Abstract
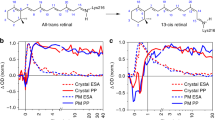
Ever since the conversion of the 11-cis retinal chromophore to its all-trans form in rhodopsin was identified as the primary photochemical event in vision1, experimentalists and theoreticians have tried to unravel the molecular details of this process. The high quantum yield of 0.65 (ref. 2), the production of the primary ground-state rhodopsin photoproduct within a mere 200 fs (refs 3–7), and the storage of considerable energy in the first stable bathorhodopsin intermediate8 all suggest an unusually fast and efficient photoactivated one-way reaction9. Rhodopsin's unique reactivity is generally attributed to a conical intersection between the potential energy surfaces of the ground and excited electronic states10,11 enabling the efficient and ultrafast conversion of photon energy into chemical energy12,13,14,15,16. But obtaining direct experimental evidence for the involvement of a conical intersection is challenging: the energy gap between the electronic states of the reacting molecule changes significantly over an ultrashort timescale, which calls for observational methods that combine high temporal resolution with a broad spectral observation window. Here we show that ultrafast optical spectroscopy with sub-20-fs time resolution and spectral coverage from the visible to the near-infrared allows us to follow the dynamics leading to the conical intersection in rhodopsin isomerization. We track coherent wave-packet motion from the photoexcited Franck–Condon region to the photoproduct by monitoring the loss of reactant emission and the subsequent appearance of photoproduct absorption, and find excellent agreement between the experimental observations and molecular dynamics calculations that involve a true electronic state crossing. Taken together, these findings constitute the most compelling evidence to date for the existence and importance of conical intersections in visual photochemistry.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Yoshizawa, T. & Wald, G. Pre-lumirhodopsin and the bleaching of visual pigments. Nature 197, 1279–1286 (1963)
Kim, J. E., Tauber, M. J. & Mathies, R. A. Wavelength dependent cis-trans isomerization in vision. Biochemistry 40, 13774–13778 (2001)
Schoenlein, R. W., Peteanu, L. A., Mathies, R. A. & Shank, C. V. The first step in vision: femtosecond isomerization of rhodopsin. Science 254, 412–415 (1991)
Haran, G., Morlino, E. A., Matthes, J., Callender, R. H. & Hochstrasser, R. M. Femtosecond polarized pump-probe and stimulated emission spectroscopy of the isomerization reaction of rhodopsin. J. Phys. Chem. A 103, 2202–2207 (1999)
Chosrowjan, H. et al. Rhodopsin emission in real time: a new aspect of the primary event in vision. J. Am. Chem. Soc. 120, 9706–9707 (1998)
Kandori, H. et al. Excited-state dynamics of rhodopsin probed by femtosecond fluorescence spectroscopy. Chem. Phys. Lett. 334, 271–276 (2001)
Kochendoerfer, G. G. & Mathies, R. A. Spontaneous emission study of the femtosecond isomerization dynamics of rhodopsin. J. Phys. Chem. 100, 14526–14532 (1996)
Schick, G. A., Cooper, T. M., Holloway, R. A., Murray, L. P. & Birge, R. R. Energy storage in the primary photochemical events of rhodopsin and isorhodopsin. Biochemistry 26, 2556–2562 (1987)
Mathies, R. A. & Lugtenburg, J. in Handbook of Biological Physics Vol. 3, Molecular Mechanisms in Visual Transduction (eds Stavenga, D. G., DeGrip, W. J. & Pugh E. N.) 55–90 (Elsevier Science, 2000)
Garavelli, M., Celani, P., Bernardi, F., Robb, M. A. & Olivucci, M. The C5H6NH2 + protonated Shiff base: an ab initio minimal model for retinal photoisomerization. J. Am. Chem. Soc. 119, 6891–6901 (1997)
González-Luque, R. et al. Computational evidence in favor of a two-state, two-mode model of the retinal chromophore photoisomerization. Proc. Natl Acad. Sci. USA 97, 9379–9384 (2000)
