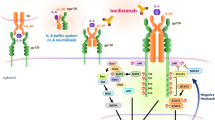
Abstract
The innate immune response is essential for combating infectious disease. Macrophages and other cells respond to infection by releasing cytokines, such as interleukin-1β (IL-1β), which in turn activate a well-described, myeloid-differentiation factor 88 (MYD88)-mediated, nuclear factor-κB (NF-κB)-dependent transcriptional pathway that results in inflammatory-cell activation and recruitment1,2,3,4. Endothelial cells, which usually serve as a barrier to the movement of inflammatory cells out of the blood and into tissue, are also critical mediators of the inflammatory response5,6. Paradoxically, the cytokines vital to a successful immune defence also have disruptive effects on endothelial cell–cell interactions and can trigger degradation of barrier function and dissociation of tissue architecture7,8,9. The mechanism of this barrier dissolution and its relationship to the canonical NF-κB pathway remain poorly defined. Here we show that the direct, immediate and disruptive effects of IL-1β on endothelial stability in a human in vitro cell model are NF-κB independent and are instead the result of signalling through the small GTPase ADP-ribosylation factor 6 (ARF6) and its activator ARF nucleotide binding site opener (ARNO; also known as CYTH2). Moreover, we show that ARNO binds directly to the adaptor protein MYD88, and thus propose MYD88–ARNO–ARF6 as a proximal IL-1β signalling pathway distinct from that mediated by NF-κB. Finally, we show that SecinH3, an inhibitor of ARF guanine nucleotide-exchange factors such as ARNO, enhances vascular stability and significantly improves outcomes in animal models of inflammatory arthritis and acute inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Li, Q. & Verma, I. M. NF-κB regulation in the immune system. Nature Rev. Immunol. 2, 725–734 (2002)
Collins, T. et al. Transcriptional regulation of endothelial cell adhesion molecules: NF-κB and cytokine-inducible enhancers. FASEB J. 9, 899–909 (1995)
Liu, S. F. & Malik, A. B. NF-κB activation as a pathological mechanism of septic shock and inflammation. Am. J. Physiol. Lung Cell. Mol. Physiol. 290, L622–L645 (2006)
Rothwarf, D. M. & Karin, M. The NF-κB activation pathway: a paradigm in information transfer from membrane to nucleus. Sci. STKE 1999, RE1 (1999)
Pober, J. S. & Sessa, W. C. Evolving functions of endothelial cells in inflammation. Nature Rev. Immunol. 7, 803–815 (2007)
Muller, W. A. Leukocyte–endothelial cell interactions in the inflammatory response. Lab. Invest. 82, 521–533 (2002)
Royall, J. A. et al. Tumor necrosis factor and interleukin 1 alpha increase vascular endothelial permeability. Am. J. Physiol. Lung Cell. Mol. Physiol. 257, L399–L410 (1989)
West, X. Z. et al. Oxidative stress induces angiogenesis by activating TLR2 with novel endogenous ligands. Nature 467, 972–976 (2010)
London, N. R. et al. Targeting Robo4-dependent Slit signaling to survive the cytokine storm in sepsis and influenza. Sci. Transl. Med. 2, 23ra19 (2010)
Zhu, J. & Mohan, C. Toll-like receptor signaling pathways–therapeutic opportunities. Mediators Inflamm. 2010, 781235 (2010)
Kishore, N. et al. A selective IKK-2 inhibitor blocks NF-κB-dependent gene expression in interleukin-1β-stimulated synovial fibroblasts. J. Biol. Chem. 278, 32861–32871 (2003)
Matthews, J. S. & O’Neill, L. A. Distinct roles for p42/p44 and p38 mitogen-activated protein kinases in the induction of IL-2 by IL-1. Cytokine 11, 643–655 (1999)
Martin, M. U. & Wesche, H. Summary and comparison of the signaling mechanisms of the Toll/interleukin-1 receptor family. Biochim. Biophys. Acta 1592, 265–280 (2002)
Kaltschmidt, B., Linker, R. A., Deng, J. & Kaltschmidt, C. Cyclooxygenase-2 is a neuronal target gene of NF-κB. BMC Mol. Biol. 3, 16 (2002)
Funk, C. D. Prostaglandins and leukotrienes: advances in eicosanoid biology. Science 294, 1871–1875 (2001)
Palacios, F., Price, L., Schweitzer, J., Collard, J. G. & D’Souza-Schorey, C. An essential role for ARF6-regulated membrane traffic in adherens junction turnover and epithelial cell migration. EMBO J. 20, 4973–4986 (2001)
D’Souza-Schorey, C., Li, G., Colombo, M. I. & Stahl, P. D. A regulatory role for ARF6 in receptor-mediated endocytosis. Science 267, 1175–1178 (1995)
Riley, K. N., Maldonado, A. E., Tellier, P., D’Souza-Schorey, C. & Herman, I. M. Betacap73–ARF6 interactions modulate cell shape and motility after injury in vitro. Mol. Biol. Cell 14, 4155–4161 (2003)
Zhang, Q. et al. Small-molecule synergist of the Wnt/β-catenin signaling pathway. Proc. Natl Acad. Sci. USA 104, 7444–7448 (2007)
Hafner, M. et al. Inhibition of cytohesins by SecinH3 leads to hepatic insulin resistance. Nature 444, 941–944 (2006)
