Abstract
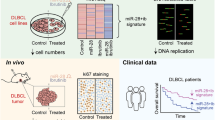
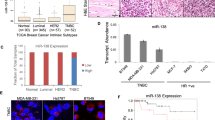
MicroRNAs (miRNAs) belong to a recently discovered class of small RNA molecules that regulate gene expression at the post-transcriptional level. miRNAs have crucial functions in the development and establishment of cell identity, and aberrant metabolism or expression of miRNAs has been linked to human diseases, including cancer1. Components of the miRNA machinery and miRNAs themselves are involved in many cellular processes that are altered in cancer, such as differentiation, proliferation and apoptosis. Some miRNAs, referred to as oncomiRs2, show differential expression levels in cancer and are able to affect cellular transformation, carcinogenesis and metastasis, acting either as oncogenes or tumour suppressors. The phenomenon of ‘oncogene addiction’ reveals that despite the multistep nature of tumorigenesis, targeting of certain single oncogenes can have therapeutic value3,4, and the possibility of oncomiR addiction has been proposed but never demonstrated3. MicroRNA-21 (miR-21) is a unique miRNA in that it is overexpressed in most tumour types analysed so far. Despite great interest in miR-21, most of the data implicating it in cancer have been obtained through miRNA profiling and limited in vitro functional assays. To explore the role of miR-21 in cancer in vivo, we used Cre and Tet-off technologies to generate mice conditionally expressing miR-21. Here we show that overexpression of miR-21 leads to a pre-B malignant lymphoid-like phenotype, demonstrating that mir-21 is a genuine oncogene. When miR-21 was inactivated, the tumours regressed completely in a few days, partly as a result of apoptosis. These results demonstrate that tumours can become addicted to oncomiRs and support efforts to treat human cancers through pharmacological inactivation of miRNAs such as miR-21.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Medina, P. P. & Slack, F. J. microRNAs and cancer: an overview. Cell Cycle 7, 2485–2492 (2008)
Esquela-Kerscher, A. & Slack, F. J. Oncomirs—microRNAs with a role in cancer. Nature Rev. Cancer 6, 259–269 (2006)
Sharma, S. V. & Settleman, J. Oncogene addiction: setting the stage for molecularly targeted cancer therapy. Genes Dev. 21, 3214–3231 (2007)
Weinstein, I. B. & Joe, A. Oncogene addiction. Cancer Res. 68, 3077–3080 (2008)
Costinean, S. et al. Ship and C/ebp-β are targeted by miR-155 in B cells of Eμ-miR-155 transgenic mice. Blood 117, 1374–1382 (2009)
Si, M. L. et al. miR-21-mediated tumor growth. Oncogene 26, 2799–2803 (2007)
Iorio, M. V. et al. MicroRNA gene expression deregulation in human breast cancer. Cancer Res. 65, 7065–7070 (2005)
Chan, J. A., Krichevsky, A. M. & Kosik, K. S. MicroRNA-21 is an antiapoptotic factor in human glioblastoma cells. Cancer Res. 65, 6029–6033 (2005)
Schetter, A. J. et al. MicroRNA expression profiles associated with prognosis and therapeutic outcome in colon adenocarcinoma. J. Am. Med. Assoc. 299, 425–436 (2008)
Volinia, S. et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc. Natl Acad. Sci. USA 103, 2257–2261 (2006)
Fulci, V. et al. Quantitative technologies establish a novel microRNA profile of chronic lymphocytic leukemia. Blood 109, 4944–4951 (2007)
Lawrie, C. H. et al. MicroRNA expression distinguishes between germinal center B cell-like and activated B cell-like subtypes of diffuse large B cell lymphoma. Int. J. Cancer 121, 1156–1161 (2007)
Jongen-Lavrencic, M., Sun, S. M., Dijkstra, M. K., Valk, P. J. & Lowenberg, B. MicroRNA expression profiling in relation to the genetic heterogeneity of acute myeloid leukemia. Blood 111, 5078–5085 (2008)
Navarro, A. et al. MicroRNA expression profiling in classic Hodgkin lymphoma. Blood 111, 2825–2832 (2008)
Mao, J., Barrow, J., McMahon, J., Vaughan, J. & McMahon, A. P. An ES cell system for rapid, spatial and temporal analysis of gene function in vitro and in vivo . Nucleic Acids Res. 33, e155 (2005)
Zambrowicz, B. P. et al. Disruption of overlapping transcripts in the ROSA βgeo 26 gene trap strain leads to widespread expression of β-galactosidase in mouse embryos and hematopoietic cells. Proc. Natl Acad. Sci. USA 94, 3789–3794 (1997)
Petersen, P. H., Zou, K., Hwang, J. K., Jan, Y. N. & Zhong, W. Progenitor cell maintenance requires numb and numblike during mouse neurogenesis. Nature 419, 929–934 (2002)