Klessinger, M. & Michl, J. Excited States and Photochemistry of Organic Molecules (VCH, 1994)
Levine, B. G. & Martinez, T. M. Isomerization through conical intersections. Annu. Rev. Phys. Chem. 58, 613–634 (2007)
Frutos, L. M., Andruniow, T., Santoro, F., Ferre, N. & Olivucci, M. Tracking the excited-state time evolution of the visual pigment with multiconfigurational quantum chemistry. Proc. Natl Acad. Sci. USA 104, 7764–7769 (2007)
Hayashi, S., Tajkhorshid, E. & Schulten, K. Photochemical reaction dynamics of the primary event of vision studied by means of a hybrid molecular simulation. Biophys. J. 96, 403–416 (2009)
Tomasello, G. et al. Electrostatic control of the photoisomerization efficiency and optical properties in visual pigments: on the role of counterion quenching. J. Am. Chem. Soc. 131, 5172–5186 (2009)
Manzoni, C., Polli, D. & Cerullo, G. Two-color pump-probe system broadly tunable over the visible and the near infrared with sub-30 fs temporal resolution. Rev. Sci. Instrum. 77, 023103 (2006)
Weingart, O. The twisted C11 = C12 bond of the rhodopsin chromophore — a photochemical hot spot. J. Am. Chem. Soc. 129, 10618–10619 (2007)
Hayashi, S., Tajkhorshid, E. & Schulten, K. Molecular dynamics simulation of bacteriorhodopsin’s photoisomerization using ab initio forces for the excited chromophore. Biophys. J. 85, 1440–1449 (2003)
Groenhof, G. et al. Photoactivation of the photoactive yellow protein: why photon absorption triggers a trans-to-cis isomerization of the chromophore in the protein. J. Am. Chem. Soc. 126, 4228–4233 (2004)
Groenhof, G. et al. Ultrafast deactivation of an excited cytosine-guanine base pair in DNA. J. Am. Chem. Soc. 129, 6812–6819 (2007)
Hudock, H. R. et al. Ab initio molecular dynamics and time-resolved photoelectron spectroscopy of electronically excited uracil and thymine. J. Phys. Chem. A 111, 8500–8508 (2007)
Hudock, H. R. & Martinez, T. J. Excited-state dynamics of cytosine reveal multiple intrinsic subpicosecond pathways. ChemPhysChem 9, 2486–2490 (2008)
Andersson, K., Malmqvist, P.-A. & Roos, B. O. Second-order perturbation theory with a complete active space self-consistent field reference function. J. Chem. Phys. 96, 1218–1226 (1992)
Tao, H., Levine, B. G. & Martinez, T. J. Ab initio multiple spawning dynamics using multi-state second-order perturbation. J. Phys. Chem. A 113, 13656–13662 (2009)
Kukura, P., McCamant, D. W., Yoon, S., Wandschneider, D. B. & Mathies, R. A. Structural observation of the primary isomerization in vision with femtosecond-stimulated Raman. Science 310, 1006–1009 (2005)
Wang, Q., Schoenlein, R. W., Peteanu, L. A., Mathies, R. A. & Shank, C. V. Vibrationally coherent photochemistry in the femtosecond primary event of vision. Science 266, 422–424 (1994)
Warshel, A. Bicycle-pedal model for the first step in the vision process. Nature 260, 679–683 (1976)
Warshel, A. & Barboy, N. Energy storage and reaction pathways in the first step of the vision process. J. Am. Chem. Soc. 104, 1469–1476 (1982)
Okada, T. et al. The retinal conformation and its environment in rhodopsin in light of a new 2.2 Å crystal structure. J. Mol. Biol. 342, 571–583 (2004)
De Grip, W. J., Daemen, F. J. M. & Bonting, S. L. Isolation and purification of bovine rhodopsin. Methods Enzymol. 67, 301–320 (1980)
Polli, D., Lüer, L. & Cerullo, G. High-time-resolution pump-probe system with broadband detection for the study of time-domain vibrational dynamics. Rev. Sci. Instrum. 78, 103108 (2007)
Lin, H. & Truhlar, D. G. QM/MM: what have we learned, where are we, and where do we go from here? Theor. Chem. Acc. 117, 185–199 (2007)
Altoè, P., Stenta, M., Bottoni, A. & Garavelli, M. A tunable QM/MM approach to chemical reactivity, structure and physico-chemical properties prediction. Theor. Chem. Acc. 118, 219–240 (2007)
