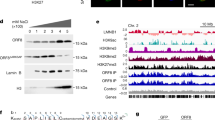
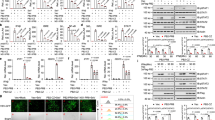
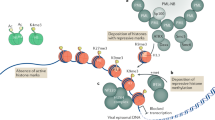
Abstract
Viral infection is commonly associated with virus-driven hijacking of host proteins. Here we describe a novel mechanism by which influenza virus affects host cells through the interaction of influenza non-structural protein 1 (NS1) with the infected cell epigenome. We show that the NS1 protein of influenza A H3N2 subtype possesses a histone-like sequence (histone mimic) that is used by the virus to target the human PAF1 transcription elongation complex (hPAF1C). We demonstrate that binding of NS1 to hPAF1C depends on the NS1 histone mimic and results in suppression of hPAF1C-mediated transcriptional elongation. Furthermore, human PAF1 has a crucial role in the antiviral response. Loss of hPAF1C binding by NS1 attenuates influenza infection, whereas hPAF1C deficiency reduces antiviral gene expression and renders cells more susceptible to viruses. We propose that the histone mimic in NS1 enables the influenza virus to affect inducible gene expression selectively, thus contributing to suppression of the antiviral response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kornberg, R. D. & Thomas, J. O. Chromatin structure—oligomers of histones. Science 184, 865–868 (1974)
Campos, E. I. & Reinberg, D. Histones: annotating chromatin. Ann. Rev. Genet. 43, 559–599 (2009)
Taverna, S. D., Li, H., Ruthenburg, A. J., Allis, C. D. & Patel, D. J. How chromatin-binding modules interpret histone modifications: lessons from professional pocket pickers. Nature Struct. Mol. Biol. 14, 1025–1040 (2007)
Kouzarides, T. Chromatin modifications and their function. Cell 128, 693–705 (2007)
Kelly, A. E. et al. Survivin reads phosphorylated histone H3 threonine 3 to activate the mitotic kinase Aurora B. Science 330, 235–239 (2010)
Fernandez-Capetillo, O. et al. DNA damage-induced G2-M checkpoint activation by histone H2AX and 53BP1. Nature Cell Biol. 4, 993–997 (2002)
Li, B., Carey, M. & Workman, J. L. The role of chromatin during transcription. Cell 128, 707–719 (2007)
Filippakopoulos, P. et al. Selective inhibition of BET bromodomains. Nature 468, 1067–1073 (2010)
Nicodeme, E. et al. Suppression of inflammation by a synthetic histone mimic. Nature 468, 1119–1123 (2010)
Nishiyama, A. et al. Intracellular delivery of acetyl-histone peptides inhibits native bromodomain-chromatin interactions and impairs mitotic progression. Febs Lett. 582, 1501–1507 (2008)
Hargreaves, D. C., Horng, T. & Medzhitov, R. Control of inducible gene expression by signal-dependent transcriptional elongation. Cell 138, 129–145 (2009)
Sampath, S. C. et al. Methylation of a histone mimic within the histone methyltransferase G9a regulates protein complex assembly. Mol. Cell 27, 596–608 (2007)
Elde, N. C. & Malik, H. S. The evolutionary conundrum of pathogen mimicry. Nature Rev. Microbiol. 7, 787–797 (2009)
Hale, B. G., Randall, R. E., Ortin, J. & Jackson, D. The multifunctional NS1 protein of influenza A viruses. J. Gen. Virol. 89, 2359–2376 (2008)
Garcia-Sastre, A. et al. Influenza A virus lacking the NS1 gene replicates in interferon-deficient systems. Virology 252, 324–330 (1998)
Lu, Y., Wambach, M., Katze, M. G. & Krug, R. M. Binding of the influenza-virus NS1 protein to double-stranded-RNA inhibits the activation of the protein-kinase that phosphorylates the Elf-2 translation initiation-factor. Virology 214, 222–228 (1995)
Gack, M. U. et al. Influenza A Virus NS1 targets the ubiquitin ligase TRIM25 to evade recognition by the host viral RNA sensor RIG-I. Cell Host Microbe 5, 439–449 (2009)
Pichlmair, A. et al. RIG-I-mediated antiviral responses to single-stranded RNA bearing 5'-phosphates. Science 314, 997–1001 (2006)
Hale, B. G., Jackson, D., Chen, Y. H., Lamb, R. A. & Randall, R. E. Influenza A virus NS1 protein binds p85b and activates phosphatidylinositol-3-kinase signaling. Proc. Natl Acad. Sci. USA 103, 14194–14199 (2006)
Krug, R. M., Yuan, W. M., Noah, D. L. & Latham, A. G. Intracellular warfare between human influenza viruses and human cells: the roles of the viral NS1 protein. Virology 309, 181–189 (2003)
Nemeroff, M. E., Barabino, S. M. L., Li, Y. Z., Keller, W. & Krug, R. M. Influenza virus NS1 protein interacts with the cellular 30 kDa subunit of CPSF and inhibits 3' end formation of cellular pre-mRNAs. Mol. Cell 1, 991–1000 (1998)
Das, K. et al. Structural basis for suppression of a host antiviral response by influenza A virus. Proc. Natl Acad. Sci. USA 105, 13093–13098 (2008)
