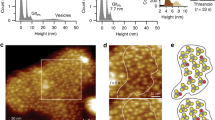
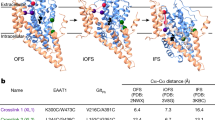
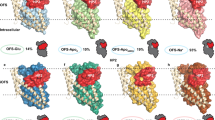
Abstract
Glutamate transporters are integral membrane proteins that catalyse neurotransmitter uptake from the synaptic cleft into the cytoplasm of glial cells and neurons1. Their mechanism of action involves transitions between extracellular (outward)-facing and intracellular (inward)-facing conformations, whereby substrate binding sites become accessible to either side of the membrane2. This process has been proposed to entail transmembrane movements of three discrete transport domains within a trimeric scaffold3. Using single-molecule fluorescence resonance energy transfer (smFRET) imaging4, we have directly observed large-scale transport domain movements in a bacterial homologue of glutamate transporters. We find that individual transport domains alternate between periods of quiescence and periods of rapid transitions, reminiscent of bursting patterns first recorded in single ion channels using patch-clamp methods5,6. We propose that the switch to the dynamic mode in glutamate transporters is due to separation of the transport domain from the trimeric scaffold, which precedes domain movements across the bilayer. This spontaneous dislodging of the substrate-loaded transport domain is approximately 100-fold slower than subsequent transmembrane movements and may be rate determining in the transport cycle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Danbolt, N. C. Glutamate uptake. Prog. Neurobiol. 65, 1–105 (2001)
Krishnamurthy, H., Piscitelli, C. L. & Gouaux, E. Unlocking the molecular secrets of sodium-coupled transporters. Nature 459, 347–355 (2009)
Reyes, N., Ginter, C. & Boudker, O. Transport mechanism of a bacterial homologue of glutamate transporters. Nature 462, 880–885 (2009)
Weiss, S. Fluorescence spectroscopy of single biomolecules. Science 283, 1676–1683 (1999)
Sakmann, B., Patlak, J. & Neher, E. Single acetylcholine-activated channels show burst-kinetics in presence of desensitizing concentrations of agonist. Nature 286, 71–73 (1980)
Cull-Candy, S. G. & Parker, I. Rapid kinetics of single glutamate-receptor channels. Nature 295, 410–412 (1982)
Tzingounis, A. V. & Wadiche, J. I. Glutamate transporters: confining runaway excitation by shaping synaptic transmission. Nature Rev. Neurosci. 8, 935–947 (2007)
Zerangue, N. & Kavanaugh, M. P. Flux coupling in a neuronal glutamate transporter. Nature 383, 634–637 (1996)
Yernool, D., Boudker, O., Jin, Y. & Gouaux, E. Structure of a glutamate transporter homologue from Pyrococcus horikoshii. Nature 431, 811–818 (2004)
Boudker, O. et al. Coupling substrate and ion binding to extracellular gate of a sodium-dependent aspartate transporter. Nature 445, 387–393 (2007)
Boudker, O. & Verdon, G. Structural perspectives on secondary active transporters. Trends Pharmacol. Sci. 31, 418–426 (2010)
Ryan, R. M., Compton, E. L. & Mindell, J. A. Functional characterization of a Na+-dependent aspartate transporter from Pyrococcus horikoshii. J. Biol. Chem. 284, 17540–17548 (2009)
Blanchard, S. C. Single-molecule observations of ribosome function. Curr. Opin. Struct. Biol. 19, 103–109 (2009)
Groeneveld, M. & Slotboom, D. J. Rigidity of the subunit interfaces of the trimeric glutamate transporter GltT during translocation. J. Mol. Biol. 372, 565–570 (2007)
Reyes, N., Oh, S. & Boudker, O. Binding thermodynamics of a glutamate transporter homolog. Nature Struct. Mol. Biol. 20, 634–640 (2013)
Groeneveld, M. & Slotboom, D. J. Na+:aspartate coupling stoichiometry in the glutamate transporter homologue GltPh . Biochemistry 49, 3511–3513 (2010)
Roy, R., Hohng, S. & Ha, T. A practical guide to single-molecule FRET. Nature Methods 5, 507–516 (2008)
Dave, R., Terry, D. S., Munro, J. B. & Blanchard, S. C. Mitigating unwanted photophysical processes for improved single-molecule fluorescence imaging. Biophys. J. 96, 2371–2381 (2009)
Verdon, G. & Boudker, O. Crystal structure of an asymmetric trimer of a bacterial glutamate transporter homolog. Nature Struct. Mol. Biol. 19, 355–357 (2012)
McKinney, S. A., Joo, C. & Ha, T. Analysis of single-molecule FRET trajectories using hidden Markov modeling. Biophys. J. 91, 1941–1951 (2006)
