Abstract
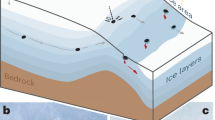
Ediacaran (635–542 million years ago) fossils have been regarded as early animal ancestors of the Cambrian evolutionary explosion of marine invertebrate phyla1, as giant marine protists2 and as lichenized fungi3. Recent documentation of palaeosols in the Ediacara Member of the Rawnsley Quartzite of South Australia4 confirms past interpretations of lagoonal–aeolian deposition based on synsedimentary ferruginization and loessic texture5,6. Further evidence for palaeosols comes from non-marine facies, dilation cracks, soil nodules, sand crystals, stable isotopic data and mass balance geochemistry4. Here I show that the uppermost surfaces of the palaeosols have a variety of fossils in growth position, including Charniodiscus, Dickinsonia, Hallidaya, Parvancorina, Phyllozoon, Praecambridium, Rugoconites, Tribrachidium and ‘old-elephant skin’ (ichnogenus Rivularites7). These fossils were preserved as ferruginous impressions, like plant fossils8, and biological soil crusts9,10 of Phanerozoic eon sandy palaeosols. Sand crystals after gypsum11 and nodules of carbonate12 are shallow within the palaeosols4, even after correcting for burial compaction13. Periglacial involutions and modest geochemical differentiation of the palaeosols are evidence of a dry, cold temperate Ediacaran palaeoclimate in South Australia4. This new interpretation of some Ediacaran fossils as large sessile organisms of cool, dry soils, is compatible with observations that Ediacaran fossils were similar in appearance and preservation to lichens and other microbial colonies of biological soil crusts3, rather than marine animals1, or protists2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Erwin, D. H. et al. The Cambrian conundrum: early divergence and later ecological success in the history of animals. Science 334, 1091–1097 (2011)
Seilacher, A., Buatois, L. A. & Mangano, M. G. Trace fossils in the Ediacaran–Cambrian transition: behavioral diversification, ecological turnover and environmental shift. Palaeogeogr. Palaeoclimatol. Palaeoecol. 227, 323–356 (2005)
Retallack, G. J. Growth, decay and burial compaction of Dickinsonia, an iconic Ediacaran fossil. Alcheringa 31, 215–240 (2007)
Retallack, G. J. Were Ediacaran siliciclastics of South Australia coastal or deep marine? Sedimentology 59, 1208–1236 (2012)
Mawson, D. & Segnit, E. R. Purple slates of the Adelaide System. Trans Roy. Soc. S. Australia 72, 276–280 (1949)
Jenkins, R. J. F., Ford, C. H. & Gehling, J. G. The Ediacara Member of the Rawnsley Quartzite: the context of the Ediacara assemblage (late Precambrian, Flinders Ranges). J. Geol. Soc. Australia 30, 101–119 (1983)
Retallack, G. J. Criteria for distinguishing microbial mats and earths. Soc. Econ. Paleont. Mineral. Spec. Pap. 101, 136–152 (2012)
Retallack, G. J. & Dilcher, D. L. Core and geophysical logs versus outcrop for interpretation of Cretaceous paleosols in the Dakota Formation of Kansas. Palaeogeogr. Palaeoclimatol. Palaeoecol. 329–330, 47–63 (2012)
Retallack, G. J. Cambrian–Ordovician non-marine fossils from South Australia. Alcheringa 33, 355–391 (2009)
Simpson, W. S. et al. A preserved Late Cretaceous biological soil crust in the capping sandstone member, Wahweap Formation, Grand Staircase-Escalante National Monument, Utah: paleoclimatic implications. Sedim. Geol. 230, 139–145 (2010)
Retallack, G. J. & Huang, C.-M. Depth to gypsic horizon as a proxy for paleoprecipitation in paleosols of sedimentary environments. Geology 38, 403–406 (2010)
Retallack, G. J. Pedogenic carbonate proxies for amount and seasonality of precipitation in paleosols. Geology 33, 333–336 (2005)
Sheldon, N. D. & Retallack, G. J. Equation for compaction of paleosols due to burial. Geology 29, 247–250 (2001)
