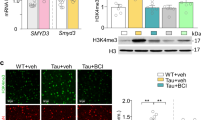
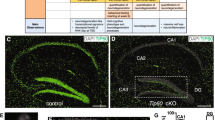
Abstract
Cognitive decline is a debilitating feature of most neurodegenerative diseases of the central nervous system, including Alzheimer’s disease1. The causes leading to such impairment are only poorly understood and effective treatments are slow to emerge2. Here we show that cognitive capacities in the neurodegenerating brain are constrained by an epigenetic blockade of gene transcription that is potentially reversible. This blockade is mediated by histone deacetylase 2, which is increased by Alzheimer’s-disease-related neurotoxic insults in vitro, in two mouse models of neurodegeneration and in patients with Alzheimer’s disease. Histone deacetylase 2 associates with and reduces the histone acetylation of genes important for learning and memory, which show a concomitant decrease in expression. Importantly, reversing the build-up of histone deacetylase 2 by short-hairpin-RNA-mediated knockdown unlocks the repression of these genes, reinstates structural and synaptic plasticity, and abolishes neurodegeneration-associated memory impairments. These findings advocate for the development of selective inhibitors of histone deacetylase 2 and suggest that cognitive capacities following neurodegeneration are not entirely lost, but merely impaired by this epigenetic blockade.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Walsh, D. M. & Selkoe, D. J. Deciphering the molecular basis of memory failure in Alzheimer’s disease. Neuron 44, 181–193 (2004)
Holtzman, D. M., Morris, J. C. & Goate, A. M. Alzheimer’s disease: the challenge of the second century. Sci. Transl. Med. 3, 77sr1 (2011)
Kandel, E. R. The molecular biology of memory storage: a dialogue between genes and synapses. Science 294, 1030–1038 (2001)
Gräff, J., Kim, D., Dobbin, M. M. & Tsai, L. H. Epigenetic regulation of gene expression in physiological and pathological brain processes. Physiol. Rev. 91, 603–649 (2011)
Brownell, J. E. & Allis, C. D. Special HATs for special occasions: linking histone acetylation to chromatin assembly and gene activation. Curr. Opin. Genet. Dev. 6, 176–184 (1996)
Kazantsev, A. G. & Thompson, L. M. Therapeutic application of histone deacetylase inhibitors for central nervous system disorders. Nature Rev. Drug Discov. 7, 854–868 (2008)
Guan, J. S. et al. HDAC2 negatively regulates memory formation and synaptic plasticity. Nature 459, 55–60 (2009)
Akhtar, M. W. et al. Histone deacetylases 1 and 2 form a developmental switch that controls excitatory synapse maturation and function. J. Neurosci. 29, 8288–8297 (2009)
Cruz, J. C., Tseng, H. C., Goldman, J. A., Shih, H. & Tsai, L. H. Aberrant Cdk5 activation by p25 triggers pathological events leading to neurodegeneration and neurofibrillary tangles. Neuron 40, 471–483 (2003)
Cruz, J. C. et al. p25/cyclin-dependent kinase 5 induces production and intraneuronal accumulation of amyloid beta in vivo. J. Neurosci. 26, 10536–10541 (2006)
Cruz, J. C. & Tsai, L. H. A Jekyll and Hyde kinase: roles for Cdk5 in brain development and disease. Curr. Opin. Neurobiol. 14, 390–394 (2004)
Patrick, G. N. et al. Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402, 615–622 (1999)
Fischer, A., Sananbenesi, F., Pang, P. T., Lu, B. & Tsai, L. H. Opposing roles of transient and prolonged expression of p25 in synaptic plasticity and hippocampus-dependent memory. Neuron 48, 825–838 (2005)
Frankland, P. W. & Bontempi, B. The organization of recent and remote memories. Nature Rev. Neurosci. 6, 119–130 (2005)
McQuown, S. C. et al. HDAC3 is a critical negative regulator of long-term memory formation. J. Neurosci. 31, 764–774 (2011)
Oakley, H. et al. Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: potential factors in amyloid plaque formation. J. Neurosci. 26, 10129–10140 (2006)
Kimura, R., Devi, L. & Ohno, M. Partial reduction of BACE1 improves synaptic plasticity, recent and remote memories in Alzheimer’s disease transgenic mice. J. Neurochem. 113, 248–261 (2010)
Lauterborn, J. C. et al. Differential effects of protein synthesis inhibition on the activity-dependent expression of BDNF transcripts: evidence for immediate-early gene responses from specific promoters. J. Neurosci. 16, 7428–7436 (1996)
Sun, J. M., Chen, H. Y. & Davie, J. R. Differential distribution of unmodified and phosphorylated histone deacetylase 2 in chromatin. J. Biol. Chem. 282, 33227–33236 (2007)
Fischer, A., Sananbenesi, F., Wang, X. Y., Dobbin, M. & Tsai, L. H. Recovery of learning and memory is associated with chromatin remodelling. Nature 447, 178–182 (2007)
Markesbery, W. R. & Carney, J. M. Oxidative alterations in Alzheimer’s disease. Brain Pathol. 9, 133–146 (1999)
Yankner, B. A. Mechanisms of neuronal degeneration in Alzheimer’s disease. Neuron 16, 921–932 (1996)
Sandelin, A., Alkema, W., Engstrom, P., Wasserman, W. W. & Lenhard, B. JASPAR: an open-access database for eukaryotic transcription factor binding profiles. Nucleic Acids Res. 32, D91–D94 (2004)
Chrousos, G. P. & Kino, T. Glucocorticoid signaling in the cell. Expanding clinical implications to complex human behavioral and somatic disorders. Ann. NY Acad. Sci. 1179, 153–166 (2009)
