Abstract
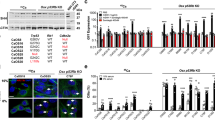
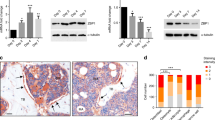
Mutation of the retinoblastoma gene (RB1) tumour suppressor occurs in one-third of all human tumours and is particularly associated with retinoblastoma and osteosarcoma1. Numerous functions have been ascribed to the product of the human RB1 gene, the retinoblastoma protein (pRb). The best known is pRb’s ability to promote cell-cycle exit through inhibition of the E2F transcription factors and the transcriptional repression of genes encoding cell-cycle regulators1. In addition, pRb has been shown in vitro to regulate several transcription factors that are master differentiation inducers2. Depending on the differentiation factor and cellular context, pRb can either suppress or promote their transcriptional activity. For example, pRb binds to Runx2 and potentiates its ability to promote osteogenic differentiation in vitro3. In contrast, pRb acts with E2F to suppress peroxisome proliferator-activated receptor γ subunit (PPAR-γ), the master activator of adipogenesis4,5. Because osteoblasts and adipocytes can both arise from mesenchymal stem cells, these observations suggest that pRb might play a role in the choice between these two fates. However, so far, there is no evidence for this in vivo. Here we use mouse models to address this hypothesis in mesenchymal tissue development and tumorigenesis. Our data show that Rb status plays a key role in establishing fate choice between bone and brown adipose tissue in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Burkhart, D. L. & Sage, J. Cellular mechanisms of tumour suppression by the retinoblastoma gene. Nature Rev. Cancer 8, 671–682 (2008)
Korenjak, M. & Brehm, A. E2F-Rb complexes regulating transcription of genes important for differentiation and development. Curr. Opin. Genet. Dev. 15, 520–527 (2005)
Thomas, D. M. et al. The retinoblastoma protein acts as a transcriptional coactivator required for osteogenic differentiation. Mol. Cell 8, 303–316 (2001)
Fajas, L. et al. The retinoblastoma-histone deacetylase 3 complex inhibits PPARγ and adipocyte differentiation. Dev. Cell 3, 903–910 (2002)
Fajas, L. et al. E2Fs regulate adipocyte differentiation. Dev. Cell 3, 39–49 (2002)
Clark, J. C., Dass, C. R. & Choong, P. F. A review of clinical and molecular prognostic factors in osteosarcoma. J. Cancer Res. Clin. Oncol. 134, 281–297 (2008)
Kansara, M. & Thomas, D. M. Molecular pathogenesis of osteosarcoma. DNA Cell Biol. 26, 1–18 (2007)
Sage, J. et al. Acute mutation of retinoblastoma gene function is sufficient for cell cycle re-entry. Nature 424, 223–228 (2003)
Jonkers, J. et al. Synergistic tumor suppressor activity of BRCA2 and p53 in a conditional mouse model for breast cancer. Nature Genet. 29, 418–425 (2001)
Logan, M. et al. Expression of Cre recombinase in the developing mouse limb bud driven by a Prxl enhancer. Genesis 33, 77–80 (2002)
Hansen, J. B. et al. Retinoblastoma protein functions as a molecular switch determining white versus brown adipocyte differentiation. Proc. Natl Acad. Sci. USA 101, 4112–4117 (2004)
Scime, A. et al. Rb and p107 regulate preadipocyte differentiation into white versus brown fat through repression of PGC-1alpha. Cell Metab. 2, 283–295 (2005)
Dali-Youcef, N. et al. Adipose tissue-specific inactivation of the retinoblastoma protein protects against diabesity because of increased energy expenditure. Proc. Natl Acad. Sci. USA 104, 10703–10708 (2007)
Rodda, S. J. & McMahon, A. P. Distinct roles for Hedgehog and canonical Wnt signaling in specification, differentiation and maintenance of osteoblast progenitors. Development 133, 3231–3244 (2006)
Berman, S. D. et al. Metastatic osteosarcoma induced by inactivation of Rb and p53 in the osteoblast lineage. Proc. Natl Acad. Sci. USA 105, 11851–11856 (2008)
Lengner, C. J. et al. Osteoblast differentiation and skeletal development are regulated by Mdm2-p53 signaling. J. Cell Biol. 172, 909–921 (2006)
Tallquist, M. D. & Soriano, P. Epiblast-restricted Cre expression in MORE mice: a tool to distinguish embryonic vs. extra-embryonic gene function. Genesis 26, 113–115 (2000)
Wu, L. et al. Extra-embryonic function of Rb is essential for embryonic development and viability. Nature 421, 942–947 (2003)
Berman, S. D. et al. The retinoblastoma protein tumor suppressor is important for appropriate osteoblast differentiation and bone development. Mol. Cancer Res. 6, 1440–1451 (2008)
Barski, A., Pregizer, S. & Frenkel, B. Identification of transcription factor target genes by ChIP display. Methods Mol. Biol. 455, 177–190 (2008)
Acknowledgements
We thank T. Jacks, C. Tabin and A. McMahon for providing key mutant mouse strains, K. Lane for pCW22, M. Hemann for the Rb shRNA, the University of Iowa Gene Transfer Vector Core for the adenoviral Cre and GFP vectors, and G. Karsenty for p6OSE2-Luc and control p4Luc. We also thank members of the Lees laboratory, S. Mukherjee and M. Hemann for input during this study. This work was supported by an National Cancer Institute/National Institutes of Health grant to J.A.L., who is a Ludwig Scholar at MIT.
Author information
Authors and Affiliations
Contributions
E.C. conducted all the experiments with assistance from J.A.Q.-E. in the Rb re-introduction study, P.S.D. and S.D.B. in the generation and analysis of compound mutant mouse strains and S.N. for the LSL-LacZ;Prx1-Cre embryo analysis. E.C. and J.A.L. were responsible for conceiving this study, interpreting the data and manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-6 with legends. (PDF 4373 kb)
Rights and permissions
About this article
Cite this article
Calo, E., Quintero-Estades, J., Danielian, P. et al. Rb regulates fate choice and lineage commitment in vivo. Nature 466, 1110–1114 (2010). https://doi.org/10.1038/nature09264
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09264
This article is cited by
-
RETINOBLASTOMA-RELATED interactions with key factors of the RNA-directed DNA methylation (RdDM) pathway and its influence on root development
Planta (2023)
-
Hepatocellular carcinoma evades RB1-induced senescence by activating the FOXM1–FOXO1 axis
Oncogene (2022)
-
Retinoblastoma patient-derived stem cells—an in vivo model to study the role of RB1 in adipogenesis
Histochemistry and Cell Biology (2022)
-
Spinal cord injury reprograms muscle fibroadipogenic progenitors to form heterotopic bones within muscles
Bone Research (2022)
-
Histone demethylase KDM5A promotes tumorigenesis of osteosarcoma tumor
Cell Death Discovery (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.