Abstract
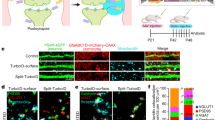
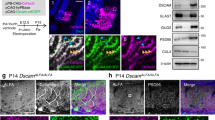
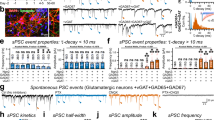
In the developing central nervous system (CNS), the control of synapse number and function is critical to the formation of neural circuits. We previously demonstrated that astrocyte-secreted factors powerfully induce the formation of functional excitatory synapses between CNS neurons1. Astrocyte-secreted thrombospondins induce the formation of structural synapses, but these synapses are postsynaptically silent2. Here we use biochemical fractionation of astrocyte-conditioned medium to identify glypican 4 (Gpc4) and glypican 6 (Gpc6) as astrocyte-secreted signals sufficient to induce functional synapses between purified retinal ganglion cell neurons, and show that depletion of these molecules from astrocyte-conditioned medium significantly reduces its ability to induce postsynaptic activity. Application of Gpc4 to purified neurons is sufficient to increase the frequency and amplitude of glutamatergic synaptic events. This is achieved by increasing the surface level and clustering, but not overall cellular protein level, of the GluA1 subunit of the AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid) glutamate receptor (AMPAR). Gpc4 and Gpc6 are expressed by astrocytes in vivo in the developing CNS, with Gpc4 expression enriched in the hippocampus and Gpc6 enriched in the cerebellum. Finally, we demonstrate that Gpc4-deficient mice have defective synapse formation, with decreased amplitude of excitatory synaptic currents in the developing hippocampus and reduced recruitment of AMPARs to synapses. These data identify glypicans as a family of novel astrocyte-derived molecules that are necessary and sufficient to promote glutamate receptor clustering and receptivity and to induce the formation of postsynaptically functioning CNS synapses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ullian, E. M., Sapperstein, S. K., Christopherson, K. S. & Barres, B. A. Control of synapse number by glia. Science 291, 657–661 (2001)
Christopherson, K. S. et al. Thrombospondins are astrocyte-secreted proteins that promote CNS synaptogenesis. Cell 120, 421–433 (2005)
Traynelis, S. F. et al. Glutamate receptor ion channels: structure, regulation, and function. Pharmacol. Rev. 62, 405–496 (2010)
Kucukdereli, H. et al. Control of excitatory CNS synaptogenesis by astrocyte-secreted proteins Hevin and SPARC. Proc. Natl Acad. Sci. USA 108, E440–E449 (2011)
Foo, L. C. et al. Development of a method for the purification and culture of rodent astrocytes. Neuron 71, 799–811 (2011)
Ybot-Gonzalez, P., Copp, A. J. & Greene, N. D. Expression pattern of glypican-4 suggests multiple roles during mouse development. Dev. Dyn. 233, 1013–1017 (2005)
Song, H. H. & Filmus, J. The role of glypicans in mammalian development. Biochim. Biophys. Acta 1573, 241–246 (2002)
Watanabe, K., Yamada, H. & Yamaguchi, Y. K-glypican: a novel GPI-anchored heparan sulfate proteoglycan that is highly expressed in developing brain and kidney. J. Cell Biol. 130, 1207–1218 (1995)
De Cat, B. & David, G. Developmental roles of the glypicans. Semin. Cell Dev. Biol. 12, 117–125 (2001)
Van Vactor, D., Wall, D. P. & Johnson, K. G. Heparan sulfate proteoglycans and the emergence of neuronal connectivity. Curr. Opin. Neurobiol. 16, 40–51 (2006)
Johnson, K. G. et al. The HSPGs Syndecan and Dally-like bind the receptor phosphatase LAR and exert distinct effects on synaptic development. Neuron 49, 517–531 (2006)
Cahoy, J. D. et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J. Neurosci. 28, 264–278 (2008)
Hsia, A. Y., Malenka, R. C. & Nicoll, R. A. Development of excitatory circuitry in the hippocampus. J. Neurophysiol. 79, 2013–2024 (1998)
Lu, W. et al. Subunit composition of synaptic AMPA receptors revealed by a single-cell genetic approach. Neuron 62, 254–268 (2009)
Andrasfalvy, B. K., Smith, M. A., Borchardt, T., Sprengel, R. & Magee, J. C. Impaired regulation of synaptic strength in hippocampal neurons from GluR1-deficient mice. J. Physiol. (Lond.) 552, 35–45 (2003)
O’Brien, R. J. et al. Synaptic clustering of AMPA receptors by the extracellular immediate-early gene product Narp. Neuron 23, 309–323 (1999)
Beattie, E. C. et al. Control of synaptic strength by glial TNF-α. Science 295, 2282–2285 (2002)
Frischknecht, R. et al. Brain extracellular matrix affects AMPA receptor lateral mobility and short-term synaptic plasticity. Nature Neurosci. 12, 897–904 (2009)
