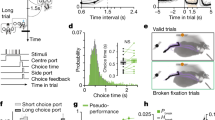
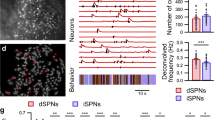
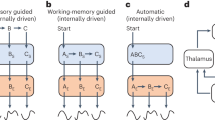
Abstract
Learning new action sequences subserves a plethora of different abilities such as escaping a predator, playing the piano, or producing fluent speech. Proper initiation and termination of each action sequence is critical for the organization of behaviour, and is compromised in nigrostriatal disorders like Parkinson’s and Huntington’s diseases. Using a self-paced operant task in which mice learn to perform a particular sequence of actions to obtain an outcome, we found neural activity in nigrostriatal circuits specifically signalling the initiation or the termination of each action sequence. This start/stop activity emerged during sequence learning, was specific for particular actions, and did not reflect interval timing, movement speed or action value. Furthermore, genetically altering the function of striatal circuits disrupted the development of start/stop activity and selectively impaired sequence learning. These results have important implications for understanding the functional organization of actions and the sequence initiation and termination impairments observed in basal ganglia disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lashley, K. S. in Cerebral Mechanisms in Behavior (ed. Jeffress, L. A.) (John Wiley, 1951)
Gallistel, C. R. The Organization of Action: A New Synthesis (Lawrence Erlbaum Associates, 1980)
Grillner, S. & Wallén, P. Central pattern generators for locomotion, with special reference to vertebrates. Annu. Rev. Neurosci. 8, 233–261 (1985)
Marder, E. & Bucher, D. Central pattern generators and the control of rhythmic movements. Curr. Biol. 11, R986–R996 (2001)
Hikosaka, O. et al. Parallel neural networks for learning sequential procedures. Trends Neurosci. 22, 464–471 (1999)
Yin, H. H. et al. Dynamic reorganization of striatal circuits during the acquisition and consolidation of a skill. Nature Neurosci. 12, 333–341 (2009)
Graybiel, A. M. The basal ganglia and chunking of action repertoires. Neurobiol. Learn. Mem. 70, 119–136 (1998)
Hikosaka, O., Miyashita, K., Miyachi, S., Sakai, K. & Lu, X. Differential roles of the frontal cortex, basal ganglia, and cerebellum in visuomotor sequence learning. Neurobiol. Learn. Mem. 70, 137–149 (1998)
Bailey, K. R. & Mair, R. G. The role of striatum in initiation and execution of learned action sequences in rats. J. Neurosci. 26, 1016–1025 (2006)
Benecke, R., Rothwell, J. C., Dick, J. P., Day, B. L. & Marsden, C. D. Disturbance of sequential movements in patients with Parkinson’s disease. Brain 110, 361–379 (1987)
Agostino, R., Berardelli, A., Formica, A., Accornero, N. & Manfredi, M. Sequential arm movements in patients with Parkinson’s disease, Huntington’s disease and dystonia. Brain 115, 1481–1495 (1992)
Castiello, U., Stelmach, G. E. & Lieberman, A. N. Temporal dissociation of the prehension pattern in Parkinson’s disease. Neuropsychologia 31, 395–402 (1993)
Phillips, J. G., Chiu, E., Bradshaw, J. L. & Iansek, R. Impaired movement sequencing in patients with Huntington’s disease: a kinematic analysis. Neuropsychologia 33, 365–369 (1995)
Willingham, D. B. & Koroshetz, W. J. Evidence for dissociable motor skills in Huntington’s disease patients. Psychobiology 21, 173–182 (1993)
Stefanova, E. D., Kostic, V. S., Ziropadja, L., Markovic, M. & Ocic, G. G. Visuomotor skill learning on serial reaction time task in patients with early Parkinson’s disease. Mov. Disord. 15, 1095–1103 (2000)
Boyd, L. A. et al. Motor sequence chunking is impaired by basal ganglia stroke. Neurobiol. Learn. Mem. 92, 35–44 (2009)
Fujii, N. & Graybiel, A. M. Representation of action sequence boundaries by macaque prefrontal cortical neurons. Science 301, 1246–1249 (2003)
Aldridge, J. W. & Berridge, K. C. Coding of serial order by neostriatal neurons: a “natural action” approach to movement sequence. J. Neurosci. 18, 2777–2787 (1998)
Meyer-Luehmann, M., Thompson, J. F., Berridge, K. C. & Aldridge, J. W. Substantia nigra pars reticulata neurons code initiation of a serial pattern: implications for natural action sequences and sequential disorders. Eur. J. Neurosci. 16, 1599–1608 (2002)
Dang, M. T. et al. Disrupted motor learning and long-term synaptic plasticity in mice lacking NMDAR1 in the striatum. Proc. Natl Acad. Sci. USA 103, 15254–15259 (2006)
Gong, S. et al. Targeting Cre recombinase to specific neuron populations with bacterial artificial chromosome constructs. J. Neurosci. 27, 9817–9823 (2007)
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nature Neurosci. 8, 1263–1268 (2005)
