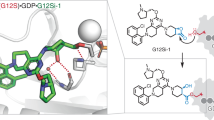
Abstract
Somatic mutations in the small GTPase K-Ras are the most common activating lesions found in human cancer, and are generally associated with poor response to standard therapies1,2,3. Efforts to target this oncogene directly have faced difficulties owing to its picomolar affinity for GTP/GDP4 and the absence of known allosteric regulatory sites. Oncogenic mutations result in functional activation of Ras family proteins by impairing GTP hydrolysis5,6. With diminished regulation by GTPase activity, the nucleotide state of Ras becomes more dependent on relative nucleotide affinity and concentration. This gives GTP an advantage over GDP7 and increases the proportion of active GTP-bound Ras. Here we report the development of small molecules that irreversibly bind to a common oncogenic mutant, K-Ras(G12C). These compounds rely on the mutant cysteine for binding and therefore do not affect the wild-type protein. Crystallographic studies reveal the formation of a new pocket that is not apparent in previous structures of Ras, beneath the effector binding switch-II region. Binding of these inhibitors to K-Ras(G12C) disrupts both switch-I and switch-II, subverting the native nucleotide preference to favour GDP over GTP and impairing binding to Raf. Our data provide structure-based validation of a new allosteric regulatory site on Ras that is targetable in a mutant-specific manner.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
Protein Data Bank
Data deposits
Atomic coordinates and structure factors for the reported crystal structures have been deposited with the Protein Data Bank (PDB), and accession numbers can be found in Extended Data Table 2.
References
Slebos, R. J. C. et al. K-ras oncogene activation as a prognostic marker in adenocarcinoma of the lung. N. Engl. J. Med. 323, 561–565 (1990)
Pao, W. et al. KRAS mutations and primary resistance of lung adenocarcinomas to gefitinib or erlotinib. PLoS Med. 2, e17 (2005)
Lièvre, A. et al. KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res. 66, 3992–3995 (2006)
John, J. et al. Kinetics of interaction of nucleotides with nucleotide-free H-ras p21. Biochemistry 29, 6058–6065 (1990)
Gibbs, J. B., Sigal, I. S., Poe, M. & Scolnick, E. M. Intrinsic GTPase activity distinguishes normal and oncogenic ras p21 molecules. Proc. Natl Acad. Sci. USA 81, 5704–5708 (1984)
Trahey, M. & McCormick, F. A cytoplasmic protein stimulates normal N-ras p21 GTPase, but does not affect oncogenic mutants. Science 238, 542–545 (1987)
Scherer, A. et al. Crystallization and preliminary X-ray analysis of the human c-H-ras-oncogene product p21 complexed with GTP analogues. J. Mol. Biol. 206, 257–259 (1989)
Erlanson, D. A. et al. Site-directed ligand discovery. Proc. Natl Acad. Sci. USA 97, 9367–9372 (2000)
Burlingame, M. A., Tom, C. T. M. B. & Renslo, A. R. Simple one-pot synthesis of disulfide fragments for use in disulfide-exchange screening. ACS Comb. Sci. 13, 205–208 (2011)
Sadowsky, J. D. et al. Turning a protein kinase on or off from a single allosteric site via disulfide trapping. Proc. Natl Acad. Sci. USA 108, 6056–6061 (2011)
Forbes, S. A. et al. The catalogue of somatic mutations in cancer (COSMIC). Curr. Protoc. Hum. Genet. 57, 10.11.1–10.11.26 (2008)
Bar-Sagi, D. A Ras by any other name. Mol. Cell. Biol. 21, 1441–1443 (2001)
Milburn, M. V. et al. Molecular switch for signal transduction: structural differences between active and inactive forms of protooncogenic ras proteins. Science 247, 939–945 (1990)
Taveras, A. G. et al. Ras oncoprotein inhibitors: the discovery of potent, ras nucleotide exchange inhibitors and the structural determination of a drug–protein complex. Bioorg. Med. Chem. 5, 125–133 (1997)
Naven, R. T., Kantesaria, S., Nadanaciva, S., Schroeter, T. & Leach, K. L. High throughput glutathione and Nrf2 assays to assess chemical and biological reactivity of cysteine-reactive compounds. Toxicol. Rev. 2, 235–244 (2013)
John, J. et al. Kinetic and structural analysis of the Mg2+-binding site of the guanine nucleotide-binding protein p21H-ras. J. Biol. Chem. 268, 923–929 (1993)
Feig, L. A. & Cooper, G. M. Inhibition of NIH 3T3 cell proliferation by a mutant ras protein with preferential affinity for GDP. Mol. Cell. Biol. 8, 3235–3243 (1988)
