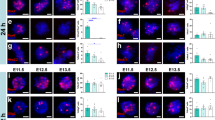
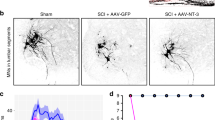
Abstract
Spinal cord injuries often occur at the cervical level above the phrenic motor pools, which innervate the diaphragm. The effects of impaired breathing are a leading cause of death from spinal cord injuries, underscoring the importance of developing strategies to restore respiratory activity. Here we show that, after cervical spinal cord injury, the expression of chondroitin sulphate proteoglycans (CSPGs) associated with the perineuronal net (PNN) is upregulated around the phrenic motor neurons. Digestion of these potently inhibitory extracellular matrix molecules with chondroitinase ABC (denoted ChABC) could, by itself, promote the plasticity of tracts that were spared and restore limited activity to the paralysed diaphragm. However, when combined with a peripheral nerve autograft, ChABC treatment resulted in lengthy regeneration of serotonin-containing axons and other bulbospinal fibres and remarkable recovery of diaphragmatic function. After recovery and initial transection of the graft bridge, there was an unusual, overall increase in tonic electromyographic activity of the diaphragm, suggesting that considerable remodelling of the spinal cord circuitry occurs after regeneration. This increase was followed by complete elimination of the restored activity, proving that regeneration is crucial for the return of function. Overall, these experiments present a way to markedly restore the function of a single muscle after debilitating trauma to the central nervous system, through both promoting the plasticity of spared tracts and regenerating essential pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Davies, S. J. et al. Regeneration of adult axons in white matter tracts of the central nervous system. Nature 390, 680–683 (1997)
Bradbury, E. J. et al. Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature 416, 636–640 (2002)
Silver, J. & Miller, J. H. Regeneration beyond the glial scar. Nature Rev. Neurosci. 5, 146–156 (2004)
Massey, J. M. et al. Chondroitinase ABC digestion of the perineuronal net promotes functional collateral sprouting in the cuneate nucleus after cervical spinal cord injury. J. Neurosci. 26, 4406–4414 (2006)
García-Alias, G., Barkhuysen, S., Buckle, M. & Fawcett, J. W. Chondroitinase ABC treatment opens a window of opportunity for task-specific rehabilitation. Nature Neurosci. 12, 1145–1151 (2009)
Hunanyan, A. S. et al. Role of chondroitin sulfate proteoglycans in axonal conduction in mammalian spinal cord. J. Neurosci. 30, 7761–7769 (2010)
Schachtrup, C. et al. Fibrinogen triggers astrocyte scar formation by promoting the availability of active TGF-β after vascular damage. J. Neurosci. 30, 5843–5854 (2010)
Choi, H. et al. Respiratory abnormalities resulting from midcervical spinal cord injury and their reversal by serotonin 1A agonists in conscious rats. J. Neurosci. 25, 4550–4559 (2005)
Fuller, D. D. et al. Cervical spinal cord injury upregulates ventral spinal 5-HT2A receptors. J. Neurotrauma 22, 203–213 (2005)
Golder, F. J. & Mitchell, G. S. Spinal synaptic enhancement with acute intermittent hypoxia improves respiratory function after chronic cervical spinal cord injury. J. Neurosci. 25, 2925–2932 (2005)
Goshgarian, H. G. The crossed phrenic phenomenon: a model for plasticity in the respiratory pathways following spinal cord injury. J. Appl. Physiol. 94, 795–810 (2003)
Hadley, S. D., Walker, P. D. & Goshgarian, H. G. Effects of the serotonin synthesis inhibitor p-CPA on the expression of the crossed phrenic phenomenon 4 h following C2 spinal cord hemisection. Exp. Neurol. 160, 479–488 (1999)
Hodges, M. R. et al. Defects in breathing and thermoregulation in mice with near-complete absence of central serotonin neurons. J. Neurosci. 28, 2495–2505 (2008)
David, S. & Aguayo, A. J. Axonal elongation into peripheral nervous system ‘bridges’ after central nervous system injury in adult rats. Science 214, 931–933 (1981)
Munz, M. et al. Functional activity of rat brainstem neurons regenerating axons along peripheral nerve grafts. Brain Res. 340, 115–125 (1985)
Gauthier, P. & Rasminsky, M. Activity of medullary respiratory neurons regenerating axons into peripheral nerve grafts in the adult rat. Brain Res. 438, 225–236 (1988)
Gauthier, P. et al. Functional reconnections established by central respiratory neurons regenerating axons into a nerve graft bridging the respiratory centers to the cervical spinal cord. J. Neurosci. Res. 70, 65–81 (2002)
Fouad, K. et al. Combining Schwann cell bridges and olfactory-ensheathing glia grafts with chondroitinase promotes locomotor recovery after complete transection of the spinal cord. J. Neurosci. 25, 1169–1178 (2005)
