Abstract
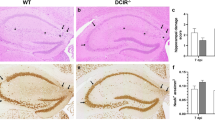
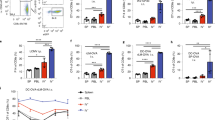
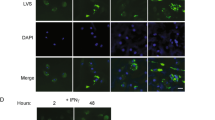
Lymph nodes (LNs) capture microorganisms that breach the body’s external barriers and enter draining lymphatics, limiting the systemic spread of pathogens1. Recent work has shown that CD11b+CD169+ macrophages, which populate the subcapsular sinus (SCS) of LNs, are critical for the clearance of viruses from the lymph and for initiating antiviral humoral immune responses2,3,4. Here we show, using vesicular stomatitis virus (VSV), a relative of rabies virus transmitted by insect bites, that SCS macrophages perform a third vital function: they prevent lymph-borne neurotropic viruses from infecting the central nervous system (CNS). On local depletion of LN macrophages, about 60% of mice developed ascending paralysis and died 7–10 days after subcutaneous infection with a small dose of VSV, whereas macrophage-sufficient animals remained asymptomatic and cleared the virus. VSV gained access to the nervous system through peripheral nerves in macrophage-depleted LNs. In contrast, within macrophage-sufficient LNs VSV replicated preferentially in SCS macrophages but not in adjacent nerves. Removal of SCS macrophages did not compromise adaptive immune responses against VSV, but decreased type I interferon (IFN-I) production within infected LNs. VSV-infected macrophages recruited IFN-I-producing plasmacytoid dendritic cells to the SCS and in addition were a major source of IFN-I themselves. Experiments in bone marrow chimaeric mice revealed that IFN-I must act on both haematopoietic and stromal compartments, including the intranodal nerves, to prevent lethal infection with VSV. These results identify SCS macrophages as crucial gatekeepers to the CNS that prevent fatal viral invasion of the nervous system on peripheral infection.
This is a preview of subscription content, access via your institution
Access options



Similar content being viewed by others
References
von Andrian, U. H. & Mempel, T. R. Homing and cellular traffic in lymph nodes. Nature Rev. Immunol. 3, 867–878 (2003)
Junt, T. et al. Subcapsular sinus macrophages in lymph nodes clear lymph-borne viruses and present them to antiviral B cells. Nature 450, 110–114 (2007)
Phan, T. G., Grigorova, I., Okada, T. & Cyster, J. G. Subcapsular encounter and complement-dependent transport of immune complexes by lymph node B cells. Nature Immunol. 8, 992–1000 (2007)
Carrasco, Y. R. & Batista, F. D. B cells acquire particulate antigen in a macrophage-rich area at the boundary between the follicle and the subcapsular sinus of the lymph node. Immunity 27, 160–171 (2007)
Lyles, D. S. & Rupprecht, C. E. in Fields Virology 5th edn, Vol. 1 (ed. Howley, P. M & Knipe, D. M.) 1363–1408 (Lippincott Williams & Wilkins, 2007)
Hangartner, L., Zinkernagel, R. M. & Hengartner, H. Antiviral antibody responses: the two extremes of a wide spectrum. Nature Rev. Immunol. 6, 231–243 (2006)
Probst, H. C. et al. Histological analysis of CD11c-DTR/GFP mice after in vivo depletion of dendritic cells. Clin. Exp. Immunol. 141, 398–404 (2005)
Purtha, W. E., Chachu, K. A., Virgin, H. W. & Diamond, M. S. Early B-cell activation after West Nile virus infection requires α/β interferon but not antigen receptor signaling. J. Virol. 82, 10964–10974 (2008)
Muller, U. et al. Functional role of type I and type II interferons in antiviral defense. Science 264, 1918–1921 (1994)
Delemarre, F. G., Kors, N., Kraal, G. & van Rooijen, N. Repopulation of macrophages in popliteal lymph nodes of mice after liposome-mediated depletion. J. Leukoc. Biol. 47, 251–257 (1990)
Chandran, K., Sullivan, N. J., Felbor, U., Whelan, S. P. & Cunningham, J. M. Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. Science 308, 1643–1645 (2005)
Hickman, H. D. et al. Direct priming of antiviral CD8+ T cells in the peripheral interfollicular region of lymph nodes. Nature Immunol. 9, 155–165 (2008)
Brundler, M. A. et al. Immunity to viruses in B cell-deficient mice: influence of antibodies on virus persistence and on T cell memory. Eur. J. Immunol. 26, 2257–2262 (1996)
Thomsen, A. R. et al. Cooperation of B cells and T cells is required for survival of mice infected with vesicular stomatitis virus. Int. Immunol. 9, 1757–1766 (1997)
Asselin-Paturel, C. et al. Mouse type I IFN-producing cells are immature APCs with plasmacytoid morphology. Nature Immunol. 2, 1144–1150 (2001)
