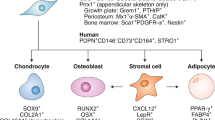
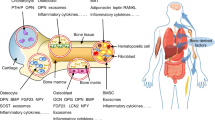
Abstract
The mouse genetic revolution has shown repeatedly that most organs have more functions than expected. This has led to the realization that, in addition to a molecular and cellular approach, there is a need for a whole-organism study of physiology. The skeleton is an example of how a whole-organism approach to physiology can broaden the functions of a given organ, reveal connections of this organ with others such as the brain, pancreas and gut, and shed new light on the pathogenesis of degenerative diseases affecting multiple organs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bernard, C. Introduction à l'Etude de la Médecine Expérimentale (Flammarion, 1865).
Gurney, C. W. Erythropoietin, erythropoiesis, and the kidney. J. Am. Med. Assoc. 173, 1828–1829 (1960).
Friedman, J. M. & Halaas, J. L. Leptin and the regulation of body weight in mammals. Nature 395, 763–770 (1998).
Chen, S. K. et al. Hematopoietic origin of pathological grooming in Hoxb8 mutant mice. Cell 141, 775–785 (2010).
Cannon, W. B. The Wisdom of the Body (Norton, 1932).
Monod, J. & Jacob, F. Teleonomic mechanisms in cellular metabolism, growth, and differentiation. Cold Spring Harb. Symp. Quant. Biol. 26, 389–401 (1961).
Teitelbaum, S. L. Bone resorption by osteoclasts. Science 289, 1504–1508 (2000).
Ducy, P., Schinke, T. & Karsenty, G. The osteoblast: a sophisticated fibroblast under central surveillance. Science 289, 1501–1504 (2000).
Legroux-Gerot, I., Vignau, J., Collier, F. & Cortet, B. Bone loss associated with anorexia nervosa. Joint Bone Spine 72, 489–495 (2005).
Misra, M. & Klibanski, A. The neuroendocrine basis of anorexia nervosa and its impact on bone metabolism. Neuroendocrinology 93, 65–73 (2011).
Riggs, B. L., Khosla, S. & Melton, L. J. A unitary model for involutional osteoporosis: estrogen deficiency causes both type I and type II osteoporosis in postmenopausal women and contributes to bone loss in aging men. J. Bone Miner. Res. 13, 763–773 (1998).
Riggs, B. L. & Melton, L. J. Involutional osteoporosis. N. Engl. J. Med. 314, 1676–1686 (1986).
Hauschka, P. V., Lian, J. B., Cole, D. E. & Gundberg, C. M. Osteocalcin and matrix Gla protein: vitamin K-dependent proteins in bone. Physiol. Rev. 69, 990–1047 (1989).
Oury, F. et al. Endocrine regulation of male fertility by the skeleton. Cell 144, 796–809 (2011).
Lee, N. K. et al. Endocrine regulation of energy metabolism by the skeleton. Cell 130, 456–469 (2007). This paper describes for the first time the endocrine function of bone.
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 425–432 (1994). This report describes the identification of the leptin gene.
Halaas, J. L. et al. Weight-reducing effects of the plasma protein encoded by the obese gene. Science 269, 543–546 (1995).
Chehab, F. F., Lim, M. E. & Lu, R. Correction of the sterility defect in homozygous obese female mice by treatment with the human recombinant leptin. Nature Genet. 12, 318–320 (1996).
Ducy, P. et al. Leptin inhibits bone formation through a hypothalamic relay: a central control of bone mass. Cell 100, 197–207 (2000).
Pogoda, P. et al. Leptin inhibits bone formation not only in rodents, but also in sheep. J. Bone Miner. Res. 21, 1591–1599 (2006).
Elefteriou, F. et al. Serum leptin level is a regulator of bone mass. Proc. Natl Acad. Sci. USA 101, 3258–3263 (2004).
Gibson, W. T. et al. Congenital leptin deficiency due to homozygosity for the Δ133G mutation: report of another case and evaluation of response to four years of leptin therapy. J. Clin. Endocrinol. Metab. 89, 4821–4826 (2004).
Bjorbaek, C., Elmquist, J. K., Frantz, J. D., Shoelson, S. E. & Flier, J. S. Identification of SOCS-3 as a potential mediator of central leptin resistance. Mol. Cell 1, 619–625 (1998).
Bjornholm, M. et al. Mice lacking inhibitory leptin receptor signals are lean with normal endocrine function. J. Clin. Invest. 117, 1354–1360 (2007).