Houle, J. D. et al. Combining an autologous peripheral nervous system ‘bridge’ and matrix modification by chondroitinase allows robust, functional regeneration beyond a hemisection lesion of the adult rat spinal cord. J. Neurosci. 26, 7405–7415 (2006)
Yang, L. J. et al. Sialidase enhances spinal axon outgrowth in vivo . Proc. Natl Acad. Sci. USA 103, 11057–11062 (2006)
Zhou, L., Baumgartner, B. J., Hill-Felberg, S. J., McGowen, L. R. & Shine, H. D. Neurotrophin-3 expressed in situ induces axonal plasticity in the adult injured spinal cord. J. Neurosci. 23, 1424–1431 (2003)
Alto, L. T. et al. Chemotropic guidance facilitates axonal regeneration and synapse formation after spinal cord injury. Nature Neurosci. 12, 1106–1113 (2009)
Liu, K. et al. PTEN deletion enhances the regenerative ability of adult corticospinal neurons. Nature Neurosci. 13, 1075–1081 (2010)
McPhail, L. T., Plunet, W. T., Das, P. & Ramer, M. S. The astrocytic barrier to axonal regeneration at the dorsal root entry zone is induced by rhizotomy. Eur. J. Neurosci. 21, 267–270 (2005)
Hayashi, F., Hinrichsen, C. F. & McCrimmon, D. R. Short-term plasticity of descending synaptic input to phrenic motoneurons in rats. J. Appl. Physiol. 94, 1421–1430 (2003)
Lane, M. A. et al. Cervical prephrenic interneurons in the normal and lesioned spinal cord of the adult rat. J. Comp. Neurol. 511, 692–709 (2008)
Turrigiano, G. G. & Nelson, S. B. Homeostatic plasticity in the developing nervous system. Nature Rev. Neurosci. 5, 97–107 (2004)
Nantwi, K. D., El-Bohy, A. & Goshgarian, H. G. Actions of systemic theophylline on hemidiaphragmatic recovery in rats following cervical spinal cord hemisection. Exp. Neurol. 140, 53–59 (1996)
Fuller, D. D., Johnson, S. M., Olson, E. B., Jr & Mitchell, G. S. Synaptic pathways to phrenic motoneurons are enhanced by chronic intermittent hypoxia after cervical spinal cord injury. J. Neurosci. 23, 2993–3000 (2003)
Teng, Y. D. et al. Serotonin 1A receptor agonists reverse respiratory abnormalities in spinal cord-injured rats. J. Neurosci. 23, 4182–4189 (2003)
Alilain, W. J. et al. Light-induced rescue of breathing after spinal cord injury. J. Neurosci. 28, 11862–11870 (2008)
Houle, J. D. et al. Combining peripheral nerve grafting and matrix modulation to repair the injured rat spinal cord. J. Vis. Exp. 10.3791/1324 (2009)
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Coordinates (Academic, 1998)
Acknowledgements
This project was funded by The Christopher and Dana Reeve Foundation (W.J.A.), the International Spinal Research Trust (W.J.A.), the National Institute of Neurological Disorders and Stroke, grants NS25713 and NS060767 (J.S.), and the National Heart, Lung and Blood Institute, grant HL080318 (T.E.D.). We offer special thanks to the Brumagin Memorial Fund (J.S.) and the Ellen Becker Neuroscience Regenerative Medicine Research Fund (J.S.). We also thank J. D. Houle and V. J. Tom for teaching us the peripheral nerve grafting technique.
Author information
Authors and Affiliations
Contributions
W.J.A. performed the surgeries, recorded and analysed the electrophysiological data, and completed the immunocytochemical, tracing and histological experiments. K.P.H. helped with the immunocytochemical detection and quantification. H.H. assisted with the animal care, surgeries and data quantification, as well as performed all of the tissue processing. T.E.D. gave technical guidance and helped discuss the results. W.J.A. and J.S. designed all of the studies, analysed the data and wrote the paper. All authors discussed the data and helped to prepare the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-10 with legends. (PDF 1287 kb)
Rights and permissions
About this article
Cite this article
Alilain, W., Horn, K., Hu, H. et al. Functional regeneration of respiratory pathways after spinal cord injury. Nature 475, 196–200 (2011). https://doi.org/10.1038/nature10199
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10199
This article is cited by
-
Osteopontin enhances the effect of treadmill training and promotes functional recovery after spinal cord injury
Molecular Biomedicine (2023)
-
Regulation of axonal regeneration after mammalian spinal cord injury
Nature Reviews Molecular Cell Biology (2023)
-
Chondroitin sulfate proteoglycans prevent immune cell phenotypic conversion and inflammation resolution via TLR4 in rodent models of spinal cord injury
Nature Communications (2022)
-
Natural and targeted circuit reorganization after spinal cord injury
Nature Neuroscience (2022)
-
New insights into glial scar formation after spinal cord injury
Cell and Tissue Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.