Abstract
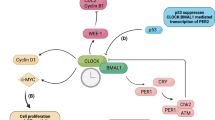
Sudden cardiac death exhibits diurnal variation in both acquired and hereditary forms of heart disease1,2, but the molecular basis of this variation is unknown. A common mechanism that underlies susceptibility to ventricular arrhythmias is abnormalities in the duration (for example, short or long QT syndromes and heart failure)3,4,5 or pattern (for example, Brugada’s syndrome)6 of myocardial repolarization. Here we provide molecular evidence that links circadian rhythms to vulnerability in ventricular arrhythmias in mice. Specifically, we show that cardiac ion-channel expression and QT-interval duration (an index of myocardial repolarization) exhibit endogenous circadian rhythmicity under the control of a clock-dependent oscillator, krüppel-like factor 15 (Klf15). Klf15 transcriptionally controls rhythmic expression of Kv channel-interacting protein 2 (KChIP2), a critical subunit required for generating the transient outward potassium current7. Deficiency or excess of Klf15 causes loss of rhythmic QT variation, abnormal repolarization and enhanced susceptibility to ventricular arrhythmias. These findings identify circadian transcription of ion channels as a mechanism for cardiac arrhythmogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Muller, J. E. et al. Circadian variation in the frequency of sudden cardiac death. Circulation 75, 131–138 (1987)
Matsuo, K. et al. The circadian pattern of the development of ventricular fibrillation in patients with Brugada syndrome. Eur. Heart J. 20, 465–470 (1999)
Goldenberg, I. & Moss, A. J. Long QT syndrome. J. Am. Coll. Cardiol. 51, 2291–2300 (2008)
Patel, U. & Pavri, B. B. Short QT syndrome: a review. Cardiol. Rev. 17, 300–303 (2009)
Tomaselli, G. F. & Marban, E. Electrophysiological remodeling in hypertrophy and heart failure. Cardiovasc. Res. 42, 270–283 (1999)
Antzelevitch, C. Role of spatial dispersion of repolarization in inherited and acquired sudden cardiac death syndromes. Am. J. Physiol. Heart Circ. Physiol. 293, H2024–H2038 (2007)
Kuo, H. C. et al. A defect in the Kv channel-interacting protein 2 (KChIP2) gene leads to a complete loss of Ito and confers susceptibility to ventricular tachycardia. Cell 107, 801–813 (2001)
Antzelevitch, C. & Yan, G. X. J wave syndromes. Heart Rhythm 7, 549–558 (2010)
Reppert, S. M. & Weaver, D. R. Coordination of circadian timing in mammals. Nature 418, 935–941 (2002)
Bexton, R. S., Vallin, H. O. & Camm, A. J. Diurnal variation of the QT interval—influence of the autonomic nervous system. Br. Heart J. 55, 253–258 (1986)
Kong, T. Q., Jr, Goldberger, J. J., Parker, M., Wang, T. & Kadish, A. H. Circadian variation in human ventricular refractoriness. Circulation 92, 1507–1516 (1995)
Martino, T. A. & Sole, M. J. Molecular time: an often overlooked dimension to cardiovascular disease. Circ. Res. 105, 1047–1061 (2009)
Paschos, G. K. & FitzGerald, G. A. Circadian clocks and vascular function. Circ. Res. 106, 833–841 (2010)
Durgan, D. J. & Young, M. E. The cardiomyocyte circadian clock: emerging roles in health and disease. Circ. Res. 106, 647–658 (2010)
Yamashita, T. et al. Circadian variation of cardiac K+ channel gene expression. Circulation 107, 1917–1922 (2003)
Haldar, S. M. et al. Klf15 deficiency is a molecular link between heart failure and aortic aneurysm formation. Sci. Transl. Med. 2, 26ra26 (2010)
Wang, B. et al. The Krüppel-like factor KLF15 inhibits connective tissue growth factor (CTGF) expression in cardiac fibroblasts. J. Mol. Cell. Cardiol. 45, 193–197 (2008)
Fisch, S. et al. Krüppel-like factor 15 is a regulator of cardiomyocyte hypertrophy. Proc. Natl Acad. Sci. USA 104, 7074–7079 (2007); correction. 104, 13851 (2007)
Bunger, M. K. et al. Mop3 is an essential component of the master circadian pacemaker in mammals. Cell 103, 1009–1017 (2000)