Farnsworth, C. L. & Feig, L. A. Dominant inhibitory mutations in the Mg2+-binding site of RasH prevent its activation by GTP. Mol. Cell. Biol. 11, 4822–4829 (1981)
Hall, B. E., Yang, S. S., Boriack-Sjodin, P. A., Kuriyan, J. & Bar-Sagi, D. Structure-based mutagenesis reveals distinct functions for Ras switch 1 and switch 2 in Sos-catalyzed guanine nucleotide exchange. J. Biol. Chem. 276, 27629–27637 (2001)
Pai, E. F. et al. Structure of the guanine-nucleotide-binding domain of the Ha-ras oncogene product p21 in the triphosphate conformation. Nature 341, 209–214 (1989)
Sung, Y.-J., Carter, M., Zhong, J.-M. & Hwang, Y.-W. Mutagenesis of the H-ras p21 at glycine-60 residue disrupts GTP-induced conformational change. Biochemistry 34, 3470–3477 (1995)
Hwang, M.-C. C., Sung, Y.-J. & Hwang, Y.-W. The differential effects of the Gly-60 to Ala mutation on the interaction of H-Ras p21 with different downstream targets. J. Biol. Chem. 271, 8196–8202 (1996)
Sunaga, N. et al. Knockdown of oncogenic KRAS in non-small cell lung cancers suppresses tumor growth and sensitizes tumor cells to targeted therapy. Mol. Cancer Ther. 10, 336–346 (2011)
Barbie, D. A. et al. Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature 462, 108–112 (2009)
Peyroche, A. et al. Brefeldin A acts to stabilize an abortive ARF–GDP–Sec7 domain protein complex. Mol. Cell 3, 275–285 (1999)
Nishimura, A. et al. Structural basis for the specific inhibition of heterotrimeric Gq protein by a small molecule. Proc. Natl Acad. Sci. USA 107, 13666–13671 (2010)
Maurer, T. et al. Small-molecule ligands bind to a distinct pocket in Ras and inhibit SOS-mediated nucleotide exchange activity. Proc. Natl Acad. Sci. USA 109, 5299–5304 (2012)
Sun, Q. et al. Discovery of small molecules that bind to K-Ras and inhibit Sos-mediated activation. Angew. Chem. 124, 6244–6247 (2012)
Shima, F. et al. In silico discovery of small-molecule Ras inhibitors that display antitumor activity by blocking the Ras-effector interaction. Proc. Natl Acad. Sci. USA 110, 8182–8187 (2013)
Ahmadian, M. R. et al. Guanosine triphosphatase stimulation of oncogenic Ras mutants. Proc. Natl Acad. Sci. USA 96, 7065–7070 (1999)
Acknowledgements
We are grateful to M. Burlingame and J. Sadowsky for assistance with the tethering screen; P. Ren and Y. Liu for assistance in chemical design and discussions; N. Younger for preparing several compounds; J. Kuriyan for sharing SOS and H-Ras constructs; F. McCormick and T. Yuan for discussion and sharing K-Ras reagents; R. Goody, K. Shannon and F. Wittinghofer for discussion. U.P. was supported by a postdoctoral fellowship of the Tobacco-related Disease Research Program (19FT-0069). The Advanced Light Source is supported by the Director, Office of Science, Office of Basic Energy Sciences, of the US Department of Energy under Contract No. DE-AC02-05CH11231. M.L.S. is a fellow of the International Association for the Study of Lung Cancer (IASLC) and receives a Young Investigator Award of the Prostate Cancer Foundation (PCF).
Author information
Authors and Affiliations
Contributions
J.M.O., U.P., J.A.W. and K.M.S. designed the study. J.M.O., U.P. and K.M.S. designed the molecules and wrote the manuscript. J.M.O. and U.P. performed the initial screen, synthesized the molecules and performed biochemical assays. U.P. expressed and purified the proteins and performed structural studies. J.M.O. and M.L.S. performed the cellular assays. J.M.O., U.P., M.L.S. and K.M.S performed analysis. All authors edited and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.M.O., U.P. and K.M.S. are joint inventors on a UC Regents-owned patent application covering these molecules, which has been licensed to Araxes Pharma LLC. J.M.O., U.P. and K.M.S. hold stock in and are consultants to Araxes Pharma LLC.
Extended data figures and tables
Extended Data Figure 1 Comparison of co-crystal structure of 6 with K-Ras(G12C) to known structures of Ras.
a, Compound 6 (cyan) bound in the S-IIP of K-Ras(G12C). b, Compound 6 (aligned and overlayed) with GDP-bound wild-type H-Ras showing groove near S-IIP (PDB accession 4Q21)13. c, Clash of compound 6 (aligned and overlayed) with GTPγS-bound K-Ras(G12D), which shows glycerol molecule adjacent to S-IIP (PDB accession 4DSO)27.
