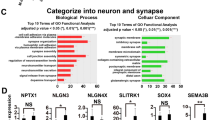
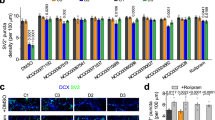
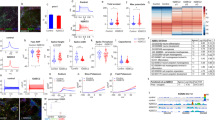
Abstract
Phelan–McDermid syndrome (PMDS) is a complex neurodevelopmental disorder characterized by global developmental delay, severely impaired speech, intellectual disability, and an increased risk of autism spectrum disorders (ASDs)1. PMDS is caused by heterozygous deletions of chromosome 22q13.3. Among the genes in the deleted region is SHANK3, which encodes a protein in the postsynaptic density (PSD)2,3. Rare mutations in SHANK3 have been associated with idiopathic ASDs4,5,6,7, non-syndromic intellectual disability8, and schizophrenia9. Although SHANK3 is considered to be the most likely candidate gene for the neurological abnormalities in PMDS patients10, the cellular and molecular phenotypes associated with this syndrome in human neurons are unknown. We generated induced pluripotent stem (iPS) cells from individuals with PMDS and autism and used them to produce functional neurons. We show that PMDS neurons have reduced SHANK3 expression and major defects in excitatory, but not inhibitory, synaptic transmission. Excitatory synaptic transmission in PMDS neurons can be corrected by restoring SHANK3 expression or by treating neurons with insulin-like growth factor 1 (IGF1). IGF1 treatment promotes formation of mature excitatory synapses that lack SHANK3 but contain PSD95 and N-methyl-d-aspartate (NMDA) receptors with fast deactivation kinetics. Our findings provide direct evidence for a disruption in the ratio of cellular excitation and inhibition in PMDS neurons, and point to a molecular pathway that can be recruited to restore it.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Phelan, K. & McDermid, H. E. The 22q13.3 deletion syndrome (Phelan-McDermid syndrome). Mol. Syndromol. 2, 186–201 (2012)
Boeckers, T. M., Bockmann, J., Kreutz, M. R. & Gundelfinger, E. D. ProSAP/Shank proteins – a family of higher order organizing molecules of the postsynaptic density with an emerging role in human neurological disease. J. Neurochem. 81, 903–910 (2002)
Sheng, M. & Kim, E. The Shank family of scaffold proteins. J. Cell Sci. 113, 1851–1856 (2000)
Boccuto, L. et al. Prevalence of SHANK3 variants in patients with different subtypes of autism spectrum disorders. Eur. J. Hum. Genet. 21, 310–316 (2013)
Durand, C. M. et al. Mutations in the gene encoding the synaptic scaffolding protein SHANK3 are associated with autism spectrum disorders. Nature Genet. 39, 25–27 (2007)
Gauthier, J. et al. Novel de novo SHANK3 mutation in autistic patients. Am. J. Med. Genet. B. Neuropsychiatr. Genet. 150B, 421–424 (2009)
Moessner, R. et al. Contribution of SHANK3 mutations to autism spectrum disorder. Am. J. Hum. Genet. 81, 1289–1297 (2007)
Hamdan, F. F. et al. Excess of de novo deleterious mutations in genes associated with glutamatergic systems in nonsyndromic intellectual disability. Am. J. Hum. Genet. 88, 306–316 (2011)
Gauthier, J. et al. De novo mutations in the gene encoding the synaptic scaffolding protein SHANK3 in patients ascertained for schizophrenia. Proc. Natl Acad. Sci. USA 107, 7863–7868 (2010)
Wilson, H. L. et al. Molecular characterisation of the 22q13 deletion syndrome supports the role of haploinsufficiency of SHANK3/PROSAP2 in the major neurological symptoms. J. Med. Genet. 40, 575–584 (2003)
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007)
Yazawa, M. et al. Using induced pluripotent stem cells to investigate cardiac phenotypes in Timothy syndrome. Nature 471, 230–234 (2011)
Paşca, S. P. et al. Using iPSC-derived neurons to uncover cellular phenotypes associated with Timothy syndrome. Nature Med. 17, 1657–1662 (2011)
Yoo, A. S. et al. MicroRNA-mediated conversion of human fibroblasts to neurons. Nature 476, 228–231 (2011)
Dittgen, T. et al. Lentivirus-based genetic manipulations of cortical neurons and their optical and electrophysiological monitoring in vivo. Proc. Natl Acad. Sci. USA 101, 18206–18211 (2004)
Nathanson, J. L., Yanagawa, Y., Obata, K. & Callaway, E. M. Preferential labeling of inhibitory and excitatory cortical neurons by endogenous tropism of adeno-associated virus and lentivirus vectors. Neuroscience 161, 441–450 (2009)
Akhtar, M. W. et al. Histone deacetylases 1 and 2 form a developmental switch that controls excitatory synapse maturation and function. J. Neurosci. 29, 8288–8297 (2009)
Maunakea, A. K. et al. Conserved role of intragenic DNA methylation in regulating alternative promoters. Nature 466, 253–257 (2010)
Rinaldi, T., Kulangara, K., Antoniello, K. & Markram, H. Elevated NMDA receptor levels and enhanced postsynaptic long-term potentiation induced by prenatal exposure to valproic acid. Proc. Natl Acad. Sci. USA 104, 13501–13506 (2007)
Goold, C. P. & Nicoll, R. A. Single-cell optogenetic excitation drives homeostatic synaptic depression. Neuron 68, 512–528 (2010)
