Abstract
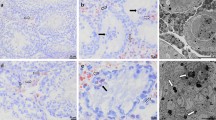
Apoptosis and the subsequent clearance of dying cells occurs throughout development and adult life in many tissues. Failure to promptly clear apoptotic cells has been linked to many diseases1,2,3. ELMO1 is an evolutionarily conserved cytoplasmic engulfment protein that functions downstream of the phosphatidylserine receptor BAI1, and, along with DOCK1 and the GTPase RAC1, promotes internalization of the dying cells4,5,6,7. Here we report the generation of ELMO1-deficient mice, which we found to be unexpectedly viable and grossly normal. However, they had a striking testicular pathology, with disrupted seminiferous epithelium, multinucleated giant cells, uncleared apoptotic germ cells and decreased sperm output. Subsequent in vitro and in vivo analyses revealed a crucial role for ELMO1 in the phagocytic clearance of apoptotic germ cells by Sertoli cells lining the seminiferous epithelium. The engulfment receptor BAI1 and RAC1 (upstream and downstream of ELMO1, respectively) were also important for Sertoli-cell-mediated engulfment. Collectively, these findings uncover a selective requirement for ELMO1 in Sertoli-cell-mediated removal of apoptotic germ cells and make a compelling case for a relationship between engulfment and tissue homeostasis in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Henson, P. M. Dampening inflammation. Nature Immunol. 6, 1179–1181 (2005)
Nagata, S., Hanayama, R. & Kawane, K. Autoimmunity and the clearance of dead cells. Cell 140, 619–630 (2010)
Gregory, C. D. & Pound, J. D. in Phagocytosis of Dying Cells Vol. 1. (eds Krysko, D. V. & Vandenabeele, P.) 271–298 (Springer, 2009)
Zhou, Z., Caron, E., Hartwieg, E., Hall, A. & Horvitz, H. R. The C. elegans PH domain protein CED-12 regulates cytoskeletal reorganization via a Rho/Rac GTPase signaling pathway. Dev. Cell 1, 477–489 (2001)
Wu, Y. C., Tsai, M. C., Cheng, L. C., Chou, C. J. & Weng, N. Y. C. elegans CED-12 acts in the conserved crkII/DOCK180/Rac pathway to control cell migration and cell corpse engulfment. Dev. Cell 1, 491–502 (2001)
Gumienny, T. L. et al. CED-12/ELMO, a novel member of the CrkII/Dock180/Rac pathway, is required for phagocytosis and cell migration. Cell 107, 27–41 (2001)
Park, D. et al. BAI1 is an engulfment receptor for apoptotic cells upstream of the ELMO/Dock180/Rac module. Nature 450, 430–434 (2007)
Lauber, K., Blumenthal, S. G., Waibel, M. & Wesselborg, S. Clearance of apoptotic cells: getting rid of the corpses. Mol. Cell 14, 277–287 (2004)
Zhou, Z., Mangahas, P. M. & Yu, X. The genetics of hiding the corpse: engulfment and degradation of apoptotic cells in C. elegans and D. melanogaster. Curr. Top. Dev. Biol. 63, 91–143 (2004)
deBakker, C. D. et al. Phagocytosis of apoptotic cells is regulated by a UNC-73/TRIO-MIG-2/RhoG signaling module and armadillo repeats of CED-12/ELMO. Curr. Biol. 14, 2208–2216 (2004)
Cool, J. & Capel, B. Mixed signals: development of the testis. Semin. Reprod. Med. 27, 5–13 (2009)
Loveland, K. L. et al. Drivers of germ cell maturation. Ann. NY Acad. Sci. 1061, 173–182 (2005)
Griswold, M. D. The central role of Sertoli cells in spermatogenesis. Semin. Cell Dev. Biol. 9, 411–416 (1998)
Bailey, R. W., Aronow, B., Harmony, J. A. & Griswold, M. D. Heat shock-initiated apoptosis is accelerated and removal of damaged cells is delayed in the testis of clusterin/ApoJ knock-out mice. Biol. Reprod. 66, 1042–1053 (2002)
Braun, R. E. Every sperm is sacred–or is it? Nature Genet. 18, 202–204 (1998)
Rotter, V. et al. Mice with reduced levels of p53 protein exhibit the testicular giant-cell degenerative syndrome. Proc. Natl Acad. Sci. USA 90, 9075–9079 (1993)
Holstein, A. F. & Eckmann, C. Multinucleated spermatocytes and spermatids in human seminiferous tubules. Andrologia 18, 5–16 (1986)
Lysiak, J. J., Zheng, S., Woodson, R. & Turner, T. T. Caspase-9-dependent pathway to murine germ cell apoptosis: mediation by oxidative stress, BAX, and caspase 2. Cell Tissue Res. 328, 411–419 (2007)
Lysiak, J. J., Turner, S. D. & Turner, T. T. Molecular pathway of germ cell apoptosis following ischemia/reperfusion of the rat testis. Biol. Reprod. 63, 1465–1472 (2000)
Holdcraft, R. W. & Braun, R. E. Androgen receptor function is required in Sertoli cells for the terminal differentiation of haploid spermatids. Development 131, 459–467 (2004)
Brugnera, E. et al. Unconventional Rac-GEF activity is mediated through the Dock180-ELMO complex. Nature Cell Biol. 4, 574–582 (2002)
Miyanishi, M. et al. Identification of Tim4 as a phosphatidylserine receptor. Nature 450, 435–439 (2007)