Grewer, C. et al. Individual subunits of the glutamate transporter EAAC1 homotrimer function independently of each other. Biochemistry 44, 11913–11923 (2005)
Shimamoto, K. et al. dl-threo-β-benzyloxyaspartate, a potent blocker of excitatory amino acid transporters. Mol. Pharmacol. 53, 195–201 (1998)
Liu, S., Bokinsky, G., Walter, N. G. & Zhuang, X. Dissecting the multistep reaction pathway of an RNA enzyme by single-molecule kinetic “fingerprinting”. Proc. Natl Acad. Sci. USA 104, 12634–12639 (2007)
Stolzenberg, S., Khelashvili, G. & Weinstein, H. Structural intermediates in a model of the substrate translocation path of the bacterial glutamate transporter homologue GltPh . J. Phys. Chem. B 116, 5372–5383 (2012)
Fairman, W. A. et al. An excitatory amino-acid transporter with properties of a ligand-gated chloride channel. Nature 375, 599–603 (1995)
Wadiche, J. I., Amara, S. G. & Kavanaugh, M. P. Ion fluxes associated with excitatory amino acid transport. Neuron 15, 721–728 (1995)
Grewer, C., Watzke, N., Wiessner, M. & Rauen, T. Glutamate translocation of the neuronal glutamate transporter EAAC1 occurs within milliseconds. Proc. Natl Acad. Sci. USA 97, 9706–9711 (2000)
Otis, T. S. & Kavanaugh, M. P. Isolation of current components and partial reaction cycles in the glial glutamate transporter EAAT2. J. Neurosci. 20, 2749–2757 (2000)
Blanchard, S. C. et al. tRNA dynamics on the ribosome during translation. Proc. Natl Acad. Sci. USA 101, 12893–12898 (2004)
Zhao, Y. et al. Single-molecule dynamics of gating in a neurotransmitter transporter homologue. Nature 465, 188–193 (2010)
Munro, J. B., Altman, R. B., O’Connor, N. & Blanchard, S. C. Identification of two distinct hybrid state intermediates on the ribosome. Mol. Cell 25, 505–517 (2007)
Qin, F., Auerbach, A. & Sachs, F. A direct optimization approach to hidden Markov modeling for single channel kinetics. Biophys. J. 79, 1915–1927 (2000)
Qin, F. Restoration of single-channel currents using the segmental k-means method based on hidden Markov modeling. Biophys. J. 86, 1488–1501 (2004)
Motulsky, H. & Christopoulos, A. Fitting Models to Biological Data Using Linear and Nonlinear Regression: A Practical Guide to Curve Fitting 256–311 (Oxford Univ. Press, 2004)
Sigworth, F. J. & Sine, S. M. Data transformations for improved display and fitting of single-channel dwell time histograms. Biophys. J. 52, 1047–1054 (1987)
Acknowledgements
We would like to thank D. S. Terry for his help with the design of smFRET experiments and discussions; Z. Zhou for the synthesis of cyanine fluorophores; E. Georgieva for initial biochemical characterization of the single-cysteine mutants; G. Verdon, A. Accardi and N. Reyes for helpful discussions and comments on the manuscript. The work was supported in part by the National Institute of Health grants 5U54GM087519 and R01NS064357.
Author information
Authors and Affiliations
Contributions
N.A. purified GltPh mutants, carried out the experiments and analysed the data. R.B.A. prepared reagents for smFRET experiments. N.A., O.B. and S.C.B. together designed, analysed and interpreted the experiments and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information file contains Supplementary Figures 1-10, Supplementary Methods, Supplementary Tables 1-2, Supplementary Discussion and Supplementary References. (PDF 3709 kb)
Rights and permissions
About this article
Cite this article
Akyuz, N., Altman, R., Blanchard, S. et al. Transport dynamics in a glutamate transporter homologue. Nature 502, 114–118 (2013). https://doi.org/10.1038/nature12265
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12265
This article is cited by
-
Regulation of the mammalian-brain V-ATPase through ultraslow mode-switching
Nature (2022)
-
Kinetic mechanism of Na+-coupled aspartate transport catalyzed by GltTk
Communications Biology (2021)
-
Millisecond dynamics of an unlabeled amino acid transporter
Nature Communications (2020)
-
Amino Acid Transporters and Exchangers from the SLC1A Family: Structure, Mechanism and Roles in Physiology and Cancer
Neurochemical Research (2020)
-
Quantifying secondary transport at single-molecule resolution
Nature (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.