Abstract
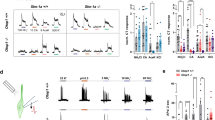
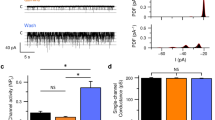
Recognition of sweet, bitter and umami tastes requires the non-vesicular release from taste bud cells of ATP, which acts as a neurotransmitter to activate afferent neural gustatory pathways1. However, how ATP is released to fulfil this function is not fully understood. Here we show that calcium homeostasis modulator 1 (CALHM1), a voltage-gated ion channel2,3, is indispensable for taste-stimuli-evoked ATP release from sweet-, bitter- and umami-sensing taste bud cells. Calhm1 knockout mice have severely impaired perceptions of sweet, bitter and umami compounds, whereas their recognition of sour and salty tastes remains mostly normal. Calhm1 deficiency affects taste perception without interfering with taste cell development or integrity. CALHM1 is expressed specifically in sweet/bitter/umami-sensing type II taste bud cells. Its heterologous expression induces a novel ATP permeability that releases ATP from cells in response to manipulations that activate the CALHM1 ion channel. Knockout of Calhm1 strongly reduces voltage-gated currents in type II cells and taste-evoked ATP release from taste buds without affecting the excitability of taste cells by taste stimuli. Thus, CALHM1 is a voltage-gated ATP-release channel required for sweet, bitter and umami taste perception.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chaudhari, N. & Roper, S. D. The cell biology of taste. J. Cell Biol. 190, 285–296 (2010)
Ma, Z. et al. Calcium homeostasis modulator 1 (CALHM1) is the pore-forming subunit of an ion channel that mediates extracellular Ca2+ regulation of neuronal excitability. Proc. Natl Acad. Sci. USA 109, E1963–E1971 (2012)
Siebert, A. P. et al. Structural and functional similarities of calcium homeostasis modulator 1 (CALHM1) ion channel with connexins, pannexins and innexins. J. Biol. Chem. 288, 6140–6153 (2013)
Chandrashekar, J., Hoon, M. A., Ryba, N. J. & Zuker, C. S. The receptors and cells for mammalian taste. Nature 444, 288–294 (2006)
Finger, T. E. et al. ATP signaling is crucial for communication from taste buds to gustatory nerves. Science 310, 1495–1499 (2005)
Dreses-Werringloer, U. et al. A polymorphism in CALHM1 influences Ca2+ homeostasis, Aβ levels, and Alzheimer’s disease risk. Cell 133, 1149–1161 (2008)
Koppel, J. et al. CALHM1 P86L polymorphism modulates CSF Aβ levels in cognitively healthy individuals at risk for Alzheimer’s disease. Mol. Med. 17, 974–979 (2011)
Lambert, J. C. et al. The CALHM1 P86L polymorphism is a genetic modifier of age at onset in Alzheimer’s disease: a meta-analysis study. J. Alzheimers Dis. 22, 247–255 (2010)
Moyer, B. D. et al. Expression of genes encoding multi-transmembrane proteins in specific primate taste cell populations. PLoS ONE 4, e7682 (2009)
Matsumoto, I., Ohmoto, M., Narukawa, M., Yoshihara, Y. & Abe, K. Skn-1a (Pou2f3) specifies taste receptor cell lineage. Nature Neurosci. 14, 685–687 (2011)
Zhang, Y. et al. Coding of sweet, bitter, and umami tastes: different receptor cells sharing similar signaling pathways. Cell 112, 293–301 (2003)
Huang, Y. J. et al. The role of pannexin 1 hemichannels in ATP release and cell-cell communication in mouse taste buds. Proc. Natl Acad. Sci. USA 104, 6436–6441 (2007)
Romanov, R. A. et al. Afferent neurotransmission mediated by hemichannels in mammalian taste cells. EMBO J. 26, 657–667 (2007)
Vandenbeuch, A., Zorec, R. & Kinnamon, S. C. Capacitance measurements of regulated exocytosis in mouse taste cells. J. Neurosci. 30, 14695–14701 (2010)
Romanov, R. A. et al. Dispensable ATP permeability of pannexin 1 channels in a heterologous system and in mammalian taste cells. J. Cell Sci. 125, 5514–5523 (2012)
Sabirov, R. Z. & Okada, Y. ATP release via anion channels. Purinergic Signal. 1, 311–328 (2005)
Silverman, W., Locovei, S. & Dahl, G. Probenecid, a gout remedy, inhibits pannexin 1 channels. Am. J. Physiol. Cell Physiol. 295, C761–C767 (2008)
Romanov, R. A. & Kolesnikov, S. S. Electrophysiologically identified subpopulations of taste bud cells. Neurosci. Lett. 395, 249–254 (2006)
Burnstock, G. & Kennedy, C. P2X receptors in health and disease. Adv. Pharmacol. 61, 333–372 (2011)
Edwards, F. A., Gibb, A. J. & Colquhoun, D. ATP receptor-mediated synaptic currents in the central nervous system. Nature 359, 144–147 (1992)
Evans, R. J., Derkach, V. & Surprenant, A. ATP mediates fast synaptic transmission in mammalian neurons. Nature 357, 503–505 (1992)
Cotrina, M. L., Lin, J. H., Lopez-Garcia, J. C., Naus, C. C. & Nedergaard, M. ATP-mediated glia signaling. J. Neurosci. 20, 2835–2844 (2000)
