Abstract
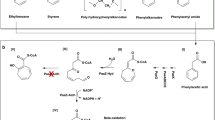
Catabolism may give rise to toxic intermediates that compromise cell vitality, such as epoxide formation in the recently elucidated and apparently universal bacterial coenzyme A (CoA)-dependent degradation of phenylacetic acid1. This compound is central to the catabolism of a variety of aromatics, such as phenylalanine, lignin-related compounds or environmental contaminants2,3. The key phenylacetyl-CoA monooxygenase (epoxidase) of the pathway, PaaABCE1,4,5, is also connected to the production of various primary and secondary metabolites6,7,8,9, as well as to the virulence of certain pathogens1,10,11. However, the enzyme complex has so far not been investigated in detail. Here we characterize the bacterial multicomponent monooxygenase PaaABCE that, surprisingly, not only transforms phenylacetyl-CoA into its ring-1,2-epoxide, but also mediates the NADPH-dependent removal of the epoxide oxygen, regenerating phenylacetyl-CoA with formation of water. We provide evidence for a catalytic di-iron centre that is probably the key to the unprecedented deoxygenation of an organic compound by an oxygenase. Presumably, the bifunctionality is vital to avoid toxic intracellular epoxide levels if the subsequent catabolic steps are impeded. Our data suggest that detoxification is assisted by two thioesterases (PaaI and PaaY) forming non-reactive breakdown products. Hence, PaaABCE may harbour an intrinsic escape mechanism from its own toxic product and represents the archetype of a bifunctional oxygenase/deoxygenase. Analogous reactions may possibly be catalysed by other di-iron epoxidases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
Data deposits
For E. coli K12, the NCBI database accession numbers are as follows: PaaY, NP_415918.1; PaaI, NP_415914.1. For Pseudomonas sp. strain Y2, the EMBL database numbers are as follows: PaaA, CAD76929.1; PaaB, CAD76932.1; PaaC, CAD76933.1; PaaD, CAD76935.1; PaaE, CAD76937.1.
References
Teufel, R. et al. Bacterial phenylalanine and phenylacetate catabolic pathway revealed. Proc. Natl Acad. Sci. USA 107, 14390–14395 (2010)
Luengo, J. M., Garcia, J. L. & Olivera, E. R. The phenylacetyl-CoA catabolon: a complex catabolic unit with broad biotechnological applications. Mol. Microbiol. 39, 1434–1442 (2001)
Ismail, W. et al. Functional genomics by NMR spectroscopy. Phenylacetate catabolism in Escherichia coli. Eur. J. Biochem. 270, 3047–3054 (2003)
Grishin, A. M. et al. Structural and functional studies of the Escherichia coli phenylacetyl-CoA monooxygenase complex. J. Biol. Chem. 286, 10735–10743 (2011)
Fernández, C., Ferrandez, A., Minambres, B., Diaz, E. & Garcia, J. L. Genetic characterization of the phenylacetyl-coenzyme A oxygenase from the aerobic phenylacetic acid degradation pathway of Escherichia coli. Appl. Environ. Microbiol. 72, 7422–7426 (2006)
Teufel, R. et al. Studies on the mechanism of ring hydrolysis in phenylacetate degradation: a metabolic branching point. J. Biol. Chem. 286, 11021–11034 (2011)
Geng, H. & Belas, R. Expression of tropodithietic acid biosynthesis is controlled by a novel autoinducer. J. Bacteriol. 192, 4377–4387 (2010)
Thiel, V. et al. Identification and biosynthesis of tropone derivatives and sulfur volatiles produced by bacteria of the marine Roseobacter clade. Org. Biomol. Chem. 8, 234–246 (2010)
