Abstract
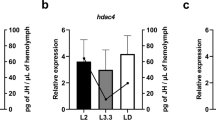
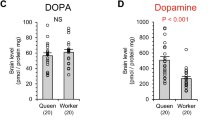
The honeybee (Apis mellifera) forms two female castes: the queen and the worker. This dimorphism depends not on genetic differences, but on ingestion of royal jelly, although the mechanism through which royal jelly regulates caste differentiation has long remained unknown. Here I show that a 57-kDa protein in royal jelly, previously designated as royalactin, induces the differentiation of honeybee larvae into queens. Royalactin increased body size and ovary development and shortened developmental time in honeybees. Surprisingly, it also showed similar effects in the fruitfly (Drosophila melanogaster). Mechanistic studies revealed that royalactin activated p70 S6 kinase, which was responsible for the increase of body size, increased the activity of mitogen-activated protein kinase, which was involved in the decreased developmental time, and increased the titre of juvenile hormone, an essential hormone for ovary development. Knockdown of epidermal growth factor receptor (Egfr) expression in the fat body of honeybees and fruitflies resulted in a defect of all phenotypes induced by royalactin, showing that Egfr mediates these actions. These findings indicate that a specific factor in royal jelly, royalactin, drives queen development through an Egfr-mediated signalling pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
25 May 2011
The legend to Fig. 1 was corrected
References
Maynard Smith, J. & Szathmary, L. The Major Transitions in Evolution (Freeman, 1995).
Haydak, M. H. Honey bee nutrition. Annu. Rev. Entomol. 15, 143–156 (1970)
Patel, N. G., Haydak, M. H. & Gochnauer, T. A. Electrophoretic components of the proteins in honeybee larval food. Nature 186, 633–634 (1960)
Weaver, N. Effects of larval age on dimorphic differentiation of the female honey bee. Ann. Entomol. Soc. Am. 50, 283–294 (1957)
Shuel, R. W. & Dixon, S. E. The early establishment of dimorphism in the female honeybee, Apis mellifera L. Insectes Soc. 7, 265–282 (1960)
Page, R. E. & Peng, C. Y. Aging and development in social insects with emphasis on the honey bee, Apis mellifera L. Exp. Gerontol. 36, 695–711 (2001)
Wheeler, D. E. Developmental and physiological determinations of caste in social Hymenoptera: evolutionary implications. Am. Nat. 128, 13–34 (1986)
Patel, A. et al. The making of a queen: TOR pathway is a key player in diphenic caste development. PLoS ONE 2, e509 (2007)
Kamakura, M., Mitani, N., Fukuda, T. & Fukushima, M. Antifatigue effect of fresh royal jelly in mice. J. Nutr. Sci. Vitaminol. (Tokyo) 47, 394–401 (2001)
Beetsma, J. The process of queen-worker differentiation in the honeybee. Bee World 60, 24–39 (1979)
Kamakura, M., Suenobu, N. & Fukushima, M. 57-kDa protein in royal jelly enhances proliferation of primary cultured rat hepatocytes and increases albumin production in the absence of serum. Biochem. Biophys. Res. Commun. 282, 865–874 (2001)
Kamakura, M. Signal transduction mechanism leading to enhanced proliferation of primary cultured adult rat hepatocytes treated with royal jelly 57-kDa protein. J. Biochem. 132, 911–919 (2002)
Bloch, G., Wheeler, D. E. & Robinson, G. E. in Hormones, Brain and Behavior Vol. 3 (eds Pfaff, D. W., Arnold, A. P., Fahrbach, S. E., Etgen, A. M. & Rubin, R. T. ) 195–235 (Academic Press, 2002)
Wirtz, P. & Beetsma, J. Induction of caste differentiation in the honeybee (Apis mellifera L.) by juvenile hormone. Entomol. Exp. Appl. 15, 517–520 (1972)
Tatar, M. et al. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 292, 107–110 (2001)
Colombani, J. et al. Antagonistic actions of ecdysone and insulins determine final size in Drosophila . Science 310, 667–670 (2005)
Gilboa, L. & Lehmann, R. Soma-germline interactions coordinate homeostasis and growth in the Drosophila gonad. Nature 443, 97–100 (2006)
Caldwell, P. E., Walkiewicz, M. & Stern, M. Ras activity in the Drosophila prothoracic gland regulates body size and developmental rate via ecdysone release. Curr. Biol. 15, 1785–1795 (2005)
Mirth, C., Truman, J. W. & Riddiford, L. M. The role of the prothoracic gland in determining critical weight for metamorphosis in Drosophila melanogaster . Curr. Biol. 15, 1796–1807 (2005)
Belgacem, Y. H. & Martin, J. R. Hmgcr in the Corpus Allatum controls sexual dimorphism of locomotor activity and body size via the insulin pathway in Drosophila . PLoS ONE 2, e187 (2007)
Colombani, J. et al. A nutrient sensor mechanism controls Drosophila growth. Cell 114, 739–749 (2003)