Cotrina, M. L., Lin, J. H. & Nedergaard, M. Cytoskeletal assembly and ATP release regulate astrocytic calcium signaling. J. Neurosci. 18, 8794–8804 (1998)
Burnstock, G. Dual control of local blood flow by purines. Ann. NY Acad. Sci. 603, 31–44 (1990)
Chizh, B. A. & Illes, P. P2X receptors and nociception. Pharmacol. Rev. 53, 553–568 (2001)
Cockayne, D. A. et al. Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice. Nature 407, 1011–1015 (2000)
Wynn, G., Rong, W., Xiang, Z. & Burnstock, G. Purinergic mechanisms contribute to mechanosensory transduction in the rat colorectum. Gastroenterology 125, 1398–1409 (2003)
Ohmoto, M., Matsumoto, I., Misaka, T. & Abe, K. Taste receptor cells express voltage-dependent potassium channels in a cell age-specific manner. Chem. Senses 31, 739–746 (2006)
Dvoryanchikov, G., Sinclair, M. S., Perea-Martinez, I., Wang, T. & Chaudhari, N. Inward rectifier channel, ROMK, is localized to the apical tips of glial-like cells in mouse taste buds. J. Comp. Neurol. 517, 1–14 (2009)
Sinclair, M. S. et al. Oxytocin signaling in mouse taste buds. PLoS ONE 5, e11980 (2010)
Tordoff, M. G. & Bachmanov, A. A. Mouse taste preference tests: why only two bottles? Chem. Senses 28, 315–324 (2003)
Damak, S. et al. Trpm5 null mice respond to bitter, sweet, and umami compounds. Chem. Senses 31, 253–264 (2006)
Eddy, M. C. et al. Double P2X2/P2X3 purinergic receptor knockout mice do not taste NaCl or the artificial sweetener SC45647. Chem. Senses 34, 789–797 (2009)
Glendinning, J. I., Gresack, J. & Spector, A. C. A high-throughput screening procedure for identifying mice with aberrant taste and oromotor function. Chem. Senses 27, 461–474 (2002)
Hallock, R. M., Tatangelo, M., Barrows, J. & Finger, T. E. Residual chemosensory capabilities in double P2X2/P2X3 purinergic receptor null mice: intraoral or postingestive detection? Chem. Senses 34, 799–808 (2009)
Spector, A. C., Andrews-Labenski, J. & Letterio, F. C. A new gustometer for psychophysical taste testing in the rat. Physiol. Behav. 47, 795–803 (1990)
Hellekant, G. & Roberts, T. W. in Experimental Cell Biology of Taste and Olfaction: Current Techniques and Protocols (eds Spielman, A. I. & Brand, J. G.) 277–290 (CRC, 1995)
Li, A., Leung, C. T., Peterson-Yantorno, K., Mitchell, C. H. & Civan, M. M. Pathways for ATP release by bovine ciliary epithelial cells, the initial step in purinergic regulation of aqueous humor inflow. Am. J. Physiol. Cell Physiol. 299, C1308–C1317 (2010)
Clapp, T. R., Medler, K. F., Damak, S., Margolskee, R. F. & Kinnamon, S. C. Mouse taste cells with G protein-coupled taste receptors lack voltage-gated calcium channels and SNAP-25. BMC Biol. 4, 7 (2006)
Acknowledgements
This work was supported by a KeySpan award to P.M., several US NIH grants (GM56328, MH059937, NS072775 to J.K.F.; DC10393 to M.G.T.; EY13624 to M.M.C.; R03DC011143 to I.M.; Core Grant P30 EY001583 to the University of Pennsylvania; Core Grant P30DC011735 to the Monell Chemical Senses Center) and the University of Minnesota’s Undergraduate Research Opportunities Program to S.L. and M.A. A.T. and M.O. are JSPS Fellows. We thank R. F. Margolskee for the TRPM5–GFP mice and Y. Ninomiya for comments on the manuscript.
Author information
Authors and Affiliations
Contributions
A.T., V.V., A.L., Z.M., M.O., I.M., H.Z., L.A., S.L., M.A., G.H., G.D. and N.C. designed and performed experiments. P.M. and V.V. generated the Calhm1 knockout mice. J.K. and P.D. designed experiments. M.G.T., M.M.C., P.M. and J.K.F. designed experiments and helped with data interpretation. A.T., J.K.F. and P.M. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-11, Supplementary Tables 1-4, a Supplementary Discussion and additional references. (PDF 2538 kb)
Rights and permissions
About this article
Cite this article
Taruno, A., Vingtdeux, V., Ohmoto, M. et al. CALHM1 ion channel mediates purinergic neurotransmission of sweet, bitter and umami tastes. Nature 495, 223–226 (2013). https://doi.org/10.1038/nature11906
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11906
This article is cited by
-
Characteristics of A-type voltage-gated K+ currents expressed on sour-sensing type III taste receptor cells in mice
Cell and Tissue Research (2024)
-
The circadian regulation of extracellular ATP
Purinergic Signalling (2023)
-
Contribution of TRPC3-mediated Ca2+ entry to taste transduction
Pflügers Archiv - European Journal of Physiology (2023)
-
Diversity and evolution of serotonergic cells in taste buds of elasmobranchs and ancestral actinopterygian fish
Cell and Tissue Research (2023)
-
Sensory Systems for Sugar-Induced Cephalic Phase Insulin Release
Current Oral Health Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.