Abstract
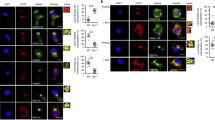
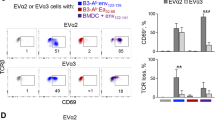
After an infection, cytotoxic T lymphocyte precursors proliferate and become effector cells by recognizing foreign peptides in the groove of major histocompatibility complex (MHC) class I molecules expressed by antigen-presenting cells (APCs)1. Professional APCs specialized for T-cell activation acquire viral antigen either by becoming infected themselves (direct presentation) or by phagocytosis of infected cells, followed by transfer of antigen to the cytosol, processing and MHC class I loading in a process referred to as cross-presentation2. An alternative way, referred to as ‘cross-dressing’, by which an uninfected APC could present antigen was postulated to be by the transfer of preformed peptide–MHC complexes from the surface of an infected cell to the APC without the need of further processing3. Here we show that this mechanism exists and boosts the antiviral response of mouse memory CD8+ T cells. A number of publications have demonstrated sharing of peptide-loaded MHC molecules in vitro4,5,6,7. Our in vitro experiments demonstrate that cross-dressing APCs do not acquire peptide–MHC complexes in the form of exosomes released by donor cells. Rather, the APCs and donor cells have to contact each other for the transfer to occur. After a viral infection, we could isolate cross-dressed APCs able to present viral antigen in vitro. Furthermore, using the diphtheria toxin system to selectively eliminate APCs that could only acquire viral peptide–MHC complexes by cross-dressing, we show that such presentation can promote the expansion of resting memory T cells. Notably, naive T cells were excluded from taking part in the response. Cross-dressing is a mechanism of antigen presentation used by dendritic cells that may have a significant role in activating previously primed CD8+ T cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vyas, J. M., Van der Veen, A. G. & Ploegh, H. L. The known unknowns of antigen processing and presentation. Nature Rev. Immunol. 8, 607–618 (2008)
Lin, M. L., Zhan, Y., Villadangos, J. A. & Lew, A. M. The cell biology of cross-presentation and the role of dendritic cell subsets. Immunol. Cell Biol. 86, 353–362 (2008)
Yewdell, J. W. & Haeryfar, S. M. Understanding presentation of viral antigens to CD8+ T cells in vivo: the key to rational vaccine design. Annu. Rev. Immunol. 23, 651–682 (2005)
Dolan, B. P., Gibbs, K. D., Jr & Ostrand-Rosenberg, S. Dendritic cells cross-dressed with peptide MHC class I complexes prime CD8+ T cells. J. Immunol. 177, 6018–6024 (2006)
Dolan, B. P., Gibbs, K. D., Jr & Ostrand-Rosenberg, S. Tumor-specific CD4+ T cells are activated by “cross-dressed” dendritic cells presenting peptide-MHC class II complexes acquired from cell-based cancer vaccines. J. Immunol. 176, 1447–1455 (2006)
Qu, C., Nguyen, V. A., Merad, M. & Randolph, G. J. MHC class I/peptide transfer between dendritic cells overcomes poor cross-presentation by monocyte-derived APCs that engulf dying cells. J. Immunol. 182, 3650–3659 (2009)
Smyth, L. A. et al. The relative efficiency of acquisition of MHC:peptide complexes and cross-presentation depends on dendritic cell type. J. Immunol. 181, 3212–3220 (2008)
Théry, C. et al. Indirect activation of naive CD4+ T cells by dendritic cell-derived exosomes. Nature Immunol. 3, 1156–1162 (2002)
Théry, C., Ostrowski, M. & Segura, E. Membrane vesicles as conveyors of immune responses. Nature Rev. Immunol. 9, 581–593 (2009)
Joly, E. & Hudrisier, D. What is trogocytosis and what is its purpose? Nature Immunol. 4, 815 (2003)
Davis, D. M. Intercellular transfer of cell-surface proteins is common and can affect many stages of an immune response. Nature Rev. Immunol. 7, 238–243 (2007)
Harshyne, L. A., Watkins, S. C., Gambotto, A. & Barratt-Boyes, S. M. Dendritic cells acquire antigens from live cells for cross-presentation to CTL. J. Immunol. 166, 3717–3723 (2001)
Matheoud, D. et al. Cross-presentation by dendritic cells from live cells induces protective immune responses in vivo. Blood 115, 4412–4420 (2010)
Wakim, L. M., Woodward-Davis, A. & Bevan, M. J. Memory T cells persisting within the brain after local infection show functional adaptations to their tissue of residence. Proc. Natl Acad. Sci. USA 107, 17872–17879 (2010)
Turner, M. J., Jellison, E. R., Lingenheld, E. G., Puddington, L. & Lefrancois, L. Avidity maturation of memory CD8 T cells is limited by self-antigen expression. J. Exp. Med. 205, 1859–1868 (2008)
Sun, J. C., Williams, M. A. & Bevan, M. J. CD4+ T cells are required for the maintenance, not programming, of memory CD8+ T cells after acute infection. Nature Immunol. 5, 927–933 (2004)
Thery, C., Amigorena, S., Raposo, G. & Clayton, A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr. Protoc. Cell Biol. 3, Unit 3.22. (2006)
Sanderson, S. & Shastri, N. LacZ inducible, antigen/MHC-specific T cell hybrids. Int. Immunol. 6, 369–376 (1994)
Acknowledgements
This work was supported by the Howard Hughes Medical Institute and National Institutes of Health. L.M.W is supported by an Overseas Biomedical Fellowship from the National Health and Medical Research Council of Australia.
Author information
Authors and Affiliations
Contributions
L.M.W. and M.J.B. devised experiments and wrote the paper. L.M.W. performed the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-10 with legend, Supplementary Methods and additional references. (PDF 673 kb)
Supplementary Movie 1
The movie shows membrane exchange between cultured DCs. DCs were labeled with either CMFDA (cytoplasmic dye - green) or Dil (membrane dye - red) and were culture together for 4 hours. Images were acquired every 4 mins. (MOV 5459 kb)
Rights and permissions
About this article
Cite this article
Wakim, L., Bevan, M. Cross-dressed dendritic cells drive memory CD8+ T-cell activation after viral infection. Nature 471, 629–632 (2011). https://doi.org/10.1038/nature09863
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09863
This article is cited by
-
Prior infection with unrelated neurotropic virus exacerbates influenza disease and impairs lung T cell responses
Nature Communications (2024)
-
Biomimetic nanovaccine-mediated multivalent IL-15 self-transpresentation (MIST) for potent and safe cancer immunotherapy
Nature Communications (2023)
-
Engineering of Trophoblast Extracellular Vesicle-Delivering Hydrogels for Localized Tolerance Induction in Cell Transplantation
Cellular and Molecular Bioengineering (2023)
-
The roles of small extracellular vesicles in cancer and immune regulation and translational potential in cancer therapy
Journal of Experimental & Clinical Cancer Research (2022)
-
Dexosomes as a cell-free vaccine for cancer immunotherapy
Journal of Experimental & Clinical Cancer Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.