Shi, Y. et al. Dissociation of the neuronal regulation of bone mass and energy metabolism by leptin in vivo . Proc. Natl Acad. Sci. USA 105, 20529–20533 (2008).
Baldock, P. A. et al. Neuropeptide Y knockout mice reveal a central role of NPY in the coordination of bone mass to body weight. PLoS ONE 4, e8415 (2009).
Sato, S. et al. Central control of bone remodeling by neuromedin U. Nature Med. 13, 1234–1240 (2007).
Cornish, J. et al. Leptin directly regulates bone cell function in vitro and reduces bone fragility in vivo . J. Endocrinol. 175, 405–415 (2002).
Bartell, S. M. et al. Central (ICV) leptin injection increases bone formation, bone mineral density, muscle mass, serum IGF-1, and the expression of osteogenic genes in leptin-deficient ob/ob mice. J. Bone Miner. Res. 26, 1710–1720 (2011).
Takeda, S. et al. Leptin regulates bone formation via the sympathetic nervous system. Cell 111, 305–317 (2002). This paper reports that leptin regulation of bone mass requires the sympathetic nervous system.
Yadav, V. K. et al. A serotonin-dependent mechanism explains the leptin regulation of bone mass, appetite, and energy expenditure. Cell 138, 976–989 (2009). This study identifies brain serotonin as a critical mediator of the central action of leptin.
Balthasar, N. et al. Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron 42, 983–991 (2004).
Dhillon, H. et al. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 49, 191–203 (2006).
Warden, S. J. et al. Psychotropic drugs have contrasting skeletal effects that are independent of their effects on physical activity levels. Bone 46, 985–992 (2010).
Bliziotes, M. Update in serotonin and bone. J. Clin. Endocrinol. Metab. 95, 4124–4132 (2010).
Kaye, W., Gendall, K. & Strober, M. Serotonin neuronal function and selective serotonin reuptake inhibitor treatment in anorexia and bulimia nervosa. Biol. Psychiatry 44, 825–838 (1998).
Mann, J. J. et al. Relationship between central and peripheral serotonin indexes in depressed and suicidal psychiatric inpatients. Arch. Gen. Psychiatry 49, 442–446 (1992).
Walther, D. J. et al. Synthesis of serotonin by a second tryptophan hydroxylase isoform. Science 299, 76 (2003). This report describes the cloning of the Tph2 gene, which is responsible for brain serotonin synthesis.
Oury, F. et al. CREB mediates brain serotonin regulation of bone mass through its expression in ventromedial hypothalamic neurons. Genes Dev. 24, 2330–2342 (2010).
Yadav, V. K. et al. Leptin-dependent serotonin control of appetite: temporal specificity, transcriptional regulation, and therapeutic implications. J. Exp. Med. 208, 41–52 (2011).
Lam, D. D. et al. Leptin does not directly affect CNS serotonin neurons to influence appetite. Cell Metab. 13, 584–591 (2011).
Elefteriou, F. et al. Leptin regulation of bone resorption by the sympathetic nervous system and CART. Nature 434, 514–520 (2005).
Bonnet, N. et al. Protective effect of beta blockers in postmenopausal women: influence on fractures, bone density, micro and macroarchitecture. Bone 40, 1209–1216 (2007).
ADHR Consortium. Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23 . Nature Genet. 26, 345–348 (2000).10.1038/81664
Hori, M., Shimizu, Y. & Fukumoto, S. Fibroblast growth factor 23 in phosphate homeostasis and bone metabolism. Endocrinology 152, 4–10 (2011).
Motyl, K. J., McCabe, L. R. & Schwartz, A. V. Bone and glucose metabolism: a two-way street. Arch. Biochem. Biophys. 503, 2–10 (2010).
Pollock, N. K. et al. Lower uncarboxylated osteocalcin concentrations in children with prediabetes is associated with beta-cell function. J. Clin. Endocrinol. Metab. 96, E1092–E1099 (2011).
Pi, M. et al. Identification of a novel extracellular cation-sensing G-protein-coupled receptor. J. Biol. Chem. 280, 40201–40209 (2005).
Merle, B. & Delmas, P. D. Normal carboxylation of circulating osteocalcin (bone Gla-protein) in Paget's disease of bone. Bone Miner. 11, 237–245 (1990).