Zinke, I. et al. Suppression of food intake and growth by amino acids in Drosophila: the role of pumpless, a fat body expressed gene with homology to vertebrate glycine cleavage system. Development 126, 5275–5284 (1999)
Navolanic, P. M., Steelman, L. S. & McCubrey, J. A. EGFR family signaling and its association with breast cancer development and resistance to chemotherapy. Int. J. Oncol. 22, 237–252 (2003)
LeVea, C. M., Reeder, J. E. & Mooney, R. A. EGF-dependent cell cycle progression is controlled by density-dependent regulation of Akt activation. Exp. Cell Res. 297, 272–284 (2004)
Rintelen, F., Stocker, H., Thomas, G. & Hafen, E. PDK1 regulates growth through Akt and S6K in Drosophila . Proc. Natl Acad. Sci. USA 98, 15020–15025 (2001)
McManus, E. J. & Alessi, D. R. TSC1–TSC2: a complex tale of PKB-mediated S6K regulation. Nature Cell Biol. 4, E214–E216 (2002)
Lee, K. S. et al. Drosophila short neuropeptide F signalling regulates growth by ERK-mediated insulin signalling. Nature Cell Biol. 10, 468–475 (2008)
Montagne, J. et al. Drosophila S6 kinase: a regulator of cell size. Science 285, 2126–2129 (1999)
Zhang, H. et al. Regulation of cellular growth by the Drosophila target of rapamycin dTOR . Genes Dev. 14, 2712–2724 (2000)
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993)
Urban, S. Rhomboid proteins: conserved membrane proteases with divergent biological functions. Genes Dev. 20, 3054–3068 (2006)
Schmitzova, J. et al. A family of royal jelly proteins of the honeybee. Apis mellifera L. Cell. Mol. Life Sci. 54, 1020–1030 (1998)
Bownes, M. The regulation of the yolk protein genes, a family of sex differentiation genes in Drosophila melanogaster . Bioessays 16, 745–752 (1994)
Goewie, E. A. & Beetsma, J. Induction of caste differentiation in the honey bee (Apis mellifera L.) after topical application of JH-III. Proc. K. Ned. Akad. Wet. C 79, 466–469 (1976)
Ebert, R. Influence of juvenile hormone on gravity orientation in the female honeybee larvae (Apis mellifera L.). J. Comp. Physiol. 137, 7–16 (1980)
Bíliková, K. et al. Apisimin, a new serine-valine-rich peptide from honeybee (Apis mellifera L.) royal jelly: purification and molecular characterization. FEBS Lett. 528, 125–129 (2002)
Kamakura, M. & Sakaki, T. A hypopharyngeal gland protein of the worker honeybee Apis mellifera L. enhances proliferation of primary-cultured rat hepatocytes and suppresses apoptosis in the absence of serum. Protein Expr. Purif. 45, 307–314 (2006)
Acknowledgements
I thank D. Yamamoto for provision of general fruitfly treatment methods and helpful advice; and S. Hayashi and T. Adachi-Yamada for instruction of dissection techniques in Drosophila. I thank T. Nonogaki and Y. Hasada for supply of honeybee larvae; K. Yu, M. Tatar, P. Leopold, G. Korge, Y. T. Ip, T. G. Wilson and D. Yamamoto for fly stocks. We are grateful to T. Oda for the gift of royal jelly, and to W. R. S. Steele for proofreading the article.
Author information
Authors and Affiliations
Contributions
M.K. designed the research and performed the experiments. M.K. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Results, a Supplementary Discussion, Supplementary Methods, additional references, Supplementary Figures 1-34 with legends and Supplementary Tables 1-14. This file was replaced online on 15 October 2012 to correct Supplementary Figures 22a and 33c and again on 23 January 2013 to correct Supplementary Figure 19b.. (PDF 4372 kb)
Rights and permissions
About this article
Cite this article
Kamakura, M. Royalactin induces queen differentiation in honeybees. Nature 473, 478–483 (2011). https://doi.org/10.1038/nature10093
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10093
This article is cited by
-
Reproductives signature revealed by protein profiling and behavioral bioassays in termite
Scientific Reports (2023)
-
Effects of simulated tropical heat waves during development on the morphological and reproductive traits of Africanized honey bee
Insectes Sociaux (2023)
-
Influence of Probiotics Feed Supplementation on Hypopharyngeal Glands Morphometric Measurements of Honeybee Workers Apis mellifera L.
Probiotics and Antimicrobial Proteins (2023)
-
DNA methylation and expression of the egfr gene are associated with worker size in monomorphic ants
Scientific Reports (2022)
-
Glucosamine-6-phosphate N-acetyltransferase gene silencing by parental RNA interference in rice leaf folder, Cnaphalocrocis medinalis (Lepidoptera: Pyralidae)
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.