Abstract
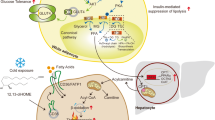
Adipose tissue mass is determined by the storage and removal of triglycerides in adipocytes1. Little is known, however, about adipose lipid turnover in humans in health and pathology. To study this in vivo, here we determined lipid age by measuring 14C derived from above ground nuclear bomb tests in adipocyte lipids. We report that during the average ten-year lifespan of human adipocytes, triglycerides are renewed six times. Lipid age is independent of adipocyte size, is very stable across a wide range of adult ages and does not differ between genders. Adipocyte lipid turnover, however, is strongly related to conditions with disturbed lipid metabolism. In obesity, triglyceride removal rate (lipolysis followed by oxidation) is decreased and the amount of triglycerides stored each year is increased. In contrast, both lipid removal and storage rates are decreased in non-obese patients diagnosed with the most common hereditary form of dyslipidaemia, familial combined hyperlipidaemia. Lipid removal rate is positively correlated with the capacity of adipocytes to break down triglycerides, as assessed through lipolysis, and is inversely related to insulin resistance. Our data support a mechanism in which adipocyte lipid storage and removal have different roles in health and pathology. High storage but low triglyceride removal promotes fat tissue accumulation and obesity. Reduction of both triglyceride storage and removal decreases lipid shunting through adipose tissue and thus promotes dyslipidaemia. We identify adipocyte lipid turnover as a novel target for prevention and treatment of metabolic disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Klein, R. A., Halliday, D. & Pittet, P. G. The use of 13-methyltetradecanoic acid as an indicator of adipose tissue turnover. Lipids 15, 572–579 (1980)
Mårin, P., Oden, B. & Björntorp, P. Assimilation and mobilization of triglycerides in subcutaneous abdominal and femoral adipose tissue in vivo in men: effects of androgens. J. Clin. Endocrinol. Metab. 80, 239–243 (1995)
Mårin, P., Rebuffe-Scrive, M. & Björntorp, P. Uptake of triglyceride fatty acids in adipose tissue in vivo in man. Eur. J. Clin. Invest. 20, 158–165 (1990)
Strawford, A., Antelo, F., Christiansen, M. & Hellerstein, M. K. Adipose tissue triglyceride turnover, de novo lipogenesis, and cell proliferation in humans measured with 2H2O. Am. J. Physiol. 286, E577–E588 (2004)
Nydal, R. & Lovseth, K. Distribution of radiocarbon from nuclear tests. Nature 206, 1029–1031 (1965)
Levin, I. & Kromer, B. The tropospheric 14CO2 level in mid latitudes of the northern hemisphere (1959–2003). Radiocarbon 46, 1261–1272 (2004)
Spalding, K. L., Bhardwaj, R. D., Buchholz, B. A., Druid, H. & Frisen, J. Retrospective birth dating of cells in humans. Cell 122, 133–143 (2005)
Perl, S. et al. Significant human β-cell turnover is limited to the first three decades of life as determined by in vivo thymidine analog incorporation and radiocarbon dating. J. Clin. Endocrinol. Metab. 95, E234–E239 (2010)
Spalding, K. L. et al. Dynamics of fat cell turnover in humans. Nature 453, 783–787 (2008)
Bergmann, O. et al. Evidence for cardiomyocyte renewal in humans. Science 324, 98–102 (2009)
Bhardwaj, R. D. et al. Neocortical neurogenesis in humans is restricted to development. Proc. Natl Acad. Sci. USA 103, 12564–12568 (2006)
Bernard, S., Frisen, J. & Spalding, K. L. A mathematical model for the interpretation of nuclear bomb test derived 14C incorporation in biological systems. Nucl. Instrum. Meth. B 268, 1295–1298 (2010)
Ekstedt, B. & Olivecrona, T. Uptake and release of fatty acids by rat adipose tissue: last in to first out? Lipids 5, 858–860 (1970)
Kerpel, S., Shafrir, E. & Shapiro, B. Mechanism of fatty acid assimilation in adipose tissue. Biochim. Biophys. Acta 46, 495–504 (1961)
Björntorp, P. Effects of age, sex, and clinical conditions on adipose tissue cellularity in man. Metabolism 23, 1091–1102 (1974)
Grundy, S. M. Chait, A & Brunzell, J. D. Familial combined hyperlipidemia workshop. Arterioscler. Thromb. Vasc. Biol. 7, 203–207 (1987)
Langin, D. et al. Adipocyte lipases and defect of lipolysis in human obesity. Diabetes 54, 3190–3197 (2005)