Ferron, M., Hinoi, E., Karsenty, G. & Ducy, P. Osteocalcin differentially regulates beta cell and adipocyte gene expression and affects the development of metabolic diseases in wild-type mice. Proc. Natl Acad. Sci. USA 105, 5266–5270 (2008).
Ferron, M. et al. Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell 142, 296–308 (2010). This study identifies a feed-forward loop linking insulin and osteocalcin.
Fulzele, K. et al. Insulin receptor signaling in osteoblasts regulates postnatal bone acquisition and body composition. Cell 142, 309–319 (2010). In this report, insulin signalling in osteoblasts is identified as a key regulator of bone mass and of energy metabolism.
Bluher, M. et al. Adipose tissue selective insulin receptor knockout protects against obesity and obesity-related glucose intolerance. Dev. Cell 3, 25–38 (2002).
Bruning, J. C. et al. A muscle-specific insulin receptor knockout exhibits features of the metabolic syndrome of NIDDM without altering glucose tolerance. Mol. Cell 2, 559–569 (1998).
Silver, I. A., Murrills, R. J. & Etherington, D. J. Microelectrode studies on the acid microenvironment beneath adherent macrophages and osteoclasts. Exp. Cell Res. 175, 266–276 (1988).
Poser, J. W. & Price, P. A. A method for decarboxylation of γ-carboxyglutamic acid in proteins. Properties of the decarboxylated γ-carboxyglutamic acid protein from calf bone. J. Biol. Chem. 254, 431–436 (1979).
Elchebly, M. et al. Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1B gene. Science 283, 1544–1548 (1999).
Delibegovic, M. et al. Liver-specific deletion of protein-tyrosine phosphatase 1B (PTP1B) improves metabolic syndrome and attenuates diet-induced endoplasmic reticulum stress. Diabetes 58, 590–599 (2009).
Delibegovic, M. et al. Improved glucose homeostasis in mice with muscle-specific deletion of protein-tyrosine phosphatase 1B. Mol. Cell. Biol. 27, 7727–7734 (2007).
Hinoi, E. et al. The sympathetic tone mediates leptin's inhibition of insulin secretion by modulating osteocalcin bioactivity. J. Cell Biol. 183, 1235–1242 (2008).
Miheller, P., Lorinczy, K. & Lakatos, P. L. Clinical relevance of changes in bone metabolism in inflammatory bowel disease. World J. Gastroenterol. 16, 5536–5542 (2010).
Rodriguez-Bores, L., Barahona-Garrido, J. & Yamamoto-Furusho, J. K. Basic and clinical aspects of osteoporosis in inflammatory bowel disease. World J. Gastroenterol. 13, 6156–6165 (2007).
Elefteriou, F. et al. ATF4 mediation of NF1 functions in osteoblast reveals a nutritional basis for congenital skeletal dysplasiae. Cell Metab. 4, 441–451 (2006).
Yang, X. et al. ATF4 is a substrate of RSK2 and an essential regulator of osteoblast biology; implication for Coffin–Lowry syndrome. Cell 117, 387–398 (2004).
Raisz, L. G. Stimulation of bone resorption by parathyroid hormone in tissue culture. Nature 197, 1015–1016 (1963).
Schinke, T. et al. Impaired gastric acidification negatively affects calcium homeostasis and bone mass. Nature Med. 15, 674–681 (2009).
Gong, Y. et al. LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell 107, 513–523 (2001). This paper identifies LRP5 as the gene mutated in OPPG.
Little, R. D. et al. A mutation in the LDL receptor-related protein 5 gene results in the autosomal dominant high-bone-mass trait. Am. J. Hum. Genet. 70, 11–19 (2002).
Boyden, L. M. et al. High bone density due to a mutation in LDL-receptor-related protein 5. N. Engl. J. Med. 346, 1513–1521 (2002). This study identifies a mutation in LRP5 as causing HBM syndrome in humans.
Kato, M. et al. Cbfa1-independent decrease in osteoblast proliferation, osteopenia, and persistent embryonic eye vascularization in mice deficient in Lrp5, a Wnt coreceptor. J. Cell Biol. 157, 303–314 (2002).
Tamai, K. et al. LDL-receptor-related proteins in Wnt signal transduction. Nature 407, 530–535 (2000).
Hay, E. et al. Interaction between LRP5 and Frat1 mediates the activation of the Wnt canonical pathway. J. Biol. Chem. 280, 13616–13623 (2005).
