Abstract
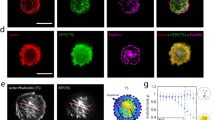
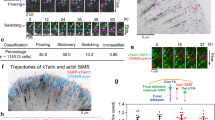
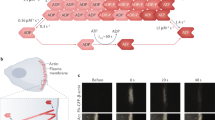
Mechanical stresses elicit cellular reactions mediated by chemical signals. Defective responses to forces underlie human medical disorders1,2,3,4 such as cardiac failure5 and pulmonary injury6. The actin cytoskeleton’s connectivity enables it to transmit forces rapidly over large distances7, implicating it in these physiological and pathological responses. Despite detailed knowledge of the cytoskeletal structure, the specific molecular switches that convert mechanical stimuli into chemical signals have remained elusive. Here we identify the actin-binding protein filamin A (FLNA)8,9 as a central mechanotransduction element of the cytoskeleton. We reconstituted a minimal system consisting of actin filaments, FLNA and two FLNA-binding partners: the cytoplasmic tail of β-integrin, and FilGAP. Integrins form an essential mechanical linkage between extracellular and intracellular environments, with β-integrin tails connecting to the actin cytoskeleton by binding directly to filamin4. FilGAP is an FLNA-binding GTPase-activating protein specific for RAC, which in vivo regulates cell spreading and bleb formation10. Using fluorescence loss after photoconversion, a novel, high-speed alternative to fluorescence recovery after photobleaching11, we demonstrate that both externally imposed bulk shear and myosin-II-driven forces differentially regulate the binding of these partners to FLNA. Consistent with structural predictions, strain increases β-integrin binding to FLNA, whereas it causes FilGAP to dissociate from FLNA, providing a direct and specific molecular basis for cellular mechanotransduction. These results identify a molecular mechanotransduction element within the actin cytoskeleton, revealing that mechanical strain of key proteins regulates the binding of signalling molecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ingber, D. E. Mechanobiology and diseases of mechanotransduction. Ann. Med. 35, 564–577 (2003)
Discher, D. E., Janmey, P. & Wang, Y. L. Tissue cells feel and respond to the stiffness of their substrate. Science 310, 1139–1143 (2005)
Moore, S. W., Roca-Cusachs, P. & Sheetz, M. P. Stretchy proteins on stretchy substrates: the important elements of integrin-mediated rigidity sensing. Dev. Cell 19, 194–206 (2010)
Hoffman, B. D. et al. Dynamic molecular processes mediate cellular mechanotransduction. Nature 475, 316–323 (2011)
Krüger, M. & Linke, W. A. Titin-based mechanical signalling in normal and failing myocardium. J. Mol. Cell. Cardiol. 46, 490–498 (2009)
Birukov, K. G. Small GTPases in mechanosensitive regulation of endothelial barrier. Microvasc. Res. 77, 46–52 (2009)
Wang, N., Tytell, J. D. & Ingber, D. E. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nature Rev. Mol. Cell Biol. 10, 75–82 (2009)
Stossel, T. P. et al. Filamins as integrators of cell mechanics and signalling. Nature Rev. Mol. Cell Biol. 2, 138–145 (2001)
Nakamura, F., Stossel, T. P. & Hartwig, J. H. The filamins: organizers of cell structure and function. Cell Adh. Migr. 5, 160–169 (2011)
Ohta, Y., Hartwig, J. H. & Stossel, T. P. FilGAP, a Rho- and ROCK-regulated GAP for Rac binds filamin A to control actin remodelling. Nature Cell Biol. 8, 803–814 (2006)
Sprague, B. L., Pego, R. L., Stavreva, D. A. & McNally, J. G. Analysis of binding reactions by fluorescence recovery after photobleaching. Biophys. J. 86, 3473–3495 (2004)
Nakamura, F., Osborn, T. M., Hartemink, C. A., Hartwig, J. H. & Stossel, T. P. Structural basis of filamin A functions. J. Cell Biol. 179, 1011–1025 (2007)
Wang, N., Butler, J. P. & Ingber, D. E. Mechanotransduction across the cell surface and through the cytoskeleton. Science 260, 1124–1127 (1993)
Calderwood, D. A. et al. Increased filamin binding to β-integrin cytoplasmic domains inhibits cell migration. Nature Cell Biol. 3, 1060–1068 (2001)
Lad, Y. et al. Structure of three tandem filamin domains reveals auto-inhibition of ligand binding. EMBO J. 26, 3993–4004 (2007)
Heikkinen, O. K. et al. Atomic structures of two novel immunoglobulin-like domain pairs in the actin cross-linking protein filamin. J. Biol. Chem. 284, 25450–25458 (2009)
Pentikäinen, U. & Ylanne, J. The regulation mechanism for the auto-inhibition of binding of human filamin A to integrin. J. Mol. Biol. 393, 644–657 (2009)
Chen, H. S., Kolahi, K. S. & Mofrad, M. R. Phosphorylation facilitates the integrin binding of filamin under force. Biophys. J. 97, 3095–3104 (2009)
Nakamura, F. et al. Molecular basis of filamin A-FilGAP interaction and its impairment in congenital disorders associated with filamin A mutations. PLoS ONE 4, e4928 (2009)
Koenderink, G. H. et al. An active biopolymer network controlled by molecular motors. Proc. Natl Acad. Sci. USA 106, 15192–15197 (2009)
