Abstract
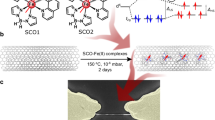
The use of molecular spin state as a quantum of information for storage, sensing and computing has generated considerable interest in the context of next-generation data storage and communication devices1,2, opening avenues for developing multifunctional molecular spintronics3. Such ideas have been researched extensively, using single-molecule magnets4,5 and molecules with a metal ion6 or nitrogen vacancy7 as localized spin-carrying centres for storage and for realizing logic operations8. However, the electronic coupling between the spin centres of these molecules is rather weak, which makes construction of quantum memory registers a challenging task9. In this regard, delocalized carbon-based radical species with unpaired spin, such as phenalenyl10, have shown promise. These phenalenyl moieties, which can be regarded as graphene fragments, are formed by the fusion of three benzene rings and belong to the class of open-shell systems. The spin structure of these molecules responds to external stimuli11,12 (such as light, and electric and magnetic fields), which provides novel schemes for performing spin memory and logic operations. Here we construct a molecular device using such molecules as templates to engineer interfacial spin transfer resulting from hybridization and magnetic exchange interaction with the surface of a ferromagnet; the device shows an unexpected interfacial magnetoresistance of more than 20 per cent near room temperature. Moreover, we successfully demonstrate the formation of a nanoscale magnetic molecule with a well-defined magnetic hysteresis on ferromagnetic surfaces. Owing to strong magnetic coupling with the ferromagnet, such independent switching of an adsorbed magnetic molecule has been unsuccessful with single-molecule magnets13. Our findings suggest the use of chemically amenable phenalenyl-based molecules as a viable and scalable platform for building molecular-scale quantum spin memory and processors for technological development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Verdaguer, M. Molecular electronics emerges from molecular magnetism. Science 272, 698–699 (1996)
Miyamachi, T. et al. Robust spin crossover and memristance across a single molecule. Nature Commun. 3, 938 (2012)
Rocha, A. R. et al. Towards molecular spintronics. Nature Mater. 4, 335–339 (2005)
Leuenberger, M. N. & Loss, D. Quantum computing in molecular magnets. Nature 410, 789–793 (2001)
Mannini, M. et al. Magnetic memory of a single-molecule quantum magnet wired to a gold surface. Nature Mater. 8, 194–197 (2009)
Sessoli, R., Gatteschi, D., Caneschi, A. & Novak, M. A. Magnetic bistability in a metal-ion cluster. Nature 365, 141–143 (1993)
Fuchs, G. D., Burkard, G., Klimov, P. V. & Awschalom, D. D. A quantum memory intrinsic to single nitrogen–vacancy centres in diamond. Nature Phys. 7, 789–793 (2011)
Ardavan, A. et al. Will spin-relaxation times in molecular magnets permit quantum information processing? Phys. Rev. Lett. 98, 057201 (2007)
Morita, Y., Suzuki, S., Sato, K. & Takui, T. Synthetic organic spin chemistry for structurally well-defined open-shell graphene fragments. Nature Chem. 3, 197–204 (2011)
Haddon, R. C. Design of organic metals and superconductors. Nature 256, 394–396 (1975)
Hicks, R. G. What's new in stable radical chemistry? Org. Biomol. Chem. 5, 1321–1338 (2007)
Itkis, M. E. et al. Magneto-opto-electronic bistability in a phenalenyl-based neutral radical. Science 296, 1443–1445 (2002)
Wende, H. et al. Substrate-induced magnetic ordering and switching of iron porphyrin molecules. Nature Mater. 6, 516–520 (2007)
Manriquez, J. M., Yee, G. T., Mclean, R. S., Epstein, A. J. & Miller, J. S. A room-temperature molecular/organic-based magnet. Science 252, 1415–1417 (1991)
Herrmann, C., Solomon, G. C. & Ratner, M. A. Organic radicals as spin filters. J. Am. Chem. Soc. 132, 3682–3684 (2010)
Gütlich, P. & Goodwin, H. A. Spin Crossover in Transition Metal Compounds I (Springer, 2004)
Morita, Y. et al. Organic tailored batteries materials using stable open-shell molecules with degenerate frontier orbitals. Nature Mater. 10, 947–951 (2011)
Xiong, Z. H., Wu, D., Vardeny, Z. V. & Shi, J. Giant magnetoresistance in organic spin-valves. Nature 427, 821–824 (2004)
Santos, T. S. et al. Room-temperature tunnel magnetoresistance and spin-polarized tunneling through an organic semiconductor barrier. Phys. Rev. Lett. 98, 016601 (2007)
Shim, J. H. et al. Large spin diffusion length in an amorphous organic semiconductor. Phys. Rev. Lett. 100, 226603 (2008)
