomposite sample that can be considered as a model of the interfacial adhesive layer. Engineering stress is plotted as a function of strain. The loading–unloading cycle at the gel preparation conditions (green line) shows a large hysteresis. At equilibrium swelling (red dashed line) the hysteresis and dissipation were very weak. Low dissipation at swelling equilibrium is responsible for a relatively weaker adhesion after immersion in water.
Supplementary information
Lap-shear test of adhesion for a PDMA S0.1 hydrogel assembly glued with TM-50 silica solution
Failure by fracture occurs outside the junctions (MOV 4958 kb)
Lap-shear test of adhesion for a PDMA S0.1 hydrogel swollen in water to maximum swelling degree Qe
The PDMA S0.1/S0.1 lap joint glued with TM-50 silica solution at as-synthesized conditions, immersed in water for two days to reach swelling equilibrium fails by interfacial peeling. (MOV 986 kb)
Rights and permissions
About this article
Cite this article
Rose, S., Prevoteau, A., Elzière, P. et al. Nanoparticle solutions as adhesives for gels and biological tissues. Nature 505, 382–385 (2014). https://doi.org/10.1038/nature12806
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12806
This article is cited by
-
Adhesive cryogel particles for bridging confined and irregular tissue defects
Military Medical Research (2023)
-
Self-healing hybrid hydrogels with sustained bioactive components release for guided bone regeneration
Journal of Nanobiotechnology (2023)
-
Designing nanohesives for rapid, universal, and robust hydrogel adhesion
Nature Communications (2023)
-
Modified bond-based peridynamic approach for modeling the thermoviscoelastic response of bimaterials with viscoelastic–elastic interface
Engineering with Computers (2023)
-
Aligned nanofibrous collagen membranes from fish swim bladder as a tough and acid-resistant suture for pH-regulated stomach perforation and tendon rupture
Biomaterials Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.