Marchetto, M. C. et al. A model for neural development and treatment of Rett syndrome using human induced pluripotent stem cells. Cell 143, 527–539 (2010)
O’Kusky, J. R., Ye, P. & D’Ercole, A. J. Insulin-like growth factor-I promotes neurogenesis and synaptogenesis in the hippocampal dentate gyrus during postnatal development. J. Neurosci. 20, 8435–8442 (2000)
Tropea, D. et al. Partial reversal of Rett syndrome-like symptoms in MeCP2 mutant mice. Proc. Natl Acad. Sci. USA 106, 2029–2034 (2009)
Chen, D. Y. et al. A critical role for IGF-II in memory consolidation and enhancement. Nature 469, 491–497 (2011)
Bozdagi, O. et al. Haploinsufficiency of the autism-associated Shank3 gene leads to deficits in synaptic function, social interaction, and social communication. Mol. Autism 1, 15 (2010)
Peça, J. et al. Shank3 mutant mice display autistic-like behaviours and striatal dysfunction. Nature 472, 437–442 (2011)
Wang, X. et al. Synaptic dysfunction and abnormal behaviors in mice lacking major isoforms of Shank3. Hum. Mol. Genet. 20, 3093–3108 (2011)
Paoletti, P., Bellone, C. & Zhou, Q. NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and disease. Nature Rev. Neurosci. 14, 383–400 (2013)
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nature Biotechnol. 27, 275–280 (2009)
Gaspard, N. et al. Generation of cortical neurons from mouse embryonic stem cells. Nature Protocols 4, 1454–1463 (2009)
Bellone, C. & Nicoll, R. A. Rapid bidirectional switching of synaptic NMDA receptors. Neuron 55, 779–785 (2007)
Sommer, C. A. et al. Induced pluripotent stem cell generation using a single lentiviral stem cell cassette. Stem Cells 27, 543–549 (2009)
Acknowledgements
We are grateful to participants and their families for their support; M. Adam for assistance with recruitment; to X. Jia, A. Cherry, C. Bangs, P. Jones, and J. Williams for assistance with tissue culture; P. Liao for help with multiplex ligation-dependent probe amplification (MLPA); M. Fabian for astrocyte preparations; H.N. Nguyen for consultations on the neural differentiation protocol and spectral karyotyping (SKY); V. Vu and G. Lin for help with data analysis; T. Sudhof, T. Boeckers, A. Grabruker, C. Garner and C. Sala for antibodies; R. Xavier for SHANK3 complementary DNA; R. Reijo-Pera and members of the Dolmetsch laboratory for commenting on the manuscript; E. Nigh for editing the manuscript. We also thank the Stanford Neuroscience Microscopy Service (supported by National Institutes of Health (NIH) NS069375). Support for this study came from the California Institute for Regenerative Medicine CIRM, the Autism Science Foundation and the Phelan-McDermid Syndrome Foundation (to A.S.), the Swiss National Science Foundation (to T.P.), the Japan Society for the Promotion of Research Abroad and American Heart Association (to M.Y.), the National Institute of Mental Health (NIMH) grant R33MH087898 (to J.F.H.); NIH Pioneer Award (5DP1OD3889), CIRM (grant RT2-01906) and Simons Foundation (to R.E.D.). We are also grateful for funding from the JDH research fund, N. Juaw, B. and F. Horowitz, M. McCafferey, B. and J. Packard, P. Kwan and K. Wang, and the Flora foundation.
Author information
Authors and Affiliations
Contributions
A.S. and R.E.D. designed experiments and wrote the manuscript; A.S. performed iPS cell maintenance, neural differentiation, electrophysiology, cloning and immunocytochemistry; O.S. maintained and characterized iPS cells, performed western blot and qRT–PCR; M.Y. generated and characterized iPS cells; T.P. performed multiplex single-cell qRT–PCR; V.S. performed teratoma assay; R.S. and A.K. performed qRT–PCR and data analysis; W.F., J.A.B. and J.F.H. recruited and characterized patients and performed the MLPA assay.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
This file contains Supplementary Figures 1-16 Supplementary Tables 1-5 and additional references. (PDF 9072 kb)
Rights and permissions
About this article
Cite this article
Shcheglovitov, A., Shcheglovitova, O., Yazawa, M. et al. SHANK3 and IGF1 restore synaptic deficits in neurons from 22q13 deletion syndrome patients. Nature 503, 267–271 (2013). https://doi.org/10.1038/nature12618
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12618
This article is cited by
-
Deficiency of FRMD5 results in neurodevelopmental dysfunction and autistic-like behavior in mice
Molecular Psychiatry (2024)
-
Disruption of DDX53 coding sequence has limited impact on iPSC-derived human NGN2 neurons
BMC Medical Genomics (2023)
-
An IGFBP2-derived peptide promotes neuroplasticity and rescues deficits in a mouse model of Phelan-McDermid syndrome
Molecular Psychiatry (2023)
-
Defects in early synaptic formation and neuronal function in Prader-Willi syndrome
Scientific Reports (2023)
-
Insulin-Like Growth Factor-1 Promotes Synaptogenesis Signaling, a Major Dysregulated Pathway in Malformation of Cortical Development, in a Rat Model
Molecular Neurobiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.