Abstract
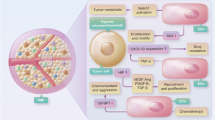
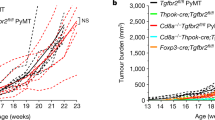
Although immune mechanisms can suppress tumour growth1,2, tumours establish potent, overlapping mechanisms that mediate immune evasion3,4,5,6. Emerging evidence suggests a link between angiogenesis and the tolerance of tumours to immune mechanisms7,8,9,10. Hypoxia, a condition that is known to drive angiogenesis in tumours, results in the release of damage-associated pattern molecules, which can trigger the rejection of tumours by the immune system11. Thus, the counter-activation of tolerance mechanisms at the site of tumour hypoxia would be a crucial condition for maintaining the immunological escape of tumours. However, a direct link between tumour hypoxia and tolerance through the recruitment of regulatory cells has not been established. We proposed that tumour hypoxia induces the expression of chemotactic factors that promote tolerance. Here we show that tumour hypoxia promotes the recruitment of regulatory T (Treg) cells through induction of expression of the chemokine CC-chemokine ligand 28 (CCL28), which, in turn, promotes tumour tolerance and angiogenesis. Thus, peripheral immune tolerance and angiogenesis programs are closely connected and cooperate to sustain tumour growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Koebel, C. M. et al. Adaptive immunity maintains occult cancer in an equilibrium state. Nature 450, 903–907 (2007)
Zhang, L. et al. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N. Engl. J. Med. 348, 203–213 (2003)
Zou, W. Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nature Rev. Cancer 5, 263–274 (2005)
Curiel, T. J. et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nature Med. 10, 942–949 (2004)
Nagaraj, S. & Gabrilovich, D. I. Myeloid-derived suppressor cells. Adv. Exp. Med. Biol. 601, 213–223 (2007)
Marigo, I., Dolcetti, L., Serafini, P., Zanovello, P. & Bronte, V. Tumor-induced tolerance and immune suppression by myeloid derived suppressor cells. Immunol. Rev. 222, 162–179 (2008)
Buckanovich, R. J. et al. Endothelin B receptor mediates the endothelial barrier to T cell homing to tumors and disables immune therapy. Nature Med. 14, 28–36 (2008)
Gabrilovich, D. I. et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nature Med. 2, 1096–1103 (1996)
Li, B. et al. Vascular endothelial growth factor blockade reduces intratumoral regulatory T cells and enhances the efficacy of a GM-CSF-secreting cancer immunotherapy. Clin. Cancer Res. 12, 6808–6816 (2006)
Osada, T. et al. The effect of anti-VEGF therapy on immature myeloid cell and dendritic cells in cancer patients. Cancer Immunol. Immunother. 57, 1115–1124 (2008)
Lotfi, R., Lee, J. J. & Lotze, M. T. Eosinophilic granulocytes and damage-associated molecular pattern molecules (DAMPs): role in the inflammatory response within tumors. J. Immunother. 30, 16–28 (2007)
Shimogai, R. et al. Expression of hypoxia-inducible factor 1α gene affects the outcome in patients with ovarian cancer. Int. J. Gynecol. Cancer 18, 499–505 (2008)
Daponte, A. et al. Prognostic significance of hypoxia-inducible factor 1α (HIF-1α) expression in serous ovarian cancer: an immunohistochemical study. BMC Cancer 8, 335 (2008)
Pan, J. et al. A novel chemokine ligand for CCR10 and CCR3 expressed by epithelial cells in mucosal tissues. J. Immunol. 165, 2943–2949 (2000)
Feng, N. et al. Redundant role of chemokines CCL25/TECK and CCL28/MEC in IgA+ plasmablast recruitment to the intestinal lamina propria after rotavirus infection. J. Immunol. 176, 5749–5759 (2006)
Eksteen, B. et al. Epithelial inflammation is associated with CCL28 production and the recruitment of regulatory T cells expressing CCR10. J. Immunol. 177, 593–603 (2006)
Wang, W. et al. Identification of a novel chemokine (CCL28), which binds CCR10 (GPR2). J. Biol. Chem. 275, 22313–22323 (2000)
Lazarus, N. H. et al. A common mucosal chemokine (mucosae-associated epithelial chemokine/CCL28) selectively attracts IgA plasmablasts. J. Immunol. 170, 3799–3805 (2003)
Roby, K. F. et al. Development of a syngeneic mouse model for events related to ovarian cancer. Carcinogenesis 21, 585–591 (2000)
Zhang, L. et al. Generation of a syngeneic mouse model to study the effects of vascular endothelial growth factor in ovarian carcinoma. Am. J. Pathol. 161, 2295–2309 (2002)
Elia, L. et al. CD4+CD25+ regulatory T-cell-inactivation in combination with adenovirus vaccines enhances T-cell responses and protects mice from tumor challenge. Cancer Gene Ther. 14, 201–210 (2007)
Kandalaft, L. E., Facciabene, A., Buckanovich, R. J. & Coukos, G. Endothelin B receptor, a new target in cancer immune therapy. Clin. Cancer Res. 15, 4521–4528 (2009)