Tolwinski, N. S. et al. Wg/Wnt signal can be transmitted through Arrow/LRP5,6 and Axin independently of Zw3/Gsk3β activity. Dev. Cell 4, 407–418 (2003).
Li, X. et al. Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling. J. Biol. Chem. 280, 19883–19887 (2005).
Ye, X. et al. Norrin, frizzled-4, and Lrp5 signaling in endothelial cells controls a genetic program for retinal vascularization. Cell 139, 285–298 (2009).
Yadav, V. K. et al. Lrp5 controls bone formation by inhibiting serotonin synthesis in the duodenum. Cell 135, 825–837 (2008).
Jackson, A. et al. Gene array analysis of Wnt-regulated genes in C3H10T1/2 cells. Bone 36, 585–598 (2005).
Glass, D. A. et al. Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Dev. Cell 8, 751–764 (2005).
Kramer, I. et al. Osteocyte Wnt/β-catenin signaling is required for normal bone homeostasis. Mol. Cell. Biol. 30, 3071–3085 (2010).
Reya, T. & Clevers, H. Wnt signalling in stem cells and cancer. Nature 434, 843–850 (2005).
Cui, Y. et al. Lrp5 functions in bone to regulate bone mass. Nature Med. 17, 684–691 (2011).
Saarinen, A. et al. Low density lipoprotein receptor-related protein 5 (LRP5) mutations and osteoporosis, impaired glucose metabolism and hypercholesterolaemia. Clin. Endocrinol. 72, 481–488 (2010).
Frost, M. et al. Patients with high-bone-mass phenotype owing to Lrp5-T253I mutation have low plasma levels of serotonin. J. Bone Miner. Res. 25, 673–675 (2010).
Frost, M. et al. Levels of serotonin, sclerostin, bone turnover markers as well as bone density and microarchitecture in patients with high bone mass phenotype due to a mutation in Lrp5. J. Bone Miner. Res. 26, 1721–1728 (2011).
Vilaca, T., Yamamoto, R. M., Carvalho, A. B. & Lazaretti-Castro, M. Neuroendocrine tumor associated with severe osteoporosis in a male patient. Endocr Rev, 32, abstr. P3-123 (2011)
Modder, U. I. et al. Relation of serum serotonin levels to bone density and structural parameters in women. J. Bone Miner. Res. 25, 415–422 (2010).
Shi, Z. C. et al. Modulation of peripheral serotonin levels by novel tryptophan hydroxylase inhibitors for the potential treatment of functional gastrointestinal disorders. J. Med. Chem. 51, 3684–3687 (2008).
Liu, Q. et al. Discovery and characterization of novel tryptophan hydroxylase inhibitors that selectively inhibit serotonin synthesis in the gastrointestinal tract. J. Pharmacol. Exp. Ther. 325, 47–55 (2008).
Inose, H. et al. Efficacy of serotonin inhibition in mouse models of bone loss. J. Bone Miner. Res. 26, 2002–2011 (2011).
Yadav, V. K. et al. Pharmacological inhibition of gut-derived serotonin synthesis is a potential bone anabolic treatment for osteoporosis. Nature Med. 16, 308–312 (2010).
Acknowledgements
We thank T. Clemens, P. Ducy, M. Gershon, M. Kassem, S. Kousteni and members of the Karsenty laboratory for comments on the manuscript. We apologize to our colleagues whose work is not directly discussed and/or cited in this article because of space constraints. G.K. is supported by the National Institutes of Health, and M.F. by the Canadian Diabetes Association.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reprints and permissions information is available at www.nature.com/reprints.
Rights and permissions
About this article
Cite this article
Karsenty, G., Ferron, M. The contribution of bone to whole-organism physiology. Nature 481, 314–320 (2012). https://doi.org/10.1038/nature10763
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10763
This article is cited by
-
Osteocalcin of maternal and embryonic origins synergize to establish homeostasis in offspring
EMBO Reports (2024)
-
Neural EGFL-like 1, a craniosynostosis-related osteochondrogenic molecule, strikingly associates with neurodevelopmental pathologies
Cell & Bioscience (2023)
-
Glucose uptake and distribution across the human skeleton using state-of-the-art total-body PET/CT
Bone Research (2023)
-
Targeting strategies for bone diseases: signaling pathways and clinical studies
Signal Transduction and Targeted Therapy (2023)
-
Bone marrow mesenchymal stem cells’ osteogenic potential: superiority or non-superiority to other sources of mesenchymal stem cells?
Cell and Tissue Banking (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.