Abstract
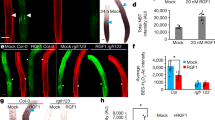
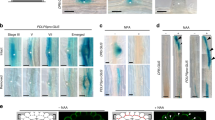
A key question in developmental biology is how cells exchange positional information for proper patterning during organ development. In plant roots the radial tissue organization is highly conserved with a central vascular cylinder in which two water conducting cell types, protoxylem and metaxylem, are patterned centripetally. We show that this patterning occurs through crosstalk between the vascular cylinder and the surrounding endodermis mediated by cell-to-cell movement of a transcription factor in one direction and microRNAs in the other. SHORT ROOT, produced in the vascular cylinder, moves into the endodermis to activate SCARECROW. Together these transcription factors activate MIR165a and MIR166b. Endodermally produced microRNA165/6 then acts to degrade its target mRNAs encoding class III homeodomain-leucine zipper transcription factors in the endodermis and stele periphery. The resulting differential distribution of target mRNA in the vascular cylinder determines xylem cell types in a dosage-dependent manner.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Du, T.-G., Schmid, M. & Jansen, R.-P. Why cells move messages: The biological functions of mRNA localization. Semin. Cell Dev. Biol. 18, 171–177 (2007)
Tretter, E. M., Alvarez, J. P., Eshed, Y. & Bowman, J. L. Activity range of Arabidopsis small RNAs derived from different biogenesis pathways. Plant Physiol. 147, 58–62 (2008)
Dunoyer, P., Himber, C., Ruiz-Ferrer, V., Alioua, A. & Voinnet, O. Intra- and intercellular RNA interference in Arabidopsis thaliana requires components of the microRNA and heterochromatic silencing pathways. Nature Genet. 39, 848–856 (2007)
Chitwood, D. H. et al. Pattern formation via small RNA mobility. Genes Dev. 23, 549–554 (2009)
Nogueira, F. T. S. et al. Regulation of small RNA accumulation in the maize shoot apex. PLoS Genet. 5, e1000320 (2009)
Juarez, M. T., Kui, J. S., Thomas, J., Heller, B. A. & Timmermans, M. C. P. microRNA-mediated repression of rolled leaf1 specifies maize leaf polarity. Nature 428, 84–88 (2004)
Lin, S.-I. et al. Regulatory network of microRNA399 and PHO2 by systemic signaling. Plant Physiol. 147, 732–746 (2008)
Esau, K. Anatomy of Seed Plants 2nd edn (Wiley, 1977)
Pryer, K. M., Schneider, H. & Magallón, S. in Assembling the Tree of Life (eds Cracraft, J. & Donoghue, M. J.) Ch. 10, 138–153 (Oxford Univ. Press, 2004)
Kevin Boyce, C., Holbrook, N. M. & Maciej, A. Z. in Vascular Transport in Plants 479–499 (Academic Press, 2005)
Gallagher, K. L., Paquette, A. J., Nakajima, K. & Benfey, P. N. Mechanisms regulating SHORT-ROOT intercellular movement. Curr. Biol. 14, 1847–1851 (2004)
Helariutta, Y. et al. The SHORT-ROOT gene controls radial patterning of the Arabidopsis root through radial signaling. Cell 101, 555–567 (2000)
Nakajima, K., Sena, G., Nawy, T. & Benfey, P. N. Intercellular movement of the putative transcription factor SHR in root patterning. Nature 413, 307–311 (2001)
Cui, H. et al. An evolutionarily conserved mechanism delimiting SHR movement defines a single layer of endodermis in plants. Science 316, 421–425 (2007)
Levesque, M. P. et al. Whole-genome analysis of the SHORT-ROOT developmental pathway in Arabidopsis . PLoS Biol. 4, e143 (2006)
Di Laurenzio, L. et al. The SCARECROW gene regulates an asymmetric cell division that is essential for generating the radial organization of the Arabidopsis root. Cell 86, 423–433 (1996)
Sabatini, S., Heidstra, R., Wildwater, M. & Scheres, B. SCARECROW is involved in positioning the stem cell niche in the Arabidopsis root meristem. Genes Dev. 17, 354–358 (2003)
Gallagher, K. L. & Benfey, P. N. Both the conserved GRAS domain and nuclear localization are required for SHORT-ROOT movement. Plant J. 57, 785–797 (2009)
