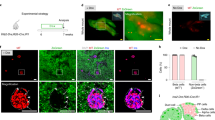
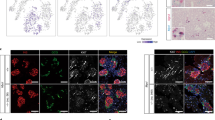
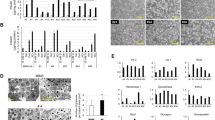
Abstract
Pancreatic insulin-producing β-cells have a long lifespan, such that in healthy conditions they replicate little during a lifetime. Nevertheless, they show increased self-duplication after increased metabolic demand or after injury (that is, β-cell loss). It is not known whether adult mammals can differentiate (regenerate) new β-cells after extreme, total β-cell loss, as in diabetes. This would indicate differentiation from precursors or another heterologous (non-β-cell) source. Here we show β-cell regeneration in a transgenic model of diphtheria-toxin-induced acute selective near-total β-cell ablation. If given insulin, the mice survived and showed β-cell mass augmentation with time. Lineage-tracing to label the glucagon-producing α-cells before β-cell ablation tracked large fractions of regenerated β-cells as deriving from α-cells, revealing a previously disregarded degree of pancreatic cell plasticity. Such inter-endocrine spontaneous adult cell conversion could be harnessed towards methods of producing β-cells for diabetes therapies, either in differentiation settings in vitro or in induced regeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zhou, Q. & Melton, D. A. Extreme makeover: converting one cell into another. Cell Stem Cell 3, 382–388 (2008)
Uhlenhaut, N. H. et al. Somatic sex reprogramming of adult ovaries to testes by FOXL2 ablation. Cell 139, 1130–1142 (2009)
Zhou, Q., Brown, J., Kanarek, A., Rajagopal, J. & Melton, D. A. In vivo reprogramming of adult pancreatic exocrine cells to β-cells. Nature 455, 627–632 (2008)
Bonal, C. et al. Pancreatic inactivation of c-Myc decreases acinar mass and transdifferentiates acinar cells into adipocytes in mice. Gastroenterology 136, 309–319 (2009)
Teta, M., Long, S. Y., Wartschow, L. M., Rankin, M. M. & Kushner, J. A. Very slow turnover of β-cells in aged adult mice. Diabetes 54, 2557–2567 (2005)
Desgraz, R. & Herrera, P. L. Pancreatic neurogenin 3-expressing cells are unipotent islet precursors. Development 136, 3567–3574 (2009)
Dor, Y., Brown, J., Martinez, O. I. & Melton, D. A. Adult pancreatic β-cells are formed by self-duplication rather than stem-cell differentiation. Nature 429, 41–46 (2004)
Rankin, M. M. & Kushner, J. A. Adaptive β-cell proliferation is severely restricted with advanced age. Diabetes 58, 1365–1372 (2009)
Matveyenko, A. V. & Butler, P. C. Relationship between β-cell mass and diabetes onset. Diabetes Obes. Metab. 10 (suppl. 4). 23–31 (2008)
Wang, R. N., Bouwens, L. & Kloppel, G. β-cell growth in adolescent and adult rats treated with streptozotocin during the neonatal period. Diabetologia 39, 548–557 (1996)
Herold, K. C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med. 346, 1692–1698 (2002)
Thyssen, S., Arany, E. & Hill, D. J. Ontogeny of regeneration of beta-cells in the neonatal rat after treatment with streptozotocin. Endocrinology 147, 2346–2356 (2006)
Bonner-Weir, S., Baxter, L. A., Schuppin, G. T. & Smith, F. E. A second pathway for regeneration of adult exocrine and endocrine pancreas. A possible recapitulation of embryonic development. Diabetes 42, 1715–1720 (1993)
Wang, R. N., Kloppel, G. & Bouwens, L. Duct- to islet-cell differentiation and islet growth in the pancreas of duct-ligated adult rats. Diabetologia 38, 1405–1411 (1995)
Xu, G., Stoffers, D. A., Habener, J. F. & Bonner-Weir, S. Exendin-4 stimulates both β-cell replication and neogenesis, resulting in increased β-cell mass and improved glucose tolerance in diabetic rats. Diabetes 48, 2270–2276 (1999)