Kino, T. et al. Cyclin-dependent kinase 5 differentially regulates the transcriptional activity of the glucocorticoid receptor through phosphorylation: clinical implications for the nervous system response to glucocorticoids and stress. Mol. Endocrinol. 21, 1552–1568 (2007)
Guan, J. S. et al. Cdk5 is required for memory function and hippocampal plasticity via the cAMP signaling pathway. PLoS ONE 6, e25735 (2011)
Citron, M. Alzheimer’s disease: strategies for disease modification. Nature Rev. Drug Discov. 9, 387–398 (2010)
Braak, H. & Braak, E. Diagnostic criteria for neuropathologic assessment of Alzheimer’s disease. Neurobiol. Aging 18, S85–S88 (1997)
Grober, E. et al. Memory and mental status correlates of modified Braak staging. Neurobiol. Aging 20, 573–579 (1999)
Palop, J. J. & Mucke, L. Amyloid-beta-induced neuronal dysfunction in Alzheimer’s disease: from synapses toward neural networks. Nature Neurosci. 13, 812–818 (2009)
Qi, Y., Wang, J. K., McMillian, M. & Chikaraishi, D. M. Characterization of a CNS cell line, CAD, in which morphological differentiation is initiated by serum deprivation. J. Neurosci. 17, 1217–1225 (1997)
Li, L. & Lindquist, S. Creating a protein-based element of inheritance. Science 287, 661–664 (2000)
Xie, Z., Sanada, K., Samuels, B. A., Shih, H. & Tsai, L. H. Serine 732 phosphorylation of FAK by Cdk5 is important for microtubule organization, nuclear movement, and neuronal migration. Cell 114, 469–482 (2003)
Koshibu, K. et al. Protein phosphatase 1 regulates the histone code for long-term memory. J. Neurosci. 29, 13079–13089 (2009)
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 - Δ Δ C T . Methods 25, 402–408 (2001)
Moffat, J. et al. A lentiviral RNAi library for human and mouse genes applied to an arrayed viral high-content screen. Cell 124, 1283–1298 (2006)
Sarbassov, D. D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005)
Price, J. L. et al. Neuropathology of nondemented aging: presumptive evidence for preclinical Alzheimer disease. Neurobiol. Aging 30, 1026–1036 (2009)
Acknowledgements
We thank A. Mungenast, S. Jemielity, R. Madabushi, F. Calderon de Anda and M. Horn for reading the manuscript, A.M. for manuscript editing, M. Eichler for mouse colony maintenance, K. Fitch for sectioning the human brain samples and M.H. for quantification of Fig. 2a. This work was partly supported by the Stanley Medical Research Institution (to S.J.H. and L.-H.T.), National Institutes of Health/National Institute on Drug Abuse (RO1DA028301, to S.J.H.) and National Institutes of Health/National Institute of Neurological Disorders and Stroke (RO1NS078839, to L.-H.T.). J.G. was supported by a Bard Richmond fellowship and by the Swiss National Science Foundation, W.Y.W. by the Simons Foundation and M.K. by the Theodor und Ida Herzog-Egli foundation. L.-H.T. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
This study was designed by J.G. and L.-H.T., and directed and coordinated by L.-H.T. J.G. planned and performed the in vitro, CK-p25 and Cdk5cKO mouse and human in vivo biochemical characterization, and all behavioural experiments. D.R. planned and contributed to the in vitro and CK-p25 in vivo experiments, generated the GR526 and the shRNA constructs, and contributed to the stereotaxic injections. J.S.G. initiated and contributed to the CK-p25 biochemical characterization, and performed the 5XFAD and HDAC2−/− experiments. W.Y.W. generated the luciferase constructs. J.S. performed the electrophysiological experiments. K.M.H., T.J.F.N., D.F. and S.J.H. characterized the shRNA constructs. M.K. contributed to the quantitative RT–PCR experiments and performed the quantification of the human data. S.C.S. performed the site-directed mutagenesis. A.S. contributed to the immunohistochemistry and the quantitative reverse transcription/quantitative PCR experiments. N.J. contributed to the behavioural and quantitative reverse transcription/quantitative PCR experiments. P.F.K. and I.D. provided the human samples and contributed to the optimization of their staining. The manuscript was written by J.G. and L.-H.T. and commented on by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-19 with legends and Supplementary Tables 1-5. (PDF 1357 kb)
Rights and permissions
About this article
Cite this article
Gräff, J., Rei, D., Guan, JS. et al. An epigenetic blockade of cognitive functions in the neurodegenerating brain. Nature 483, 222–226 (2012). https://doi.org/10.1038/nature10849
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10849
This article is cited by
-
Histone acetylation in an Alzheimer’s disease cell model promotes homeostatic amyloid-reducing pathways
Acta Neuropathologica Communications (2024)
-
Histone deacetylase inhibitors VPA and WT161 ameliorate the pathological features and cognitive impairments of the APP/PS1 Alzheimer’s disease mouse model by regulating the expression of APP secretases
Alzheimer's Research & Therapy (2024)
-
The Role of Acetylation and Methylation of Rat Hippocampal Histone H3 in the Mechanism of Aluminum-Induced Neurotoxicity
Neurochemical Research (2024)
-
Dysregulation of histone deacetylases in ocular diseases
Archives of Pharmacal Research (2024)
-
Alzheimer’s Disease-Related Epigenetic Changes: Novel Therapeutic Targets
Molecular Neurobiology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.