Mähönen, A. P. et al. A novel two-component hybrid molecule regulates vascular morphogenesis of the Arabidopsis root. Genes Dev. 14, 2938–2943 (2000)
McConnell, J. R. et al. Role of PHABULOSA and PHAVOLUTA in determining radial patterning in shoots. Nature 411, 709–713 (2001)
Han, M.-H., Goud, S., Song, L. & Fedoroff, N. The Arabidopsis double-stranded RNA-binding protein HYL1 plays a role in microRNA-mediated gene regulation. Proc. Natl Acad. Sci. USA 101, 1093–1098 (2004)
Vazquez, F., Gasciolli, V., Cr, P. & Vaucheret, H. The nuclear dsRNA binding protein HYL1 is required for microRNA accumulation and plant development, but not posttranscriptional transgene silencing. Curr. Biol. 14, 346–351 (2004)
Emery, J. F. et al. Radial patterning of Arabidopsis shoots by class III HD-ZIP and KANADI genes. Curr. Biol. 13, 1768–1774 (2003)
Mallory, A. C. et al. MicroRNA control of PHABULOSA in leaf development: importance of pairing to the microRNA 5′ region. EMBO J. 23, 3356–3364 (2004)
Zhou, G.-K., Kubo, M., Zhong, R., Demura, T. & Ye, Z.-H. Overexpression of miR165 affects apical meristem formation, organ polarity establishment and vascular development in Arabidopsis . Plant Cell Physiol. 48, 391–404 (2007)
Hawker, N. P. & Bowman, J. L. Roles for class III HD-Zip and KANADI genes in Arabidopsis root development. Plant Physiol. 135, 2261–2270 (2004)
Lee, J.-Y. et al. Transcriptional and posttranscriptional regulation of transcription factor expression in Arabidopsis roots. Proc. Natl Acad. Sci. USA 103, 6055–6060 (2006)
Jung, J.-H. & Park, C.-M. MIR166/165 genes exhibit dynamic expression patterns in regulating shoot apical meristem and floral development in Arabidopsis . Planta 225, 1327–1338 (2007)
Parizotto, E. A., Dunoyer, P., Rahm, N., Himber, C. & Voinnet, O. In vivo investigation of the transcription, processing, endonucleolytic activity, and functional relevance of the spatial distribution of a plant miRNA. Genes Dev. 18, 2237–2242 (2004)
Stark, A., Brennecke, J., Bushati, N., Russell, R. B. & Cohen, S. M. Animal MicroRNAs confer robustness to gene expression and have a significant impact on 3′UTR evolution. Cell 123, 1133–1146 (2005)
Baima, S. et al. The Arabidopsis ATHB-8 HD-zip protein acts as a differentiation-promoting transcription factor of the vascular meristems. Plant Physiol. 126, 643–655 (2001)
Ohashi-Ito, K., Kubo, M., Demura, T. & Fukuda, H. Class III homeodomain leucine-zipper proteins regulate xylem cell differentiation. Plant Cell Physiol. 46, 1646–1656 (2005)
Prigge, M. J. et al. Class III homeodomain-leucine zipper gene family members have overlapping, antagonistic, and distinct roles in Arabidopsis development. Plant Cell 17, 61–76 (2005)
Muñiz, L. et al. ACAULIS5 controls Arabidopsis xylem specification through the prevention of premature cell death. Development 135, 2573–2582 (2008)
Mähönen, A. P. et al. Cytokinin signaling and its inhibitor AHP6 regulate cell fate during vascular development. Science 311, 94–98 (2006)
Floyd, S. K. & Bowman, J. The ancestral developmental tool kit of land plants. Int. J. Plant Sci. 168, 1–35 (2007)
Floyd, S. K. & Bowman, J. L. Gene regulation: ancient microRNA target sequences in plants. Nature 428, 485–486 (2004)
Levine, E., McHale, P. & Levine, H. Small regulatory RNAs may sharpen spatial expression patterns. PLoS Comp. Biol. 3, e233 (2007)
Shi, R. & Chiang, V. L. Facile means for quantifying microRNA expression by real-time PCR. Biotechniques 39, 519–525 (2005)
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana . Plant J. 16, 735–743 (1998)
Acknowledgements