Park, S.-Y. et al. Rapid cell corpse clearance by stabilin-2, a membrane phosphatidylserine receptor. Cell Death Differ. 15, 192–201 (2008)
Shiratsuchi, A., Kawasaki, Y., Ikemoto, M., Arai, H. & Nakanishi, Y. Role of class B scavenger receptor type I in phagocytosis of apoptotic rat spermatogenic cells by Sertoli cells. J. Biol. Chem. 274, 5901–5908 (1999)
Gillot, I. et al. Germ cells and fatty acids induce translocation of CD36 scavenger receptor to the plasma membrane of Sertoli cells. J. Cell Sci. 118, 3027–3035 (2005)
Lu, Q. et al. Tyro-3 family receptors are essential regulators of mammalian spermatogenesis. Nature 398, 723–728 (1999)
Chen, Y. et al. Functions of TAM RTKs in regulating spermatogenesis and male fertility in mice. Reproduction 138, 655–666 (2009)
Hanayama, R. et al. Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science 304, 1147–1150 (2004)
Akakura, S. et al. The opsonin MFG-E8 is a ligand for the alphavbeta5 integrin and triggers DOCK180-dependent Rac1 activation for the phagocytosis of apoptotic cells. Exp. Cell Res. 292, 403–416 (2004)
A-Gonzalez, N. et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity 31, 245–258 (2009)
Meyers, E. N., Lewandoski, M. & Martin, G. R. An Fgf8 mutant allelic series generated by Cre- and Flp-mediated recombination. Nature Genet. 18, 136–141 (1998)
Lakso, M. et al. Efficient in vivo manipulation of mouse genomic sequences at the zygote stage. Proc. Natl Acad. Sci. USA 93, 5860–5865 (1996)
Srinivas, S. et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 1, 4 (2001)
Xiong, W. et al. Gas6 and the Tyro 3 receptor tyrosine kinase subfamily regulate the phagocytic function of Sertoli cells. Reproduction 135, 77–87 (2008)
Lu, M. et al. PH domain of ELMO functions in trans to regulate Rac activation via Dock180. Nature Struct. Mol. Biol. 11, 756–762 (2004)
Lysiak, J. J. et al. Essential role of neutrophils in germ cell-specific apoptosis following ischemia/reperfusion injury of the mouse testis. Biol. Reprod. 65, 718–725 (2001)
Ogawa, T. Aréchaga, J. M., Avarbock, M. R. & Brinster, R. L. Transplantation of testis germinal cells into mouse seminiferous tubules. Int. J. Dev. Biol. 41, 111–122 (1997)
Acknowledgements
We thank T. Turner, K. Tung and members of the Ravichandran and Lysiak groups for suggestions. We also thank L. Haney, A. C. Tosello-Trampont, J. Kim and S. Clugston for technical assistance. This work was supported by funding from the National Institutes of Health (to K.S.R. and J.J.L.) and the American Cancer Society (to M.R.E.). K.S.R. is a Bill Benter fellow of the American Asthma Foundation.
Author information
Authors and Affiliations
Contributions
The project was designed and the experiments planned by J.J.L., K.S.R and M.R.E. M.R.E. generated the Elmo1 knockout mice and performed and analysed most of the experiments in this study. S.Z. carried out the experiments using ELMO–GFP constructs. R.I.W. performed all of the immunohistochemistry, Sertoli cell extraction and sperm counts. M.A.R. performed the microbubble uptake experiments. I.J.J. performed the phagocytosis assays on non-Sertoli cells. D.P. performed phagocytosis assays and synthesized and characterized BAI1-TSR. Caenorhabditis elegans mismigration analysis using Elmo1 transgenes was conducted by J.M.K. J.Z. assisted with immunoblotting and quantitative PCR studies of Sertoli cells. J.J.L. performed intratesticular injections. M.R.E., K.S.R. and J.J.L. wrote the manuscript with input from the co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-6 with legends. (PDF 6096 kb)
Rights and permissions
About this article
Cite this article
Elliott, M., Zheng, S., Park, D. et al. Unexpected requirement for ELMO1 in clearance of apoptotic germ cells in vivo. Nature 467, 333–337 (2010). https://doi.org/10.1038/nature09356
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09356
This article is cited by
-
Mesenchymal stem cell-derived apoptotic bodies alleviate alveolar bone destruction by regulating osteoclast differentiation and function
International Journal of Oral Science (2023)
-
Identifying pleiotropic variants and candidate genes for fertility and reproduction traits in Holstein cattle via association studies based on imputed whole-genome sequence genotypes
BMC Genomics (2022)
-
PA1 participates in the maintenance of blood–testis barrier integrity via cooperation with JUN in the Sertoli cells of mice
Cell & Bioscience (2022)
-
Biasing the conformation of ELMO2 reveals that myoblast fusion can be exploited to improve muscle regeneration
Nature Communications (2022)
-
Role of ELMO1 in inflammation and cancer—clinical implications
Cellular Oncology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.