Kumar, S. S., Bacci, A., Kharazia, V. & Huguenard, J. R. A developmental switch of AMPA receptor subunits in neocortical pyramidal neurons. J. Neurosci. 22, 3005–3015 (2002)
Friedman, H. V., Bresler, T., Garner, C. C. & Ziv, N. E. Assembly of new individual excitatory synapses: time course and temporal order of synaptic molecule recruitment. Neuron 27, 57–69 (2000)
Groc, L., Gustafsson, B. & Hanse, E. AMPA signalling in nascent glutamatergic synapses: there and not there!. Trends Neurosci. 29, 132–139 (2006)
Malinow, R. & Malenka, R. C. AMPA receptor trafficking and synaptic plasticity. Annu. Rev. Neurosci. 25, 103–126 (2002)
Dunah, A. W. et al. LAR receptor protein tyrosine phosphatases in the development and maintenance of excitatory synapses. Nature Neurosci. 8, 458–467 (2005)
Tang, T. et al. A mouse knockout library for secreted and transmembrane proteins. Nature Biotechnol. 28, 749–755 (2010)
Varoqueaux, F. et al. Neuroligins determine synapse maturation and function. Neuron 51, 741–754 (2006)
Lesch, K.-P. et al. Molecular genetics of adult ADHD: converging evidence from genome-wide association and extended pedigree linkage studies. J. Neural Transm. 115, 1573–1585 (2008)
Calboli, F. C. et al. A genome-wide association study of neuroticism in a population-based sample. PLoS ONE 5, e11504 (2010)
Eroglu, C. et al. Gabapentin receptor α2δ-1 is a neuronal thrombospondin receptor responsible for excitatory CNS synaptogenesis. Cell 139, 380–392 (2009)
Williams, E. H. et al. Dally-like core protein and its mammalian homologues mediate stimulatory and inhibitory effects on Hedgehog signal response. Proc. Natl Acad. Sci. USA 107, 5869–5874 (2010)
Steinmetz, C. C., Buard, I., Claudepierre, T., Nagler, K. & Pfrieger, F. W. Regional variations in the glial influence on synapse development in the mouse CNS. J. Physiol. (Lond.) 577, 249–261 (2006)
Schaeren-Wiemers, N. &. Gerfin-Moser, A. A single protocol to detect transcripts of various types and expression levels in neural tissue and cultured cells: in situ hybridization using digoxigenin-labelled cRNA probes. Histochem. Cell Biol. 100, 431–440 (1993)
Micheva, K. D. & Smith, S. J. Array tomography: a new tool for imaging the molecular architecture and ultrastructure of neural circuits. Neuron 55, 25–36 (2007)
Acknowledgements
We thank R. Winant for mass spectrometry analysis, A. Olson for array tomography assistance, S. Pasca for assistance with quantitative real-time PCR and M. Fabian for technical assistance with cell cultures. This project was funded by a NIDA grant to B.A.B. (R01DA015043). N.J.A. was supported by a Human Frontiers Long Term Fellowship. S.J.S. is supported by grants R01NS0725252 and R01NS077601 from the NIH/NINDS.
Author information
Authors and Affiliations
Contributions
N.J.A. and B.A.B. designed the experiments and wrote the manuscript; N.J.A., M.L.B., L.C.F. and C.C. performed and analysed the experiments; and G.X.W. and S.J.S. assisted with data analysis.
Corresponding author
Ethics declarations
Competing interests
Stanford University has licensed a patent for array tomography developed by S.J.S. to Aratome, LLC, of Menlo Park, California. S.J.S. has founder’s equity shares in Aratome.
Supplementary information
Supplementary Information
This file contains Supplementary Methods and Materials, Supplementary References, Supplementary Figures 1-19 and Supplementary Tables 1-2. (PDF 7806 kb)
Rights and permissions
About this article
Cite this article
Allen, N., Bennett, M., Foo, L. et al. Astrocyte glypicans 4 and 6 promote formation of excitatory synapses via GluA1 AMPA receptors. Nature 486, 410–414 (2012). https://doi.org/10.1038/nature11059
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11059
This article is cited by
-
Exosome-mediated repair of spinal cord injury: a promising therapeutic strategy
Stem Cell Research & Therapy (2024)
-
Astrocytes in the adult dentate gyrus—balance between adult and developmental tasks
Molecular Psychiatry (2024)
-
Glypican-4 serum levels are associated with cognitive dysfunction and vascular risk factors in Parkinson’s disease
Scientific Reports (2024)
-
Novel model of cortical–meningeal organoid co-culture system improves human cortical brain organoid cytoarchitecture
Scientific Reports (2023)
-
Functional roles of reactive astrocytes in neuroinflammation and neurodegeneration
Nature Reviews Neurology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.