Satterly, N. et al. Influenza virus targets the mRNA export machinery and the nuclear pore complex. Proc. Natl Acad. Sci. USA 104, 1853–1858 (2007)
Luger, K., Mader, A. W., Richmond, R. K., Sargent, D. F. & Richmond, T. J. Crystal structure of the nucleosome core particle at 2.8 Å resolution. Nature 389, 251–260 (1997)
Hale, B. G., Barclay, W. S., Randall, R. E. & Russell, R. J. Structure of an avian influenza A virus NS1 protein effector domain. Virology 378, 1–5 (2008)
Xhemalce, B. & Kouzarides, T. A chromodomain switch mediated by histone H3 Lys 4 acetylation regulates heterochromatin assembly. Genes Dev. 24, 647–652 (2010)
Becker, P. B. et al. Site-specific acetylation of ISWI by GCN5. BMC Mol. Biol. 8, (2007)
Ruthenburg, A. J., Allis, C. D. & Wysocka, J. Methylation of lysine 4 on histone H3: Intricacy of writing and reading a single epigenetic mark. Mol. Cell 25, 15–30 (2007)
Wang, P. F. et al. Global analysis of H3K4 methylation defines MLL family member targets and points to a role for MLL1-mediated H3K4 methylation in the regulation of transcriptional initiation by RNA polymerase II. Mol. Cell. Biol. 29, 6074–6085 (2009)
Guillemette, B. et al. H3 lysine 4 is acetylated at active gene promoters and is regulated by H3 lysine 4 methylation. PLoS Genet. 7, (2011)
Lachner, M., O’Carroll, N., Rea, S., Mechtler, K. & Jenuwein, T. Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins. Nature 410, 116–120 (2001)
Shi, X. B. et al. ING2 PHD domain links histone H3 lysine 4 methylation to active gene repression. Nature 442, 96–99 (2006)
Lan, F. et al. Recognition of unmethylated histone H3 lysine 4 links BHC80 to LSD1-mediated gene repression. Nature 448, 718–722 (2007)
Wysocka, J. Identifying novel proteins recognizing histone modifications using peptide pull-down assay. Methods 40, 339–343 (2006)
Sims, R. J. et al. Human but not yeast CHD1 binds directly and selectively to histone H3 methylated at lysine 4 via its tandem chromodomains. J. Biol. Chem. 280, 41789–41792 (2005)
Kim, J., Guermah, M. & Roeder, R. G. The human PAF1 complex acts in chromatin transcription elongation both independently and cooperatively with SII/TFIIS. Cell 140, 491–503 (2010)
Ramirez-Carrozzi, V. R. et al. A unifying model for the selective regulation of inducible transcription by CpG islands and nucleosome remodeling. Cell 138, 114–128 (2009)
Kim, K. Y. & Levin, D. E. Mpk1 MAPK association with the Paf1 complex blocks Sen1-mediated premature transcription termination. Cell 144, 745–756 (2011)
Chen, Y. X. et al. DSIF, the Paf1 complex, and Tat-SF1 have nonredundant, cooperative roles in RNA polymerase II elongation. Genes Dev. 23, 2765–2777 (2009)
Jaehning, J. A. The Paf1 complex: platform or player in RNA polymerase II transcription? Biochim. Bioiphys. Acta 1799, 379–388 (2010)
Core, L. J., Waterfall, J. J. & Lis, J. T. Nascent RNA sequencing reveals widespread pausing and divergent initiation at human promoters. Science 322, 1845–1848 (2008)
Min, I. M. et al. Regulating RNA polymerase pausing and transcription elongation in embryonic stem cells. Gene Dev. 25, 742–754 (2011)
Mapendano, C. K., Lykke-Andersen, S., Kjems, J., Bertrand, E. & Jensen, T. H. Crosstalk between mRNA 3′ end processing and transcription initiation. Mol. Cell 40, 410–422 (2010)
Loucaides, E. M. et al. Nuclear dynamics of influenza A virus ribonucleoproteins revealed by live-cell imaging studies. Virology 394, 154–163 (2009)
Engelhardt, O. G., Smith, M. & Fodor, E. Association of the influenza a virus RNA-dependent RNA polymerase with cellular RNA polymerase II. J. Virol. 79, 5812–5818 (2005)
Jackson, D., Hossain, M. J., Hickman, D., Perez, D. R. & Lamb, R. A. A new influenza virus virulence determinant: The NS1 protein four C-terminal residues modulate pathogenicity. Proc. Natl Acad. Sci. USA 105, 4381–4386 (2008)
Strahl, B. D. & Allis, C. D. The language of covalent histone modifications. Nature 403, 41–45 (2000)
Turner, B. M. Histone acetylation and an epigenetic code. Bioessays 22, 836–845 (2000)
Jenuwein, T. & Allis, C. D. Translating the histone code. Science 293, 1074–1080 (2001)
Yang, Y. et al. The transmissibility and control of pandemic influenza A (H1N1) virus. Science 326, 729–733 (2009)
Acknowledgements