Reynisdottir, S., Eriksson, M., Angelin, B. & Arner, P. Impaired activation of adipocyte lipolysis in familial combined hyperlipidemia. J. Clin. Invest. 95, 2161–2169 (1995)
van der Kallen, C. J. et al. Evidence of insulin resistant lipid metabolism in adipose tissue in familial combined hyperlipidemia, but not type 2 diabetes mellitus. Atherosclerosis 164, 337–346 (2002)
Ayyobi, A. F. & Brunzell, J. D. Lipoprotein distribution in the metabolic syndrome, type 2 diabetes mellitus, and familial combined hyperlipidemia. Am. J. Cardiol. 92, 27–33 (2003)
Arner, P. Is familial combined hyperlipidaemia a genetic disorder of adipose tissue? Curr. Opin. Lipidol. 8, 89–94 (1997)
de Graaf, J., Veerkamp, M. J. & Stalenhoef, A. F. Metabolic pathogenesis of familial combined hyperlipidaemia with emphasis on insulin resistance, adipose tissue metabolism and free fatty acids. J. R. Soc. Med. 95 (suppl. 42). 46–53 (2002)
Blaak, E. E. et al. Fat oxidation before and after a high fat load in the obese insulin-resistant state. J. Clin. Endocrinol. Metab. 91, 1462–1469 (2006)
Houmard, J. A. Intramuscular lipid oxidation and obesity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 294, R1111–R1116 (2008)
Acknowledgements
This study was supported by the Swedish Research Council, Swedish Foundation for Strategic Research, Swedish Heart and Lung Foundation, Novo Nordic Foundation, Swedish Diabetes Foundation, Strategic Research Program in Diabetes at the Karolinska Institutet, Swedish Cancer Society, Uppsala BIO, Sweden, NIH/NCRR Grant RR13461, ERC grant 261258-HUFATREG and by the projects ‘Hepatic and adipose tissue and functions in the metabolic syndrome’ (HEPADIP, http://www.hepadip.org/) and ‘Adipokines as drug targets to combat adverse effects of excess adipose tissue’ (ADAPT, http://www.adapt-eu.net), which were supported by the European Commission as an Integrated Project under the 6th and the 7th Framework Programmes (contract LSHM-CT-2005-018734 and contract HEALTH-F2-2008-201100). This work was performed in part under the auspices of the US Department of Energy by Lawrence Livermore National Laboratory under contract DE-AC52-07NA27344. The authors would like to acknowledge E. Sjölin, K. Wåhlén, B.-M. Leijonhufvud, K. Hertel and Y. Widlund for technical assistance. We would like to thank F. Barnabé-Heider and J. Frisén for useful comments on the manuscript.
Author information
Authors and Affiliations
Contributions
K.L.S. and P.A. designed the study and wrote the manuscript together with K.N.F. and S.B. M.R. co-ordinated writing and data assembly. S.B. and E.A. were responsible for the modelling. K.L.S. performed sample preparation. M.S., G.P., B.A.B., P.S. and J.L. performed 14C accelerator mass spectrometry measurements. P.A., M.E., T.S. and H.H. collected clinical material.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information 1
The file contains Supplementary Text and Data, Supplementary Figures 1-3 with legends and additional references. (PDF 428 kb)
Supplementary Information 2
The file contains Supplementary Text, Supplementary References, Supplementary Figures 1-3 with legends and Supplementary Tables 1-2. (PDF 718 kb)
Rights and permissions
About this article
Cite this article
Arner, P., Bernard, S., Salehpour, M. et al. Dynamics of human adipose lipid turnover in health and metabolic disease. Nature 478, 110–113 (2011). https://doi.org/10.1038/nature10426
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10426
This article is cited by
-
White adipocyte dysfunction and obesity-associated pathologies in humans
Nature Reviews Molecular Cell Biology (2024)
-
Adipose Tissue Plasticity and Insulin Signaling in the Pathogenesis of Type 2 Diabetes
Diabetology International (2024)
-
Association between remnant cholesterol, metabolic syndrome, and cardiovascular disease: post hoc analysis of a prospective national cohort study
European Journal of Medical Research (2023)
-
Lipid remodeling of adipose tissue in metabolic health and disease
Experimental & Molecular Medicine (2023)
-
The long noncoding RNA ADIPINT regulates human adipocyte metabolism via pyruvate carboxylase
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.