Sprigg, R. C. Early Cambrian (?) jellyfishes from the Flinders Ranges, South Australia. Trans Roy. Soc. S. Australia 71, 212–224 (1947)
Glaessner, M. F. Precambrian animals. Sci. Am. 204, 72–78 (1961)
Fedonkin, M. A., Gehling, J. G., Grey, K., Narbonne, G. M. & Vickers-Rich, P. The Rise of Animals: Evolution and Diversification of the Kingdom Animalia (Johns Hopkins Univ. Press, 2008)
Antcliffe, J. B. & Brasier, M. D. Charnia at 50: developmental models for Ediacaran fronds. Palaeontology 51, 11–26 (2008)
Huldtgren, T. et al. Fossilized nuclei and germination structures identify Ediacaran “animal embryos” as encysting protists. Science 334, 1696–1699 (2011)
Yin, Z. et al. Early embryogenesis of potential bilaterian animals with polar lobe formation from the Ediacaran Weng’an Biota, South China. Precambr. Res. . http://dx.doi.org/10.1016/j.precamres.2011.08.011 (9 September 2011)
Yuan, X.-L., Xiao, S.-H. & Taylor, T. N. Lichen-like symbiosis 600 million years ago. Science 308, 1017–1020 (2005)
Bengtson, S., Rasmussen, B. & Krapež, B. The Paleoproterozoic megascopic Stirling biota. Paleobiology 33, 351–381 (2007)
Gehling, J. G., Droser, M. L., Jensen, S. R. & Runnegar, B. N. in Evolving Form and Function: Fossils and Development (ed. Briggs, D. E. G. ) 45–56 (Yale Peabody Museum, 2005)
Dan, J., Moshe, R. & Alperovich, N. The soils of Sede Zin. Israel J. Earth Sci. 22, 211–227 (1973)
Dan, J., Yaalon, D. H., Moshe, R. & Nissim, S. Evolution of reg soils in southern Israel and Sinai. Geoderma 28, 173–202 (1982)
Solomina, O. & Calkin, P. E. Lichenometry as applied to moraines in Alaska, USA, and Kamchatka, Russia. Arct. Antarct. Alp. Res. 35, 129–143 (2003)
Matthews, J. A. “Little Ice Age” glacier variations in Jotunheim, southern Norway: a study in regionally controlled lichenometric dating of recessional moraines, with implications for climate and lichen growth rates. Holocene 15, 1–19 (2005)
Food & Agriculture Organization. Soil Map of the World Vol. VIII, North and Central Asia (United Nations Educ. Cult. Org., 1978)
Jenkins, R. J. F. in The Geological Record of Neoproterozoic Glaciations (eds Arnaud, E., Halverson, G. P. and Shields-Zhou, G. ) 693–698 (Geol. Soc. London Mem., 2011)
Ewing, S. A. et al. A threshold in soil formation at Earth’s arid-hyperarid transition. Geochim. Cosmochim. Acta 70, 5293–5322 (2006)
Grazhdankin, D. & Gerdes, H. Y. Ediacaran microbial colonies. Lethaia 40, 201–210 (2007)
Acknowledgements
K. Lloyd, P. Coulthard, A. Coulthard, K. Anderson and D. Crawford facilitated permission to undertake research in Flinders Ranges National Park. B. Logan and M. Willison aided sampling of drill core at PIRSA, Glenside. T. Palmer and D. Atkins provided mathematical advice. Fieldwork was funded by the PRF fund of the American Chemical Society, and aided by C. Metzger and J. Gehling.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text, and Data, Supplementary Figures 1-7, Supplementary Tables 1-6 and Supplementary References. (PDF 1855 kb)
Rights and permissions
About this article
Cite this article
Retallack, G. Ediacaran life on land. Nature 493, 89–92 (2013). https://doi.org/10.1038/nature11777
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11777
This article is cited by
-
The anchor-shaped Ediacaran organism Parvancorina from Marwar Supergroup, India
Journal of Earth System Science (2023)
-
Genomic and fossil windows into the secret lives of the most ancient fungi
Nature Reviews Microbiology (2020)
-
Early fungi from the Proterozoic era in Arctic Canada
Nature (2019)
-
Early bursts of diversification defined the faunal colonization of land
Nature Ecology & Evolution (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.