Moore, B. S. et al. Biosynthetic studies of omega-cycloheptyl fatty acids in Alicyclobacillus cycloheptanicus. Formation of cycloheptanecarboxylic acid from phenylacetic acid. J. Org. Chem. 62, 2173–2185 (1997)
Law, R. J. et al. A functional phenylacetic acid catabolic pathway is required for full pathogenicity of Burkholderia cenocepacia in the Caenorhabditis elegans host model. J. Bacteriol. 190, 7209–7218 (2008)
Hamlin, J. N., Bloodworth, R. A. & Cardona, S. T. Regulation of phenylacetic acid degradation genes of Burkholderia cenocepacia K56–2. BMC Microbiol. 9, 222 (2009)
Khomyakova, M., Bükmez, O., Thomas, L. K., Erb, T. J. & Berg, I. A. A methylaspartate cycle in haloarchaea. Science 331, 334–337 (2011)
Leahy, J. G., Batchelor, P. J. & Morcomb, S. M. Evolution of the soluble diiron monooxygenases. FEMS Microbiol. Rev. 27, 449–479 (2003)
Nordlund, P. & Eklund, H. Di-iron-carboxylate proteins. Curr. Opin. Struct. Biol. 5, 758–766 (1995)
Rather, L. J., Knapp, B., Haehnel, W. & Fuchs, G. Coenzyme A-dependent aerobic metabolism of benzoate via epoxide formation. J. Biol. Chem. 285, 20615–20624 (2010)
Rather, L. J. et al. Structure and mechanism of the diiron benzoyl-coenzyme A epoxidase BoxB. J. Biol. Chem. 286, 29241–29248 (2011)
Rosenzweig, A. C., Frederick, C. A., Lippard, S. J. & Nordlund, P. Crystal structure of a bacterial non-haem iron hydroxylase that catalyses the biological oxidation of methane. Nature 366, 537–543 (1993)
Lee, S. K., Nesheim, J. C. & Lipscomb, J. D. Transient intermediates of the methane monooxygenase catalytic cycle. J. Biol. Chem. 268, 21569–21577 (1993)
Rosenzweig, A. C., Nordlund, P., Takahara, P. M., Frederick, C. A. & Lippard, S. J. Geometry of the soluble methane monooxygenase catalytic diiron center in two oxidation states. Chem. Biol. 2, 409–418 (1995)
Shu, L. et al. An Fe2IVO2 diamond core structure for the key intermediate Q of methane monooxygenase. Science 275, 515–518 (1997)
Rowe, G. T., Rybak-Akimova, E. V. & Caradonna, J. P. Unraveling the reactive species of a functional non-heme iron monooxygenase model using stopped-flow UV-vis spectroscopy. Inorg. Chem. 46, 10594–10606 (2007)
Rowe, G. T., Rybak-Akimova, E. V. & Caradonna, J. P. Heterolytic cleavage of peroxide by a diferrous compound generates metal-based intermediates identical to those observed with reactions utilizing oxygen-atom-donor molecules. Chemistry 14, 8303–8311 (2008)
Phillips, D. H. & Farmer, P. B. Evidence for DNA and protein binding by styrene and styrene oxide. Crit. Rev. Toxicol. 24, S35–S46 (1994)
Park, J. B. et al. The efficiency of recombinant Escherichia coli as biocatalyst for stereospecific epoxidation. Biotechnol. Bioeng. 95, 501–512 (2006)
Bennett, B. D. et al. Absolute metabolite concentrations and implied enzyme active site occupancy in Escherichia coli. Nature Chem. Biol. 5, 593–599 (2009)
Fuhrer, T. & Sauer, U. Different biochemical mechanisms ensure network-wide balancing of reducing equivalents in microbial metabolism. J. Bacteriol. 191, 2112–2121 (2009)
Song, F. et al. Structure, function, and mechanism of the phenylacetate pathway hot dog-fold thioesterase PaaI. J. Biol. Chem. 281, 11028–11038 (2006)
Johansson, A. J., Noack, H., Siegbahn, P. E., Xue, G. & Que, L., Jr Observed enhancement of the reactivity of a biomimetic diiron complex by the addition of water — mechanistic insights from theoretical modeling. Dalton Trans. 6741–6750 (2009)