Extended Data Figure 2 Additional insights into Ras-compound binding and its biochemical effects.
a, Compound 6 (cyan) is attached to Cys 12 of K-Ras(G12C) and extends into an allosteric binding pocket beneath switch-II (blue), the S-IIP. The binding pocket in K-Ras (surface representation of the protein shown) fits 6 tightly and includes hydrophobic sub-pockets (dashed lines). An extension of the pocket is occupied by water molecules (red spheres) and might provide space for modified compound analogues. b–d, X-ray crystallographic studies of K-Ras(G12C) bound to several additional electrophilic analogues (14, 15 and 16, respectively) reveal a similar overall binding mode. All compounds follow a similar trajectory from Cys 12 into S-IIP but show some variability in the region of the piperidine/piperazine. The respective switch-I regions of the protein can be disordered. e, Overlay of the two different crystal forms of K-Ras(G12C) bound to 9 (space group C2 (grey) and P212121 (cyan)) is shown. The ligand orientation and conformation shows minimal changes, whereas switch-II of the protein appears disordered in the C2 form and atypical in the P212121 form. f, An overlay for several compounds including the disulphide 6 is shown (16-green, 6-yellow, 7-orange, 9-cyan). Key hydrophobic residues are labelled and hydrophobic interaction between the compounds and the (p-) or (o-) sub-pockets are indicated by dashed lines.
Extended Data Figure 3 Analysis of compound labelling rate and in vitro specificity.
a, Percentage modification of K-Ras(G12C) by compounds 9 and 12 over time (n = 3, error bars denote s.d.). b, Selective single labelling of K-Ras(G12C) by compound 12 in the presence of BSA. c, Quantitative single labelling of BSA and multiple labelling of K-Ras(G12C) by DTNB. d, Comparison of modification of K-Ras(G12C) and wild-type by 12 (n = 3, error bars denote s.d.).
Extended Data Figure 4 Comparison of active conformation and compound bound form of Ras.
a, X-ray crystal structure of the active conformation of H-Ras(G12C) with GMPPNP shows interactions of the γ-phosphate with key residues (Tyr 32, Thr 35 and Gly 60) that hold switch-I (red) and switch-II (blue) in place. The inactive GDP-bound structure of H-Ras(G12C) reveals the absence of these key interactions and increased distances between these residues and the position of the γ-phosphate (positions from GMPPNP structure indicated by spheres) coinciding with large conformational changes in both switch regions. In the P212121 crystal form of 9 bound to K-Ras(G12C) GDP switch-I is ordered (often disordered by compounds, see Extended Data Table 4), but the structure shows displacement of the γ-phosphate-binding residues beyond their positions in the inactive state. b, As indicated by the X-ray structures, removal of the γ-phosphate leads to relaxation of the ‘spring-loaded’ Ras-GTP back to the GDP state, with opening of switch-II. Compound binding moves switch-II even further away and interferes with GTP binding itself.
Extended Data Figure 5 Inhibitor sensitivity, K-Ras GTP levels and K-Ras dependency of lung cancer cell lines.
a, Percentage viability after treatment for 72 h with 12 relative to DMSO (n = 3 biological replicates, error bars denote s.e.m.). b, K-Ras GTP levels determined by incubating lysates with glutathione S-transferase (GST)-tagged RBD (Ras-binding domain of C-Raf) immobilized on glutathione beads (n = 3 biological replicates). c, Viability of cell lines evaluated 72 h after transfection with KRAS siRNA (n = 3 biological replicates). d, K-Ras immunoblot showing knockdown after KRAS siRNA (n = 3 biological replicates).
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data, additional references and Supplementary Table 1. (PDF 1175 kb)
Rights and permissions
About this article
Cite this article
Ostrem, J., Peters, U., Sos, M. et al. K-Ras(G12C) inhibitors allosterically control GTP affinity and effector interactions. Nature 503, 548–551 (2013). https://doi.org/10.1038/nature12796
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12796
This article is cited by
-
Another KRAS variant trapped
Nature Chemical Biology (2024)
-
KRAS allelic imbalance drives tumour initiation yet suppresses metastasis in colorectal cancer in vivo
Nature Communications (2024)
-
From bench to bedside: current development and emerging trend of KRAS-targeted therapy
Acta Pharmacologica Sinica (2024)
-
Strain-release alkylation of Asp12 enables mutant selective targeting of K-Ras-G12D
Nature Chemical Biology (2024)
-
The energetic and allosteric landscape for KRAS inhibition
Nature (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.