Lima, S. Q., Hromadka, T., Znamenskiy, P. & Zador, A. M. PINP: a new method of tagging neuronal populations for identification during in vivo electrophysiological recording. PLoS ONE 4 e6099 10.1371/journal.pone.0006099 (2009)
Meck, W. H., Penney, T. B. & Pouthas, V. Cortico-striatal representation of time in animals and humans. Curr. Opin. Neurobiol. 18, 145–152 (2008)
Samejima, K., Ueda, Y., Doya, K. & Kimura, M. Representation of action-specific reward values in the striatum. Science 310, 1337–1340 (2005)
Lau, B. & Glimcher, P. W. Value representations in the primate striatum during matching behavior. Neuron 58, 451–463 (2008)
Morris, G., Nevet, A., Arkadir, D., Vaadia, E. & Bergman, H. Midbrain dopamine neurons encode decisions for future action. Nature Neurosci. 9, 1057–1063 (2006)
Roesch, M. R., Calu, D. J. & Schoenbaum, G. Dopamine neurons encode the better option in rats deciding between differently delayed or sized rewards. Nature Neurosci. 10, 1615–1624 (2007)
Calabresi, P., Pisani, A., Mercuri, N. B. & Bernardi, G. Long-term potentiation in the striatum is unmasked by removing the voltage-dependent magnesium block of NMDA receptor channels. Eur. J. Neurosci. 4, 929–935 (1992)
Shen, W., Flajolet, M., Greengard, P. & Surmeier, D. J. Dichotomous dopaminergic control of striatal synaptic plasticity. Science 321, 848–851 (2008)
Pomata, P. E., Belluscio, M. A., Riquelme, L. A. & Murer, M. G. NMDA receptor gating of information flow through the striatum in vivo . J. Neurosci. 28, 13384–13389 (2008)
Brainard, M. S. & Doupe, A. J. What songbirds teach us about learning. Nature 417, 351–358 (2002)
Kimura, M. Behaviorally contingent property of movement-related activity of the primate putamen. J. Neurophysiol. 63, 1277–1296 (1990)
Kermadi, I. & Joseph, J. P. Activity in the caudate nucleus of monkey during spatial sequencing. J. Neurophysiol. 74, 911–933 (1995)
Jog, M. S., Kubota, Y., Connolly, C. I., Hillegaart, V. & Graybiel, A. M. Building neural representations of habits. Science 286, 1745–1749 (1999)
Miyachi, S., Hikosaka, O. & Lu, X. Differential activation of monkey striatal neurons in the early and late stages of procedural learning. Exp. Brain Res. 146, 122–126 (2002)
Schultz, W., Dayan, P. & Montague, P. R. A neural substrate of prediction and reward. Science 275, 1593–1599 (1997)
Schultz, W. Multiple dopamine functions at different time courses. Annu. Rev. Neurosci. 30, 259–288 (2007)
Redgrave, P. & Gurney, K. The short-latency dopamine signal: a role in discovering novel actions? Nature Rev. Neurosci. 7, 967–975 (2006)
Hilário, M. R. F., Clouse, E., Yin, H. H. & Costa, R. M. Endocannabinoid signaling is critical for habit formation. Front. Integr. Neurosci. 1, 6 (2007)
Acknowledgements
We thank Y. Li for the RGS9-Cre mice, C. Gerfen for the TH-Cre mice, K. Nakazawa for the NMDAR1-loxP mice, F. Tecuapetla and S. Lima for help in the optogenetics experiment, G. Luo for genotyping, and D. Lovinger, G. Cui, C. French, C. Gremel and E. Dias-Ferreira for comments on the manuscript. This research was supported by the NIAAA Division of Intramural Clinical and Biological Research, the Champalimaud Neuroscience Programme at Institute Gulbenkian de Ciência and European Research Council Grant 243393 to R.M.C.
Author information
Authors and Affiliations
Contributions
X.J. performed the experiments and analysed the data. R.M.C. conducted the optogenetics experiment. X.J. and R.M.C. designed the experiments and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, References, Supplementary Statistics for Figures 1-5 in the main paper, Supplementary Figures 1-18 with legends and Supplementary Tables 1-2. (PDF 1412 kb)
Rights and permissions
About this article
Cite this article
Jin, X., Costa, R. Start/stop signals emerge in nigrostriatal circuits during sequence learning. Nature 466, 457–462 (2010). https://doi.org/10.1038/nature09263
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09263
This article is cited by
-
Neural inhibition as implemented by an actor-critic model involves the human dorsal striatum and ventral tegmental area
Scientific Reports (2024)
-
Dynamic behaviour restructuring mediates dopamine-dependent credit assignment
Nature (2024)
-
Computational insights on asymmetrical \(D_{1}\) and \(D_{2}\) receptor-mediated chunking: implications for OCD and Schizophrenia
Cognitive Neurodynamics (2024)
-
The Sapap3−/− mouse reconsidered as a comorbid model expressing a spectrum of pathological repetitive behaviours
Translational Psychiatry (2023)
-
Mesolimbic dopamine adapts the rate of learning from action
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.