Xue, G., De Hont, R., Munck, E. & Que, L., Jr Million-fold activation of the [Fe2(μ-O)2] diamond core for C–H bond cleavage. Nature Chem. 2, 400–405 (2010)
Gallagher, S. C., Cammack, R. & Dalton, H. Alkene monooxygenase from Nocardia corallina B-276 is a member of the class of dinuclear iron proteins capable of stereospecific epoxygenation reactions. Eur. J. Biochem. 247, 635–641 (1997)
Erb, T. J. et al. Synthesis of C5-dicarboxylic acids from C2-units involving crotonyl-CoA carboxylase/reductase: the ethylmalonyl-CoA pathway. Proc. Natl Acad. Sci. USA 104, 10631–10636 (2007)
Erb, T. J., Ismail, W. & Fuchs, G. Phenylacetate metabolism in thermophiles: characterization of phenylacetate-CoA ligase, the initial enzyme of the hybrid pathway in Thermus thermophilus. Curr. Microbiol. 57, 27–32 (2008)
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970)
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976)
Ramos-Vera, W. H., Weiss, M., Strittmatter, E., Kockelkorn, D. & Fuchs, G. Identification of missing genes and enzymes for autotrophic carbon fixation in crenarchaeota. J. Bacteriol. 193, 1201–1211 (2011)
Textor, S. et al. Propionate oxidation in Escherichia coli: evidence for operation of a methylcitrate cycle in bacteria. Arch. Microbiol. 168, 428–436 (1997)
Beinert, H. & Thomson, A. J. Three-iron clusters in iron-sulfur proteins. Arch. Biochem. Biophys. 222, 333–361 (1983)
Teufel, R., Kung, J. W., Kockelkorn, D., Alber, B. E. & Fuchs, G. 3-hydroxypropionyl-coenzyme A dehydratase and acryloyl-coenzyme A reductase, enzymes of the autotrophic 3-hydroxypropionate/4-hydroxybutyrate cycle in the Sulfolobales. J. Bacteriol. 191, 4572–4581 (2009)
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990)
Tamura, K., Dudley, J., Nei, M. & Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol. 24, 1596–1599 (2007)
Saitou, N. & Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425 (1987)
Acknowledgements
We thank Deutsche Forschungsgemeinschaft for support; V. Mascaraque, G. Heijman and W. Ismail for their contributions in the initial stage of this work; M. Voss, M. Weiß, I. Sachelaru and T. Spatzal for technical assistance; M. Müller and V. Brecht for mass spectrometry analysis; and I. Berg and H. Heider for critical comments.
Author information
Authors and Affiliations
Contributions
R.T. and T.F. performed research; R.T, T.F. and G.F designed research and analysed data; and R.T. and G.F. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-11, Supplementary Tables 1-4, a Supplementary Discussion and additional references. Supplementary Figure 11 shows PaaZ inhibition by the derivative and Supplementary Table 4 substrate specificity of thioesterases PaaI and PaaY. (PDF 2463 kb)
Rights and permissions
About this article
Cite this article
Teufel, R., Friedrich, T. & Fuchs, G. An oxygenase that forms and deoxygenates toxic epoxide. Nature 483, 359–362 (2012). https://doi.org/10.1038/nature10862
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10862
This article is cited by
-
Ungewöhnliche Flavoenzyme steuern die Bildung von Troponen in Bakterien
BIOspektrum (2022)
-
A soil-borne Mn(II)-oxidizing bacterium of Providencia sp. exploits a strategy of superoxide production coupled to hydrogen peroxide consumption to generate Mn oxides
Archives of Microbiology (2022)
-
Structural basis for biomolecular recognition in overlapping binding sites in a diiron enzyme system
Nature Communications (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.