Abstract
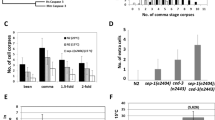
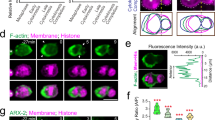
During animal development, the proper regulation of apoptosis requires the precise spatial and temporal execution of cell-death programs, which can include both caspase-dependent and caspase-independent pathways1,2. Although the mechanisms of caspase-dependent and -independent cell killing have been examined extensively, how these pathways are coordinated within a single cell that is fated to die is unknown. Here we show that the Caenorhabditis elegans Sp1 transcription factor SPTF-3 specifies the programmed cell deaths of at least two cells—the sisters of the pharyngeal M4 motor neuron and the AQR sensory neuron—by transcriptionally activating both caspase-dependent and -independent apoptotic pathways. SPTF-3 directly drives the transcription of the gene egl-1, which encodes a BH3-only protein that promotes apoptosis through the activation of the CED-3 caspase3. In addition, SPTF-3 directly drives the transcription of the AMP-activated protein kinase-related gene pig-1, which encodes a protein kinase and functions in apoptosis of the M4 sister and AQR sister independently of the pathway that activates CED-3 (refs 4, 5). Thus, a single transcription factor controls two distinct cell-killing programs that act in parallel to drive apoptosis. Our findings reveal a bivalent regulatory node for caspase-dependent and -independent pathways in the regulation of cell-type-specific apoptosis. We propose that such nodes might act as features of a general mechanism for regulating cell-type-specific apoptosis and could be therapeutic targets for diseases involving the dysregulation of apoptosis through multiple cell-killing mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fuchs, Y. & Steller, H. Programmed cell death in animal development and disease. Cell 147, 742–758 (2011)
Yuan, J. & Kroemer, G. Alternative cell death mechanisms in development and beyond. Genes Dev. 24, 2592–2602 (2010)
Metzstein, M. M., Stanfield, G. M. & Horvitz, H. R. Genetics of programmed cell death in C. elegans: past, present and future. Trends Genet. 14, 410–416 (1998)
Cordes, S., Frank, C. A. & Garriga, G. The C. elegans MELK ortholog PIG-1 regulates cell size asymmetry and daughter cell fate in asymmetric neuroblast divisions. Development 133, 2747–2756 (2006)
Denning, D. P., Hatch, V. & Horvitz, H. R. Programmed elimination of cells by caspase-independent cell extrusion in C. elegans. Nature 488, 226–230 (2012)
Avery, L. & Horvitz, H. R. A cell that dies during wild-type C. elegans development can function as a neuron in a ced-3 mutant. Cell 51, 1071–1078 (1987)
Sulston, J. E., Schierenberg, E., White, J. G. & Thomson, J. N. The embryonic cell lineage of the nematode Caenorhabditis elegans. Dev. Biol. 100, 64–119 (1983)
Hirose, T., Galvin, B. D. & Horvitz, H. R. Six and Eya promote apoptosis through direct transcriptional activation of the proapoptotic BH3-only gene egl-1 in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 107, 15479–15484 (2010)
Ulm, E. A., Sleiman, S. F. & Chamberlin, H. M. Developmental functions for the Caenorhabditis elegans Sp protein SPTF-3. Mech. Dev. 128, 428–441 (2011)
Hengartner, M. O., Ellis, R. E. & Horvitz, H. R. Caenorhabditis elegans gene ced-9 protects cells from programmed cell death. Nature 356, 494–499 (1992)
Yochem, J., Gu, T. & Han, M. A new marker for mosaic analysis in Caenorhabditis elegans indicates a fusion between hyp6 and hyp7, two major components of the hypodermis. Genetics 149, 1323–1334 (1998)
Maduro, M. & Pilgrim, D. Identification and cloning of unc-119, a gene expressed in the Caenorhabditis elegans nervous system. Genetics 141, 977–988 (1995)
Smith, C. C. T. et al. Necrostatin: a potentially novel cardioprotective agent? Cardiovasc. Drugs Ther. 21, 227–233 (2007)
Rosenbaum, D. M. et al. Necroptosis, a novel form of caspase-independent cell death, contributes to neuronal damage in a retinal ischemia-reperfusion injury model. J. Neurosci. Res. 88, 1569–1576 (2010)
Hyman, B. T. & Yuan, J. Apoptotic and non-apoptotic roles of caspases in neuronal physiology and pathophysiology. Nature Rev. Neurosci. 13, 395–406 (2012)
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974)