Moodera, J. S., Santos, T. S. & Nagahama, T. The phenomena of spin-filter tunneling. J. Phys. Condens. Matter 19, 165202 (2007)
Gould, C. et al. Tunneling anisotropic magnetoresistance: a spin-valve-like tunnel magnetoresistance using a single magnetic layer. Phys. Rev. Lett. 93, 117203 (2004)
Grünewald, M. et al. Tunneling anisotropic magnetoresistance in organic spin valves. Phys. Rev. B 84, 125208 (2011)
Atodiresei, N. et al. Design of the local spin polarization at the organic-ferromagnetic interface. Phys. Rev. Lett. 105, 066601 (2010)
Raman, K. V. et al. Effect of molecular ordering on spin and charge injection in rubrene. Phys. Rev. B 80, 195212 (2009)
Sanvito, S. Molecular spintronics: The rise of spinterface science. Nature Phys. 6, 562–564 (2010)
Gambardella, P. et al. Supramolecular control of the magnetic anisotropy in two-dimensional high-spin Fe arrays at a metal interface. Nature Mater. 8, 189–193 (2009)
Dimitrov, D. A. & Wysin, G. M. Magnetic properties of spherical fcc clusters with radial surface anisotropy. Phys. Rev. B 51, 11947–11950 (1995)
Siegmann, H. C. Surface and 2D magnetism. J. Phys. Condens. Matter 4, 8395–8434 (1992)
Miller, J. S. & Drillon, M. Magnetism: Molecules to Materials Vol. 3, Magnetism: Nanosized Magnetic Materials (Wiley-VCH, 2002)
Acknowledgements
We thank J. M. D. Coey of Trinity College, Ireland, for discussions. K.V.R. and J.S.M. were supported by the Office of Naval Research (ONR grant N00014-09-1-0177) and the National Science Foundation (grants DMR 0504158 and ULFR 09-0532-01). A.M.K. thanks the University of Groningen for partial financial support during his stay at MIT. N.A. and V.C. thank the Julich Supercomputing Centre, Forschungszentrum Julich (Germany), for performing calculations on JUROPA and JUGENE supercomputers. A.M. and T.K.S. thank IISER-Kolkata and CSIR, India, respectively, for research fellowships. S.K.M. thanks CSIR (sanction no. 01(2369)/10/EMR-II), India, for financial support. M.M. thanks the German Science foundation for support within SFB 602 and SPP 1538, and S. Demeshko for SQUID measurements. D.S. and R.M. thank the Deutsche Forschungsgemeinschaft (DFG) Priority Programme 1178 and the Danish National Research Foundation (DNRF) funded Center for Materials Crystallography (CMC) for support, and the Land Niedersachsen for providing a fellowship in the Catalysis for Sustainable Synthesis (CaSuS) Ph.D. program. J.S.M., M.M, S.K.M. and D.S. thank the Göttingen-Kolkata ‘Open shell systems (G-KOSS)’ initiative for supporting the collaboration.
Author information
Authors and Affiliations
Contributions
S.K.M., M.M. and J.S.M. designed the original research approach; A.M. and S.K.M. developed molecule synthesis; A.M.K., A.M., T.K.S., S.K.M., R.M. and D.S. performed material characterization; K.V.R. and A.M.K. prepared devices, performed transport experiments and analysed transport and magnetic characterization; N.A., P.L. and V.C. performed DFT calculations; K.V.R., A.M.K., N.A. and J.S.M. developed the organic spin-filter model; K.V.R., N.A. and J.S.M. developed the interface magnetic anisotropy model; K.V.R. coordinated collaboration; K.V.R., J.S.M. and M.M. wrote the manuscript; N.A., A.M.K., A.M. and S.K.M. provided the discussion and contributed to manuscript preparation. All authors discussed the experiments and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data, Supplementary Figures 1-7, Supplementary Table 1 and Supplementary References. (PDF 881 kb)
Rights and permissions
About this article
Cite this article
Raman, K., Kamerbeek, A., Mukherjee, A. et al. Interface-engineered templates for molecular spin memory devices. Nature 493, 509–513 (2013). https://doi.org/10.1038/nature11719
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11719
This article is cited by
-
Influence of proton irradiation on the magnetic properties of two-dimensional Ni(II) molecular magnet
Scientific Reports (2023)
-
Molecular design for enhanced spin transport in molecular semiconductors
Nano Research (2023)
-
Electronic properties and molecular electrostatic potential mapping of edge functionalized GQDs with ZnO, CuO, and TiO2
Optical and Quantum Electronics (2023)
-
Triplet-radical spin entanglement: potential of molecular materials for high-temperature quantum information processing
NPG Asia Materials (2022)
-
Comparative study of optoelectronic, thermodynamic, linear and nonlinear optical properties of methyl phenalenyl doped to zinc and copper and their applications
Journal of Materials Science: Materials in Electronics (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.