Béraud-Dufour, S. et al. A glutamic finger in the guanine nucleotide exchange factor ARNO displaces Mg2+ and the β-phosphate to destabilize GDP on ARF1. EMBO J. 17, 3651–3659 (1998)
Arend, W. P. Cytokine imbalance in the pathogenesis of rheumatoid arthritis: the role of interleukin-1 receptor antagonist. Semin. Arthritis Rheum. 30, 1–6 (2001)
Szekanecz, Z. & Koch, A. E. Vascular involvement in rheumatic diseases: ‘vascular rheumatology’. Arthritis Res. Ther. 10, 224 (2008)
van den Berg, W. B., Joosten, L. A., Helsen, M. & van de Loo, F. A. Amelioration of established murine collagen-induced arthritis with anti-IL-1 treatment. Clin. Exp. Immunol. 95, 237–243 (1994)
Joosten, L. A., Helsen, M. M., van de Loo, F. A. & van den Berg, W. B. Anticytokine treatment of established type II collagen-induced arthritis in DBA/1 mice. A comparative study using anti-TNFα, anti-IL-1α/β, and IL-1Ra. Arthritis Rheum. 39, 797–809 (1996)
Wooley, P. H., Dutcher, J., Widmer, M. B. & Gillis, S. Influence of a recombinant human soluble tumor necrosis factor receptor FC fusion protein on type II collagen-induced arthritis in mice. J. Immunol. 151, 6602–6607 (1993)
London, N. R., Whitehead, K. J. & Li, D. Y. Endogenous endothelial cell signaling systems maintain vascular stability. Angiogenesis 12, 149–158 (2009)
Keane, J. et al. Tuberculosis associated with infliximab, a tumor necrosis factor α-neutralizing agent. N. Engl. J. Med. 345, 1098–1104 (2001)
Lee, W. L. & Slutsky, A. S. Sepsis and endothelial permeability. N. Engl. J. Med. 363, 689–691 (2010)
Silva, L. C., Ortigosa, L. C. & Benard, G. Anti-TNF-α agents in the treatment of immune-mediated inflammatory diseases: mechanisms of action and pitfalls. Immunotherapy 2, 817–833 (2010)
Acknowledgements
We thank D. Lim and T. Mleynek for graphical assistance, G. Zimmerman and J. Kaplan for reading the manuscript, S. Odelberg for reading the manuscript and statistical analysis, J. Ling for help with immunostaining, R. Campbell and A. Weyrich for providing primary human blood cells, C. Rodesch and the University of Utah Cell Imaging/Fluorescence Facility as well as the University of Utah Flow Cytometry Facility, and M. P. Revelo for help with pathology. D.Y.L. and his laboratory were funded by grants from the National Heart, Lung, and Blood Institute; Burroughs Wellcome Fund; Juvenile Diabetes Research Foundation; NIAID Rocky Mountain Regional Center of Excellence in Biodefense and Emerging Infectious Disease; the American Asthma Foundation; and the Department of Defense. D.Y.L. is the HA and Edna Benning Endowed Professor of Medicine and Cardiology.
Author information
Authors and Affiliations
Contributions
W.Z., N.R.L., C.C.G. and D.Y.L. were responsible for project conceptualization, experimental design, data analysis and manuscript preparation. W.Z., C.C.G., C.T.D., Z.T., L.K.S., D.S.S. and J.G. performed and collected data for in vitro experiments. N.R.L. collected data for in vivo experiments. C.C.G. developed software techniques for immunofluorescence analysis. M.C.P.S. performed flow cytometry experiments. Z.T. and K.R.T. made constructs and adenoviruses. A.H.G. provided histology and pathology expertise. D.Y.L. was responsible for funding the project.
Corresponding author
Ethics declarations
Competing interests
The University of Utah has filed intellectual property concerning targeting signalling pathways to treat inflammatory arthritis. The University of Utah has licensed technology to Navigen, a biotechnology company owned in part by the University of Utah Research Foundation. N.R.L. and W.Z. are paid consultants for Navigen and D.Y.L. is a co-founder of Navigen.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-9, Supplementary Methods and Supplementary References. This file was replaced on December 19th 2012. (PDF 2295 kb)
Rights and permissions
About this article
Cite this article
Zhu, W., London, N., Gibson, C. et al. Interleukin receptor activates a MYD88–ARNO–ARF6 cascade to disrupt vascular stability. Nature 492, 252–255 (2012). https://doi.org/10.1038/nature11603
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11603
This article is cited by
-
Activation of NFAT by HGF and IGF-1 via ARF6 and its effector ASAP1 promotes uveal melanoma metastasis
Oncogene (2023)
-
Dietary emulsifier consumption alters gene expression in the amygdala and paraventricular nucleus of the hypothalamus in mice
Scientific Reports (2022)
-
The alarmin interleukin-1α triggers secondary degeneration through reactive astrocytes and endothelium after spinal cord injury
Nature Communications (2022)
-
Angiogenesis in Wound Healing following Pharmacological and Toxicological Exposures
Current Pathobiology Reports (2020)
-
A natural mouse model reveals genetic determinants of systemic capillary leak syndrome (Clarkson disease)
Communications Biology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.