Wicks, S. R., Yeh, R. T., Gish, W. R., Waterston, R. H. & Plasterk, R. H. Rapid gene mapping in Caenorhabditis elegans using a high density polymorphism map. Nature Genet. 28, 160–164 (2001)
Ercan, S. et al. X chromosome repression by localization of the C. elegans dosage compensation machinery to sites of transcription initiation. Nature Genet. 39, 403–408 (2007)
Marson, A. et al. Connecting microRNA genes to the core transcriptional regulatory circuitry of embryonic stem cells. Cell 134, 521–533 (2008)
Bailey, T. L. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 37, W202–W208 (2009)
Bailey, T. L. & Gribskov, M. Combining evidence using p-values: application to sequence homology searches. Bioinformatics 14, 48–54 (1998)
Beissbarth, T. & Speed, T. P. GOstat: find statistically overrepresented Gene Ontologies within a group of genes. Bioinformatics 20, 1464–1465 (2004)
Conradt, B. & Horvitz, H. R. The C. elegans protein EGL-1 is required for programmed cell death and interacts with the Bcl-2-like protein CED-9. Cell 93, 519–529 (1998)
Mello, C. C., Kramer, J. M., Stinchcomb, D. & Ambros, V. Efficient gene transfer in C. elegans: extrachromosomal maintenance and integration of transforming sequences. EMBO J. 10, 3959–3970 (1991)
Clark, S. G., Lu, X. & Horvitz, H. R. The Caenorhabditis elegans locus lin-15, a negative regulator of a tyrosine kinase signaling pathway, encodes two different proteins. Genetics 137, 987–997 (1994)
Herman, M. A., Vassilieva, L. L., Horvitz, H. R., Shaw, J. E. & Herman, R. K. The C. elegans gene lin-44, which controls the polarity of certain asymmetric cell divisions, encodes a Wnt protein and acts cell nonautonomously. Cell 83, 101–110 (1995)
Frøkjær-Jensen, C. et al. Single-copy insertion of transgenes in Caenorhabditis elegans. Nature Genet. 40, 1375–1383 (2008)
Andersen, E. C., Saffer, A. M. & Horvitz, H. R. Multiple levels of redundant processes inhibit Caenorhabditis elegans vulval cell fates. Genetics 179, 2001–2012 (2008)
Acknowledgements
We thank A. Fire, J. Gaudet, C. Barbara and Y. Iino for reporter constructs used to observe cell-type-specific apoptosis; G. Garriga for pig-1 strains; the Caenorhabditis Genetic Center, which is funded by the NIH Office of Research Infrastructure Programs (P40 OD010440) and the National BioResource project for strains; D. Denning, K. Boulias, A. Corrionero and H. Johnsen for comments about the manuscript; and members of the Horvitz laboratory for technical support and discussions. This work was supported by the Howard Hughes Medical Institute. T.H. was supported in part by the Ministry of Education, Science, Technology, Sports and Culture of Japan. H.R.H. is the David H. Koch Professor of Biology at the Massachusetts Institute of Technology and an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
T.H. and H.R.H. designed the experiments, analysed the data and wrote the manuscript. T.H. performed the experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-12 and Supplementary Tables 3-4 (see separate files for Supplementary-Tables 1-2). (PDF 4109 kb)
Supplementary Table 1
A list of SPTF-3-bound regions obtained from ChIP-seq experiments using the anti- SPTF-3 antibody N81. (XLSX 281 kb)
Supplementary Table 2
A list of SPTF-3-bound regions obtained from ChIP-seq experiments using the anti-SPTF-3 antibody M82. (XLSX 210 kb)
Rights and permissions
About this article
Cite this article
Hirose, T., Horvitz, H. An Sp1 transcription factor coordinates caspase-dependent and -independent apoptotic pathways. Nature 500, 354–358 (2013). https://doi.org/10.1038/nature12329
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12329
This article is cited by
-
Sp1 is overexpressed and associated with progression and poor prognosis in bladder urothelial carcinoma patients
International Urology and Nephrology (2022)
-
Harnessing the power of genetics: fast forward genetics in Caenorhabditis elegans
Molecular Genetics and Genomics (2021)
-
DNA methylation status of TBX20 in patients with tetralogy of Fallot
BMC Medical Genomics (2019)
-
Comparative genetic, proteomic and phosphoproteomic analysis of C. elegans embryos with a focus on ham-1/STOX and pig-1/MELK in dopaminergic neuron development
Scientific Reports (2017)
-
Both the apoptotic suicide pathway and phagocytosis are required for a programmed cell death in Caenorhabditis elegans
BMC Biology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.