Lund, J. M. et al. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl Acad. Sci. USA 101, 5598–5603 (2004)
Iparraguirre, A. et al. Two distinct activation states of plasmacytoid dendritic cells induced by influenza virus and CpG 1826 oligonucleotide. J. Leukoc. Biol. 83, 610–620 (2008)
Detje, C. N. et al. Local type I IFN receptor signaling protects against virus spread within the central nervous system. J. Immunol. 182, 2297–2304 (2009)
Jung, S. et al. In vivo depletion of CD11c+ dendritic cells abrogates priming of CD8+ T cells by exogenous cell-associated antigens. Immunity 17, 211–220 (2002)
Maloy, K. J. et al. Qualitative and quantitative requirements for CD4+ T cell-mediated antiviral protection. J. Immunol. 162, 2867–2874 (1999)
Mempel, T. R. et al. Regulatory T cells reversibly suppress cytotoxic T cell function independent of effector differentiation. Immunity 25, 129–141 (2006)
Whelan, S. P., Ball, L. A., Barr, J. N. & Wertz, G. T. Efficient recovery of infectious vesicular stomatitis virus entirely from cDNA clones. Proc. Natl Acad. Sci. USA 92, 8388–8392 (1995)
Van Rooijen, N. & Sanders, A. Liposome mediated depletion of macrophages: mechanism of action, preparation of liposomes and applications. J. Immunol. Methods 174, 83–93 (1994)
Iannacone, M. et al. Platelets mediate cytotoxic T lymphocyte-induced liver damage. Nature Med. 11, 1167–1169 (2005)
Shao, C., Liu, M., Wu, X. & Ding, F. Time-dependent expression of myostatin RNA transcript and protein in gastrocnemius muscle of mice after sciatic nerve resection. Microsurgery 27, 487–493 (2007)
Iannacone, M. et al. Platelets prevent IFN-α/β-induced lethal hemorrhage promoting CTL-dependent clearance of lymphocytic choriomeningitis virus. Proc. Natl Acad. Sci. USA 105, 629–634 (2008)
Acknowledgements
We thank G. Cheng and M. Flynn for technical support; J. Alton for secretarial assistance; D. Cureton for help and advice with VSV preparations; H. Leung for help with image quantification; R. M. Zinkernagel and H. Hengartner for providing tg7 mice; R. Bronson for help with reading neuropathology; S. Cohen for advice on nerve staining; N. van Rooijen for clodronate liposomes; and the members of the von Andrian laboratory for discussion. This work was supported by National Institutes of Health (NIH) grants AI069259, AI072252, AI078897 and AR42689 (to U.H.v.A.), the Giovanni Armenise-Harvard Foundation (to M.I.) and a NIH T32 Training Grant in Hematology (to E.A.M.).
Author information
Authors and Affiliations
Contributions
M.I., E.A.M. and U.H.v.A. designed the study. M.I., E.A.M., E.T., L.B. and T.J. performed experiments. M.I., E.A.M., E.T. and L.B. collected and analysed data. S.P.W. provided reagents and performed the RT–PCR experiment. S.E.H. contributed to the nerve imaging. L.G.G. provided mice and gave conceptual advice. M.I., E.A.M. and U.H.v.A. wrote the manuscript. M.I. and E.A.M contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-13 with legends and legends for Supplementary Movies 1-2. (PDF 1161 kb)
Supplementary Movie 1
This movie shows a three-dimensional rotation view followed by Z-stack projection of the MP-IVM stack used to generate Fig. 2e (see Supplementary Information file for full legend). (MOV 12971 kb)
Supplementary Movie 2
This movie shows a three-dimensional rotation view followed by Z-stack projection of the MP-IVM stack used to generate Fig. 2f (see Supplementary Information file for full legend). (MOV 8989 kb)
Rights and permissions
About this article
Cite this article
Iannacone, M., Moseman, E., Tonti, E. et al. Subcapsular sinus macrophages prevent CNS invasion on peripheral infection with a neurotropic virus. Nature 465, 1079–1083 (2010). https://doi.org/10.1038/nature09118
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09118
This article is cited by
-
Same yet different — how lymph node heterogeneity affects immune responses
Nature Reviews Immunology (2023)
-
Virus-based vaccine vectors with distinct replication mechanisms differentially infect and activate dendritic cells
npj Vaccines (2021)
-
Extracellular bacterial lymphatic metastasis drives Streptococcus pyogenes systemic infection
Nature Communications (2020)
-
Generation, localization and functions of macrophages during the development of testis
Nature Communications (2020)
-
Spatiotemporal regulation of type I interferon expression determines the antiviral polarization of CD4+ T cells
Nature Immunology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.