Sanbe, A. et al. Reengineering inducible cardiac-specific transgenesis with an attenuated myosin heavy chain promoter. Circ. Res. 92, 609–616 (2003)
Tuteja, G., Jensen, S. T., White, P. & Kaestner, K. H. Cis-regulatory modules in the mammalian liver: composition depends on strength of Foxa2 consensus site. Nucleic Acids Res. 36, 4149–4157 (2008)
Ripperger, J. A. & Schibler, U. Rhythmic CLOCK-BMAL1 binding to multiple E-box motifs drives circadian Dbp transcription and chromatin transitions. Nature Genet. 38, 369–374 (2006)
Mitchell, G. F., Jeron, A. & Koren, G. Measurement of heart rate and Q-T interval in the conscious mouse. Am. J. Physiol. 274, H747–H751 (1998)
Libbus, I., Wan, X. & Rosenbaum, D. S. Electrotonic load triggers remodeling of repolarizing current Ito in ventricle. Am. J. Physiol. Heart Circ. Physiol. 286, H1901–H1909 (2004)
Wagner, S. et al. Ca/calmodulin kinase II differentially modulates potassium currents. Circ. Arrhythm. Electrophysiol. 2, 285–294 (2009)
van Oort, R. J. et al. Ryanodine receptor phosphorylation by calcium/calmodulin-dependent protein kinase II promotes life-threatening ventricular arrhythmias in mice with heart failure. Circulation 122, 2669–2679 (2010)
Nelson, W., Tong, Y. L., Lee, J. K. & Halberg, F. Methods for cosinor-rhythmometry. Chronobiologia 6, 305–323 (1979)
Hu, K., Scheer, F. A., Laker, M., Smales, C. & Shea, S. A. Endogenous circadian rhythm in vasovagal response to head-up tilt. Circulation 123, 961–970 (2011)
Acknowledgements
We thank A. F. Connors Jr for support, M. Mustar for illustrations, Y. Cui for experimental assistance, and members of the Jain laboratory for discussions. Funding sources: Heart Rhythm Society Fellowship (D.J.); National Institutes of Health grants HL094660 (D.J.), HL066991 (M.D.M.), HL086614 (S.M.H.), American Heart Association postdoctoral grant (N.S.), HL089598, HL091947 (X.H.W.), HL76446 (S.A.S.), HL102241 (K.H.), HL054807 (D.S.R.), HL075427, HL076754, HL084154, HL086548 and HL097595 (M.K.J.); Swiss National Science Foundation grants 31003A/131086 (U.A.) and M01-RR02635 (B.W.H.); Leducq Foundation grants of the ENAFRA Network 07CVD03 (S.D.); and the Centre National de la Recherche Scientifique (S.D.).
Author information
Authors and Affiliations
Contributions
D.J. and M.K.J. designed the research; D.J., S.M.H., X.W., M.D.M., J.A.R., Y.L., B.L.E. and M.J.C. carried out the experiments; J.G., A.S., J.R. and R.V.K. contributed critical reagents; D.J., N.S., S.D., R.V.K., S.A.S., U.A., X.H.T.W., D.S.R. and M.K.J. supervised the research; D.J., S.M.H., X.W., M.D.M., J.A.R., K.H., B.L.E., E.F., S.A.S., U.A., X.H.T.W., D.S.R. and M.K.J. analysed and interpreted the data; and D.J. and M.K.J. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-13 with legends. (PDF 351 kb)
Rights and permissions
About this article
Cite this article
Jeyaraj, D., Haldar, S., Wan, X. et al. Circadian rhythms govern cardiac repolarization and arrhythmogenesis. Nature 483, 96–99 (2012). https://doi.org/10.1038/nature10852
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10852
This article is cited by
-
Circadian rhythm in cardiovascular diseases: a bibliometric analysis of the past, present, and future
European Journal of Medical Research (2023)
-
Branched-chain amino acids in cardiovascular disease
Nature Reviews Cardiology (2023)
-
Amplitude response and singularity behavior of circadian clock to external stimuli
npj Systems Biology and Applications (2023)
-
A defect in mitochondrial protein translation influences mitonuclear communication in the heart
Nature Communications (2023)
-
Association of accelerometer-derived circadian abnormalities and genetic risk with incidence of atrial fibrillation
npj Digital Medicine (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.