Abstract
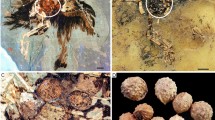
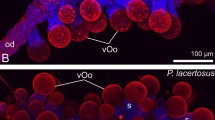
The two groups of archosaurs, crocodilians and birds, form an extant phylogenetic bracket for understanding the reproductive behaviour of dinosaurs. This behaviour is inferred from preserved nests and eggs, and even gravid individuals1. Data indicate that many ‘avian’ traits were already present in Paraves—the clade that includes birds and their close relatives—and that the early evolution of the modern avian form of reproduction was already well on its way2,3. Like living neornithine birds, non-avian maniraptorans had daily oviposition and asymmetrical eggs with complex shell microstructure, and were known to protect their clutches4,5,6. However, like crocodilians, non-avian maniraptorans had two active oviducts (one present in living birds), relatively smaller eggs, and may not have turned their eggs in the way that living birds do1,6. Here we report on the first discovery of fossilized mature or nearly mature ovarian follicles, revealing a previously undocumented stage in dinosaur reproduction: reproductively active females near ovulation. Preserved in a specimen of the long bony-tailed Jeholornis and two enantiornithine birds from the Early Cretaceous period lacustrine Jehol Biota in northeastern China, these discoveries indicate that basal birds only had one functional ovary, but retained primitive morphologies as a result of their lower metabolic rate relative to living birds. They also indicate that basal birds reached sexual maturity before skeletal maturity, as in crocodiles and paravian dinosaurs. Differences in follicular morphology between Jeholornis and the enantiornithines are interpreted as forming an evolutionary gradient from the reproductive condition in paravian dinosaurs towards neornithine birds. Furthermore, differences between the two enantiornithines indicate that this lineage might also have evolved advanced reproductive traits in parallel to the neornithine lineage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Zelenitsky, D. in Proceedings of the 2006 Goseong International Dinosaur Symposium (ed Lee, Y.-N. ) Vol. 22, 209–216 (J. Paleontol. Soc. Korea, 2006)
Zhou, Z. & Zhang, F. A precocial avian embryo from the Lower Cretaceous of China. Science 306, 653 (2004)
Chiappe, L. M., Ji, S. & Ji, Q. Juvenile birds from the Early Cretaceous of China: implications for enantiornithine ontogeny. Am. Mus. Novit. 3594, 1–46 (2007)
Norell, M. A., Clark, J. M., Chiappe, L. M. & Dashzeveg, D. A nesting dinosaur. Nature 378, 774–776 (1995)
Sato, T., Chang, Y.-N., Wu, X.-C., Zelenitsky, D. A. & Hsiao, Y.-F. A pair of shelled eggs inside a female dinosaur. Science 308, 375 (2005)
Varricchio, D. J., Jackson, F., Borkowski, J. J. & Horner, J. R. Nest and egg clutches of the dinosaur Troodon formosus and the evolution of avian reproductive traits. Nature 385, 247–250 (1997)
Gilbert, A. B. in Form and Function in Birds (eds King, A.S. & McLelland, J. ) 237–360 (Academic, 1979)
O’Connor, J. K. A Systematic Review of Enantiornithes (Aves: Ornithothoraces). PhD thesis, Univ. Southern California (2009). 600 pp
O’Connor, J. K., Chiappe, L. M. & Bell, A. in Living Dinosaurs: the Evolutionary History of Birds (eds Dyke, G. D. & Kaiser, G. ) 39–114 (J. Wiley & Sons, 2011)
Zhou, Z. & Zhang, F. A long-tailed, seed-eating bird from the Early Cretaceous of China. Nature 418, 405–409 (2002)
Zheng, X. et al. Fossil evidence of avian crops from the Early Cretaceous of China. Proc. Natl Acad. Sci. USA 108, 15904–15907 (2011)
Clark, J. M., Norell, M. A. & Chiappe, L. M. An oviraptorid skeleton from the Late Cretaceous of Ukhaa Tolgod, Mongolia, preserved in an avianlike brooding position over an oviraptorid nest. Am. Mus. Novit. 3265, 1–36 (1999)
Girling, J. E. The reptilian oviduct: a review of structure and function and directions for future research. J. Exp. Zool. 293, 141–170 (2002)
Gill, F. B. Ornithology 3rd edn 399–430 (W. H. Freeman, 2007)
Lance, V. A. Reproductive cycle of the American Alligator. Am. Zool. 29, 999–1018 (1989)