We thank P. deGross and A. Rudensky for the mass spectroscopy analysis of the NS1 binding proteins. A. Rojas Soto, D. Reinberg, M. Dobenecker and T. Zhanyun provided us with recombinant CHD1 (A.R.S., D.R.), recombinant Set7/9 (M.D.) and Set1C (T.Z.). F. Casadio, P. Lewis, O. Binda, O. Gozani, N. Levenkova, A. Mele, R. Darnell, L. Core, J. Lis and P. Palese gave us valuable technical advice and help with data analysis. We acknowledge the Rockefeller University Genomics Resource Center for technical support. We thank R. Cadagan, A. Santana, W. Huang, R. Chandramouli and H. Zebronsky for technical assistance, R. Rizzo for help with manuscript preparation and C. Nathan for discussion. L.M.K. for artwork. B.M is supported by NIH/NIAID K99 Pathway to Independence award (1K99AI095320-01). A.G.-S. is partially supported by NIAID grants R01AI046954, U19AI083025 and by CRIP (Center for Research in Influenza Pathogenesis), an NIAID funded Center of Excellence for Influenza Research and Surveillance, HHSN266200700010C. R.G.R. is supported by NIH grant CA129325. J.K. is supported by Charles H. Revson Foundation. I.M. is supported by American Italian Cancer Foundation. J.H. is supported by the Agency for Science, Technology and Research (A*STAR), Singapore. A.T. is supported by the NIH grant R01AI068058 and by Starr Cancer Consortium.
Author information
Authors and Affiliations
Contributions
I.M. contributed to design, execution, analysis of the experiments and manuscript preparation. J.S.Y.H. studied the role of PAF1 in viral infection and assisted in manuscript preparation. J.K. and R.R. studied the impact of NS1 on hPAF1C and transcriptional elongation. B.M., R.A.A. engineered the recombinant influenza viruses and studied viral infectivity. U.S. was involved in gene expression studies. S.D. performed bioinformatic analysis. C.W.S. generated antibody against viral polymerase. K.L.J. gave technical assistance. R.K.P. and K.L. contributed to manuscript preparation and enabled ChIP-seq and RNA-seq. A.G.-S. supervised and discussed the work with infectious influenza viruses. A.T. conceived and supervised this study and wrote the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
R.K.P. and K.L. are employees of GlaxoSmithKline. Research support, excluding salaries to the members of The Rockefeller University, was partially provided by GlaxoSmithKline.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-12, Supplementary Methods, additional references and full legends for Supplementary Tables 1-8. (PDF 1437 kb)
Supplementary Table 1
This table shows genes affected by Influenza Infection - see Supplementary Information file for full legend. (XLS 135 kb)
Supplementary Table 2
This table contains a list of genes used for the integrated ChIP-seq profile - see Supplementary Information file for full legend. (XLS 43 kb)
Supplementary Table 3
This table shows siPAF dependent genes in PR8/∆NS1 infected cells - see Supplementary Information file for full legend. (XLS 249 kb)
Supplementary Table 4
This table shows siPAF dependent genes in Influenza (H1N1) infected cells - see Supplementary Information file for full legend. (XLS 656 kb)
Supplementary Table 5
This table shows siPAF dependent genes in Influenza (H1N1) infected cells - see Supplementary Information file for full legend. (XLS 596 kb)
Supplementary Table 6
This table shows siPAF dependent genes in Poly(I:C) transfected cells - see Supplementary Information file for full legend. (XLS 888 kb)
Supplementary Table 7
This table shows siPAF dependent genes in IFNβ1 treated cells - see Supplementary Information file for full legend. (XLS 111 kb)
Supplementary Table 8
This table shows that expression of housekeeping genes are not affected by siPAF mediated hPAF1 deficiency - see Supplementary Information file for full legend. (XLS 24 kb)
Rights and permissions
About this article
Cite this article
Marazzi, I., Ho, J., Kim, J. et al. Suppression of the antiviral response by an influenza histone mimic. Nature 483, 428–433 (2012). https://doi.org/10.1038/nature10892
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10892
This article is cited by
-
Transcriptomic profiling implicates PAF1 in both active and repressive immune regulatory networks
BMC Genomics (2022)
-
Comparative transcriptome analysis reveals key epigenetic targets in SARS-CoV-2 infection
npj Systems Biology and Applications (2021)
-
The link among microbiota, epigenetics, and disease development
Environmental Science and Pollution Research (2021)
-
Host methylation predicts SARS-CoV-2 infection and clinical outcome
Communications Medicine (2021)
-
Acetylation at K108 of the NS1 protein is important for the replication and virulence of influenza virus
Veterinary Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.