Buckanovich, R. J. et al. Tumor vascular proteins as biomarkers in ovarian cancer. J. Clin. Oncol. 25, 852–861 (2007)
Ohta, A. et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc. Natl Acad. Sci. USA 103, 13132–13137 (2006)
Ben-Shoshan, J., Maysel-Auslender, S., Mor, A., Keren, G. & George, J. Hypoxia controls CD4+CD25+ regulatory T-cell homeostasis via hypoxia-inducible factor-1α. Eur. J. Immunol. 38, 2412–2418 (2008)
Curiel, T. J. et al. Blockade of B7-H1 improves myeloid dendritic cell-mediated antitumor immunity. Nature Med. 9, 562–567 (2003)
Yang, L. et al. Expansion of myeloid immune suppressor Gr+CD11b+ cells in tumor-bearing host directly promotes tumor angiogenesis. Cancer Cell 6, 409–421 (2004)
Shojaei, F. et al. Tumor refractoriness to anti-VEGF treatment is mediated by CD11b+Gr1+ myeloid cells. Nature Biotechnol. 25, 911–920 (2007)
Curiel, T. J. et al. Dendritic cell subsets differentially regulate angiogenesis in human ovarian cancer. Cancer Res. 64, 5535–5538 (2004)
Bertozzi, C. C. et al. Multiple initial culture conditions enhance the establishment of cell lines from primary ovarian cancer specimens. In Vitro Cell. Dev. Biol. Anim. 42, 58–62 (2006)
Bild, A. H. et al. Oncogenic pathway signatures in human cancers as a guide to targeted therapies. Nature 439, 353–357 (2006)
Tothill, R. W. et al. Novel molecular subtypes of serous and endometrioid ovarian cancer linked to clinical outcome. Clin. Cancer Res. 14, 5198–5208 (2008)
Camp, R. L., Dolled-Filhart, M. & Rimm, D. L. X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin. Cancer Res. 10, 7252–7259 (2004)
Bergeron, M. et al. Detection of hypoxic cells with the 2-nitroimidazole, EF5, correlates with early redox changes in rat brain after perinatal hypoxia–ischemia. Neuroscience 89, 1357–1366 (1999)
Lee, O. H. et al. Sphingosine 1-phosphate induces angiogenesis: its angiogenic action and signaling mechanism in human umbilical vein endothelial cells. Biochem. Biophys. Res. Commun. 264, 743–750 (1999)
Lee, Y. H. et al. Cell-retained isoforms of vascular endothelial growth factor (VEGF) are correlated with poor prognosis in osteosarcoma. Eur. J. Cancer 35, 1089–1093 (1999)
Irizarry, R. A. et al. Summaries of Affymetrix GeneChip probe level data. Nucleic Acids Res. 31, e15 (2003)
Acknowledgements
This work was supported by National Institutes of Health grant R01-CA116779; National Cancer Institute Ovarian SPORE grant P01-CA83638; and the Ovarian Cancer Research Fund. We thank M. Celeste Simon and S. Evans for generous help with the hypoxia studies; M. Feldman and the University of Pennsylvania Tumor Tissue & Biospecimen Bank for tumour processing; and G. Danet-Desnoyers and the University of Pennsylvania Xenograft Core Facility for NSG mice.
Author information
Authors and Affiliations
Contributions
A.F. designed many of the experiments and conducted most of them, analysed the data and drafted the manuscript. X.P. assisted with the experiments. I.S.H. analysed and interpreted the tissue-based studies. A.B. conducted the initial hypoxia and qPCR array experiments. K.B. assisted with the experiments. L.-P.W. carried out the tissue stains. P.A.G. analysed the Affymetrix data. C.B.G. assisted with the tissue-based studies. P.L. prepared the tissue microarrays. L.Z. assisted with the study design and provided many cell lines. G.C. conceived and supervised the study, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Tables 1-2 and Supplementary Figures 1-14 with legends. (PDF 2240 kb)
Rights and permissions
About this article
Cite this article
Facciabene, A., Peng, X., Hagemann, I. et al. Tumour hypoxia promotes tolerance and angiogenesis via CCL28 and Treg cells. Nature 475, 226–230 (2011). https://doi.org/10.1038/nature10169
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10169
This article is cited by
-
Multi-omic analyses of m5C readers reveal their characteristics and immunotherapeutic proficiency
Scientific Reports (2024)
-
Expression analysis of lymphocyte subsets and lymphocyte-to-monocyte ratio: reveling immunosuppression and chronic inflammation in breast cancer
Journal of Cancer Research and Clinical Oncology (2024)
-
The role of HIF in angiogenesis, lymphangiogenesis, and tumor microenvironment in urological cancers
Molecular Biology Reports (2024)
-
Evaluation of regulatory T-cells in cancer immunotherapy: therapeutic relevance of immune checkpoint inhibition
Medical Oncology (2024)
-
Epigenetic reprogramming of T cells: unlocking new avenues for cancer immunotherapy
Cancer and Metastasis Reviews (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.