Schweitzer, M. H., Wittmeyer, J. L. & Horner, J. R. Gender-specific reproductive tissue in ratites and Tyrannosaurus rex . Science 308, 1456–1460 (2005)
Schweitzer, M. H., Elsey, R. M., Dacke, C. G., Horner, J. R. & Lamm, E.-T. Do egg-laying crocodilian (Alligator mississippiensis) archosaurs form medullary bone? Bone 40, 1152–1158 (2007)
Richardson, K. C., Webb, G. J. W. & Manolis, S. C. Crocodiles: Inside Out 1st edn, 96–104 (Surrey Beatty & Sons, 2002)
Thorbjarnarson, J. B. Reproductive characteristics of the order Crocodylia. Herpetologica 52, 8–24 (1996)
Taylor, T. G. How an eggshell is made. Sci. Am. 222, 88–95 (1970)
Lü, J.-C. et al. An egg-adult association, gender, and reproduction in pterosaurs. Science 331, 321–324 (2011)
Astheimer, L. B., Manolis, S. C. & Grau, C. R. Egg formation in crocodiles: avian affinities in yolk deposition. Copeia 1989, 221–224 (1989)
Erickson, G. M. et al. Was dinosaurian physiology inherited by birds? Reconciling slow growth in Archaeopteryx. PLoS ONE 4, e7390 (2009)
Farlow, J. O., Hurlburt, G. R., Elsey, R. M., Britton, A. R. C. & Langston, W., Jr Femoral dimensions and body size of Alligator mississippiensis: estimating the size of extinct mesoeucrocodylians. J. Vertebr. Paleontol. 25, 354–369 (2005)
Liu, D., Zhou, Z.-H. & Zhang, Y.-G. Mass estimate and evolutionary trend in Chinese Mesozoic fossil birds. Vertebr. Palasiat. 50, 39–52 (2012)
Godfray, H. C. J., Partridge, L. & Harvey, P. H. Clutch size. Annu. Rev. Ecol. Syst. 22, 409–429 (1991)
Varricchio, D. J. et al. Avian paternal care had dinosaur origin. Science 322, 1826–1828 (2008)
Erickson, G. M., Curry Rogers, K., Varricchio, D. J., Norell, M. A. & Xu, X. Growth patterns in brooding dinosaurs reveals the timing of sexual maturity in non-avian dinosaurs and genesis of the avian condition. Biol. Lett. 3, 558–561 (2007)
Chiappe, L. M., Marugán-Lobón, J., Ji, S. & Zhou, Z. Life history of a basal bird: morphometrics of the Early Cretaceous Confuciusornis . Biol. Lett. 4, 719–723 (2008)
Acknowledgements
The specimens reported here are deposited in the Tianyu Natural History Museum, Shandong, China. We thank A. Shi for helping with illustrations, S. Zhang for help with the histological analysis and E. O’Connor for estimating body mass. The research was supported by the National Basic Research Program of China (973 Program, 2012CB821906), the National Natural Science Foundation of China (41172020) and the Chinese Academy of Sciences.
Author information
Authors and Affiliations
Contributions
X.Z., J.O’C. and Z.Z. designed the project. J.O’C., F.H., Z.Z., X.W., M.W. and Y.W. performed the research. J.O’C., F.H. and Z.Z. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Specimen identifications; Supplementary Figures 1-8, including full interpretative drawings of all specimens and counterslab photographs, Supplementary Figures 9-10 showing histological analyses; a Supplementary Discussion; Supplementary Tables 1-2 and Supplementary References. (PDF 3497 kb)
Rights and permissions
About this article
Cite this article
Zheng, X., O’Connor, J., Huchzermeyer, F. et al. Preservation of ovarian follicles reveals early evolution of avian reproductive behaviour. Nature 495, 507–511 (2013). https://doi.org/10.1038/nature11985
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11985
This article is cited by
-
Reanalysis of putative ovarian follicles suggests that Early Cretaceous birds were feeding not breeding
Scientific Reports (2020)
-
Confirmation of ovarian follicles in an enantiornithine (Aves) from the Jehol biota using soft tissue analyses
Communications Biology (2020)
-
Study on the Jehol Biota: Recent advances and future prospects
Science China Earth Sciences (2020)
-
An Early Cretaceous enantiornithine (Aves) preserving an unlaid egg and probable medullary bone
Nature Communications (2019)
-
Macrofossil evidence unveiling evolution of male cones in Ephedraceae (Gnetidae)
BMC Evolutionary Biology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.