Morse, H. C. et al. Bethesda proposals for classification of lymphoid neoplasms in mice. Blood 100, 246–258 (2002)
Pennycook, J. L., Chang, Y., Celler, J., Phillips, R. A. & Wu, G. E. High frequency of normal DJH joints in B cell progenitors in severe combined immunodeficiency mice. J. Exp. Med. 178, 1007–1016 (1993)
Li, J. et al. MiR-21 indicates poor prognosis in tongue squamous cell carcinomas as an apoptosis inhibitor. Clin. Cancer Res. 15, 3998–4008 (2009)
Seike, M. et al. MiR-21 is an EGFR-regulated anti-apoptotic factor in lung cancer in never-smokers. Proc. Natl Acad. Sci. USA 106, 12085–12090 (2009)
Imai, Y. et al. Caspase inhibitor, ZVAD-fmk, facilitates engraftment of donor hematopoietic stem cells in intra-bone marrow–bone marrow transplantation. Stem Cells Dev. 10.1089/scd.2009.0251
Felsher, D. W. & Bishop, J. M. Reversible tumorigenesis by MYC in hematopoietic lineages. Mol. Cell 4, 199–207 (1999)
Yokoyama, K. & Imamoto, F. Transcriptional control of the endogenous MYC protooncogene by antisense RNA. Proc. Natl Acad. Sci. USA 84, 7363–7367 (1987)
Li, T., Li, D., Sha, J., Sun, P. & Huang, Y. MicroRNA-21 directly targets MARCKS and promotes apoptosis resistance and invasion in prostate cancer cells. Biochem. Biophys. Res. Commun. 383, 280–285 (2009)
Zhu, S. et al. MicroRNA-21 targets tumor suppressor genes in invasion and metastasis. Cell Res. 18, 350–359 (2008)
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000)
Huettner, C. S., Zhang, P., Van Etten, R. A. & Tenen, D. G. Reversibility of acute B-cell leukaemia induced by BCR-ABL1. Nature Genet. 24, 57–60 (2000)
Weinstein, I. B. & Joe, A. K. Mechanisms of disease: oncogene addiction—a rationale for molecular targeting in cancer therapy. Nature Clin. Pract. Oncol. 3, 448–457 (2006)
Koopman, G. et al. Annexin V for flow cytometric detection of phosphatidylserine expression on B cells undergoing apoptosis. Blood 84, 1415–1420 (1994)
Acknowledgements
We thank X. Liang for help in mouse colony maintenance and genotyping; D. Scavone and T. Nottoli for assistance with generating the transgenic mice; Yale rodent care staff M. Bonk, K. Mclaughlin, J. Graham, D. Clark; C. Zeiss, L. Johnson and G. Terwilliger for help with the necropsy/pathological analysis; and I. Babar, P. Trang and J. Sklar for critical reading of the manuscript. P.P.M. is the recipient of a Postdoctoral Fellowship from Hope Funds for Cancer Research. This work was funded by a grant from the James McDonnell Foundation to F.J.S. and J. B. Weidhaas, and a pilot grant from the Yale Comprehensive Cancer Center.
Author information
Authors and Affiliations
Contributions
F.J.S. designed the project and supervised all the experiments. F.J.S. and M.N. designed the genetically engineered miR-21 mouse. M.N. constructed and tested the mir-21 vector. P.P.M. performed all the phenotyping experiments in the manuscript. F.J.S. and P.P.M. designed the phenotyping experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-22 with legends. (PDF 13808 kb)
Supplementary Movie 1
This movie shows NesCre8, mir-21LSL-Tetoff mouse phenotype. Seconds 0-4: The mouse situated at 30° suffers from a conspicuous axillary lymphadenopathy and incipient paraparesis. Seconds 5-7: The mouse situated at 30° suffers from a conspicuous paraparesis. (MOV 8928 kb)
Rights and permissions
About this article
Cite this article
Medina, P., Nolde, M. & Slack, F. OncomiR addiction in an in vivo model of microRNA-21-induced pre-B-cell lymphoma. Nature 467, 86–90 (2010). https://doi.org/10.1038/nature09284
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09284
This article is cited by
-
MiRNA-related metastasis in oral cancer: moving and shaking
Cancer Cell International (2023)
-
The various role of microRNAs in breast cancer angiogenesis, with a special focus on novel miRNA-based delivery strategies
Cancer Cell International (2023)
-
Epigenetic regulation in hematopoiesis and its implications in the targeted therapy of hematologic malignancies
Signal Transduction and Targeted Therapy (2023)
-
The relationship between microRNAs and bladder cancer: are microRNAs useful to predict bladder cancer in suspicious patients?
International Urology and Nephrology (2023)
-
Evaluation of miRNA-21 and CA-125 as a promising diagnostic biomarker in patients with ovarian cancer
Egyptian Journal of Medical Human Genetics (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.