Singh, U. C. & Kollman, P. A. A combined ab initio quantum mechanical and molecular mechanical method for carrying out simulations on complex molecular systems: applications to the CH3Cl + Cl− exchange reaction and gas phase protonation of polyethers. J. Comput. Chem. 7, 718–730 (1986)
Werner, H.-J. et al. MOLPRO, version 2008.1, a package of ab initio programs. (Cardiff University, UK, 2008)
Karlström, G. et al. MOLCAS: a program package for computational chemistry. Comput. Mater. Sci. 28, 222–239 (2003)
Case, D. A. et al. The Amber biomolecular simulation programs. J. Comput. Chem. 26, 1668–1688 (2005)
Stenta, M. et al. The catalytic activity of proline racemase: a quantum mechanical/molecular mechanical study. J. Phys. Chem. B 112, 1057–1059 (2008)
Roos, B. O. in Advances in Chemical Physics: Ab Initio Methods in Quantum Chemistry Part 2, Vol. 69 (ed. Lawley, K. P.) 399–446 (Wiley & Sons, 1987)
Cembran, A., Bernardi, F., Olivucci, M. & Garavelli, M. Counterion controlled photoisomerization of retinal chromophore models: a computational investigation. J. Am. Chem. Soc. 126, 16018–16037 (2004)
Sloane, C. S. & Hase, W. L. On the dynamics of state selected unimolecular reactions: chloroacetylene dissociation and predissociation. J. Chem. Phys. 66, 1523–1533 (1977)
Verlet, L. Computer “experiments” on classical fluids. I. Thermodynamical properties of Lennard-Jones molecules. Phys. Rev. 159, 98–103 (1967)
Weingart, O., Schapiro, I. & Buss, V. Photochemistry of visual pigment chromophore models by ab initio molecular dynamics. J. Phys. Chem. B 111, 3782–3788 (2007)
Försberg, N. & Malmqvist, P.-Å. Multiconfiguration perturbation theory with imaginary level shift. Chem. Phys. Lett. 274, 196–204 (1997)
Acknowledgements
M.G., O.W., G.O., G.T. and P.A. thank E4-Computer Engineering S.p.A. for computational time and technical assistance. Part of this study was financially supported by the DFG (FOR490) and by the PRIN programme (2008JKBBK4). P.K. is supported by a Career Acceleration Fellowship awarded by the UK Engineering and Physical Sciences Research Council (EP/H003541/1).
Author information
Authors and Affiliations
Contributions
R.A.M., P.K., M.G. and G.C. conceived the project and contributed the original scientific ideas to this work. D.P., C.M. and D.B. executed the experiments. K.M.S. carried out rhodopsin sample preparation/purification. P.A., O.W. and G.O. planned calculations, P.A. and O.W. executed them. G.T. collected calibration data. All authors discussed the results and contributed to the preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-9 with legends, Supplementary Tables 1-3, a Supplementary Discussion and additional references. (PDF 1757 kb)
Supplementary Movie 1
This movie shows the 11-cis->all-trans photoinduced motion and the frustrated isomerization path - see Supplementary Information file for full legend. (MOV 5994 kb)
Rights and permissions
About this article
Cite this article
Polli, D., Altoè, P., Weingart, O. et al. Conical intersection dynamics of the primary photoisomerization event in vision. Nature 467, 440–443 (2010). https://doi.org/10.1038/nature09346
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09346
This article is cited by
-
Tunnelling of electrons via the neighboring atom
Light: Science & Applications (2024)
-
Progress and prospects in nonlinear extreme-ultraviolet and X-ray optics and spectroscopy
Nature Reviews Physics (2023)
-
H2 formation via non-Born-Oppenheimer hydrogen migration in photoionized ethane
Nature Communications (2023)
-
Optical control of ultrafast structural dynamics in a fluorescent protein
Nature Chemistry (2023)
-
Absolute excited state molecular geometries revealed by resonance Raman signals
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.