We thank K. Kainulainen, M. Herpola, G.-B. Berglund and J. Jung for technical assistance; M. Prigge, S. Clark, C. Bellini, N. Sauer, J. Colinas, T. Vernoux, K. Gallagher, A. P. Mahonen, A. Bishopp, M. Bonke, N. Fedoroff, J. C. Fletcher, B. J. Reinhart, I. Pekker, ABRC and NASC for materials, and E. Richards and M. Harrison for comments on the manuscript; M. Tsiantis for sharing results before publication. This work was supported by the Boyce Thompson Institute and NSF IOS0818071 to J.-Y.L., Cornell Presidential Life Science Fellowship to J.Z., grants from the NIH (RO1-GM043778) and from the NSF ARABIDOPSIS 2010 programme to P.N.B., a fellowship from the MICINN, Spanish Government to M.A.M.-R., grants by the Academy of Finland, Tekes and ESF to Y.H., S.L. and A.V., European Molecular Biology Organisation (EMBO, ALTF 450-2007) to J.D., Estonian funding agencies (ETF7361 and SF0180071s07) to O.L., Nilsson-Ehle Foundation to C.J.R, and FORMAS and Carl Trygger’s Foundation for Scientific Research to A.C. Imaging at Boyce Thompson Institute was supported by NSF (NSF DBI-0618969) and Triad Foundation.
Author information
Authors and Affiliations
Contributions
A.C. and J.-Y.L. contributed equally to this work, C.J.R., J.D., S.L. and J.Z. contributed equally to this work, O.L., M.A.M.-R. and A.V. contributed equally to this work, Y.H. and P.N.B. contributed equally to this work, and the name order was determined by raffle. A.C. designed and performed experiments to characterize HD-ZIP III transcription factors and miR165/6 in vascular patterning, J.-Y.L. identified and characterized the regulatory network of SHR, SCR and miR165/6 in the xylem patterning, C.J.R. analysed the miRNA expression by in situ hybridization and participated in HD-ZIP III mutant characterization. J.D. participated in the analysis of expression of PHB (including mutant forms) and other HD-ZIP III and generated pCRE1::MIR165a as well as participated in the generation of J0571 lines to rescue scr and shr. S.L. participated in the PHB and HD-ZIP III expression analysis, phb-7d mutant characterization and generated the pSCR::MIR165a to rescue scr. J.Z. developed and characterized the xylem patterning led by non-mobile SHR and PHB-m. O.L. participated in the characterization of various HD-ZIP III mutant lines. M.A.M.-R. showed the non-cell autonomous action of non-mobile SHR. A.V. participated in positional cloning of phb-7d and establishment of the J0571 lines to rescue shr. S.T. identified the phb-7d mutant. A.C. identified the scr-6 allele. J.S. characterized shr/scr and HD-ZIP III double mutants and embryo expression patterns in GFP lines. J.L.B. shared informative non-published materials. Y.H. and P.N.B. participated in experimental design. A.C., J.-Y.L., Y.H. and P.N.B. wrote the manuscript. A.C. and J.-Y.L. are co-corresponding authors. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-20 with legends, Supplementary Tables 1-2, Supplementary Methods and References. (PDF 2022 kb)
Rights and permissions
About this article
Cite this article
Carlsbecker, A., Lee, JY., Roberts, C. et al. Cell signalling by microRNA165/6 directs gene dose-dependent root cell fate. Nature 465, 316–321 (2010). https://doi.org/10.1038/nature08977
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08977
This article is cited by
-
ARGONAUTE10 controls cell fate specification and formative cell divisions in the Arabidopsis root
The EMBO Journal (2024)
-
A diffusible small-RNA-based Turing system dynamically coordinates organ polarity
Nature Plants (2024)
-
An AGO10:miR165/6 module regulates meristem activity and xylem development in the Arabidopsis root
The EMBO Journal (2024)
-
GhHB14_D10 and GhREV_D5, two HD-ZIP III transcription factors, play a regulatory role in cotton fiber secondary cell wall biosynthesis
Plant Cell Reports (2024)
-
Global gene regulatory network underlying miR165a in Arabidopsis shoot apical meristem
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.