Humphrey, D., Duggan, C., Saha, D., Smith, D. & Kas, J. Active fluidization of polymer networks through molecular motors. Nature 416, 413–416 (2002)
Smith, D. M. et al. Molecular motor-induced instabilities and crosslinkers determine biopolymer organization. Biophys. J. 93, 4445–4452 (2007)
Kanchanawong, P. et al. Nanoscale architecture of integrin-based cell adhesions. Nature 468, 580–584 (2010)
Grashoff, C. et al. Measuring mechanical tension across vinculin reveals regulation of focal adhesion dynamics. Nature 466, 263–266 (2010)
Na, S. et al. Rapid signal transduction in living cells is a unique feature of mechanotransduction. Proc. Natl Acad. Sci. USA 105, 6626–6631 (2008)
Kiema, T. et al. The molecular basis of filamin binding to integrins and competition with talin. Mol. Cell 21, 337–347 (2006)
Sanz-Moreno, V. et al. Rac activation and inactivation control plasticity of tumor cell movement. Cell 135, 510–523 (2008)
Shifrin, Y., Arora, P. D., Ohta, Y., Calderwood, D. A. & McCulloch, C. A. The role of FilGAP-filamin A interactions in mechanoprotection. Mol. Biol. Cell 20, 1269–1279 (2009)
Johnson, C. P., Tang, H. Y., Carag, C., Speicher, D. W. & Discher, D. E. Forced unfolding of proteins within cells. Science 317, 663–666 (2007)
Krieger, C. C. et al. Cysteine shotgun-mass spectrometry (CS-MS) reveals dynamic sequence of protein structure changes within mutant and stressed cells. Proc. Natl Acad. Sci. USA 108, 8269–8274 (2011)
Fernández-Suárez, M. & Ting, A. Y. Fluorescent probes for super-resolution imaging in living cells. Nature Rev. Mol. Cell Biol. 9, 929–943 (2008)
van der Flier, A. et al. Different splice variants of filamin-B affect myogenesis, subcellular distribution, and determine binding to integrin β subunits. J. Cell Biol. 156, 361–376 (2002)
Nakamura, F., Osborn, E., Janmey, P. A. & Stossel, T. P. Comparison of filamin A-induced cross-linking and Arp2/3 complex-mediated branching on the mechanics of actin filaments. J. Biol. Chem. 277, 9148–9154 (2002)
Weissmann, C. et al. Microtubule binding and trapping at the tip of neurites regulate tau motion in living neurons. Traffic 10, 1655–1668 (2009)
Acknowledgements
The authors acknowledge the Harvard Materials Research and Engineering Center (DMR-0820484) for confocal imaging, and M. Ginsberg, J. Lippincott-Schwartz and V. Verkhusha for providing complementary DNA for PA-GFP and PA-mCherry constructs. We thank T. Collins for technical assistance, L. Jawerth and V. Zaburdaev for discussions, and J. Wilking and K. Guenthner for help with the manuscript. This work was supported by grants NIH R01 HL19429 (T.P.S.) and NIH T32 HL07680 (A.J.E.) and by the Harvard University Science and Engineering Committee Seed Fund for Interdisciplinary Science (D.A.W., T.P.S., F.N.).
Author information
Authors and Affiliations
Contributions
The project was initiated by T.P.S., F.N. and D.A.W. Experiments were designed by A.J.E., T.P.S., J.H.H. and F.N. Proteins and materials were synthesized and purified by F.N. and A.J.E. FLAC experiments were performed by A.J.E. and binding assays were performed by F.N. Data was analysed by A.J.E. All authors discussed data and aided in preparing the manuscript. A.J.E. and F.N. contributed equally to this project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-9 with legends. (PDF 3491 kb)
Supplementary Movie 1
The movie shows PA-GFP β7 Integrin binds longer to del41 mutant FLNa than wild type FLNa. (MOV 6767 kb)
Supplementary Movie 2
The movie shows PA-GFP FilGAP binds longer to wild-type FLNa than to M2474E mutant FLNa. (MOV 9969 kb)
Supplementary Movie 3
The movie shows Myosin II deforms actin networks when ATP is available. (MOV 4196 kb)
Rights and permissions
About this article
Cite this article
Ehrlicher, A., Nakamura, F., Hartwig, J. et al. Mechanical strain in actin networks regulates FilGAP and integrin binding to filamin A. Nature 478, 260–263 (2011). https://doi.org/10.1038/nature10430
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10430
This article is cited by
-
Intracellular tension sensor reveals mechanical anisotropy of the actin cytoskeleton
Nature Communications (2023)
-
Filamin-A-interacting protein 1 (FILIP1) is a dual compartment protein linking myofibrils and microtubules during myogenic differentiation and upon mechanical stress
Cell and Tissue Research (2023)
-
Activation of autophagy triggers mitochondrial loss and changes acetylation profile relevant for mechanotransduction in bladder cancer cells
Archives of Toxicology (2023)
-
A statistical mechanics model for determining the length distribution of actin filaments under cellular tensional homeostasis
Scientific Reports (2022)
-
Optineurin links Hace1-dependent Rac ubiquitylation to integrin-mediated mechanotransduction to control bacterial invasion and cell division
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.