Nir, T., Melton, D. A. & Dor, Y. Recovery from diabetes in mice by beta cell regeneration. J. Clin. Invest. 117, 2553–2561 (2007)
Baeyens, L. et al. In vitro generation of insulin-producing β cells from adult exocrine pancreatic cells. Diabetologia 48, 49–57 (2005)
Bonner-Weir, S. & Weir, G. C. New sources of pancreatic β-cells. Nature Biotechnol. 23, 857–861 (2005)
Trucco, M. Regeneration of the pancreatic β cell. J. Clin. Invest. 115, 5–12 (2005)
Xu, X. et al. β cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell 132, 197–207 (2008)
Herrera, P. L. Adult insulin- and glucagon-producing cells differentiate from two independent cell lineages. Development 127, 2317–2322 (2000)
Herrera, P. L., Nepote, V. & Delacour, A. Pancreatic cell lineage analyses in mice. Endocrine 19, 267–278 (2002)
Herrera, P. L. et al. Ablation of islet endocrine cells by targeted expression of hormone- promoter-driven toxigenes. Proc. Natl Acad. Sci. USA 91, 12999–13003 (1994)
Naglich, J. G., Metherall, J. E., Russell, D. W. & Eidels, L. Expression cloning of a diphtheria toxin receptor: identity with a heparin-binding EGF-like growth factor precursor. Cell 69, 1051–1061 (1992)
Saito, M. et al. Diphtheria toxin receptor-mediated conditional and targeted cell ablation in transgenic mice. Nature Biotechnol. 19, 746–750 (2001)
Srinivas, S. et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 1, 4 (2001)
Cano, D. A. et al. Regulated β-cell regeneration in the adult mouse pancreas. Diabetes 57, 958–966 (2008)
Wang, Z. V. et al. PANIC-ATTAC: a mouse model for inducible and reversible β-cell ablation. Diabetes 57, 2137–2148 (2008)
Gerich, J. E., Lorenzi, M., Karam, J. H., Schneider, V. & Forsham, P. H. Abnormal pancreatic glucagon secretion and postprandial hyperglycemia in diabetes mellitus. J. Am. Med. Assoc. 234, 159–165 (1975)
Perl, A. K., Wert, S. E., Nagy, A., Lobe, C. G. & Whitsett, J. A. Early restriction of peripheral and proximal cell lineages during formation of the lung. Proc. Natl Acad. Sci. USA 99, 10482–10487 (2002)
Quoix, N. et al. The GluCre-ROSA26EYFP mouse: a new model for easy identification of living pancreatic α-cells. FEBS Lett. 581, 4235–4240 (2007)
Li, W. C., Horb, M. E., Tosh, D. & Slack, J. M. In vitro transdifferentiation of hepatoma cells into functional pancreatic cells. Mech. Dev. 122, 835–847 (2005)
Fodor, A. et al. Adult rat liver cells transdifferentiated with lentiviral IPF1 vectors reverse diabetes in mice: an ex vivo gene therapy approach. Diabetologia 50, 121–130 (2007)
Chakrabarti, S. K., James, J. C. & Mirmira, R. G. Quantitative assessment of gene targeting in vitro and in vivo by the pancreatic transcription factor, Pdx1. Importance of chromatin structure in directing promoter binding. J. Biol. Chem. 277, 13286–13293 (2002)
Ritz-Laser, B. et al. Ectopic expression of the β-cell specific transcription factor Pdx1 inhibits glucagon gene transcription. Diabetologia 46, 810–821 (2003)
Schisler, J. C. et al. The Nkx6.1 homeodomain transcription factor suppresses glucagon expression and regulates glucose-stimulated insulin secretion in islet β cells. Proc. Natl Acad. Sci. USA 102, 7297–7302 (2005)
Collombat, P. et al. Opposing actions of Arx and Pax4 in endocrine pancreas development. Genes Dev. 17, 2591–2603 (2003)
Collombat, P. et al. The ectopic expression of Pax4 in the mouse pancreas converts progenitor cells into α and subsequently β cells. Cell 138, 449–462 (2009)
Bonal, C. & Herrera, P. L. Genes controlling pancreas ontogeny. Int. J. Dev. Biol. 52, 823–835 (2008)
Rorsman, P., Salehi, S. A., Abdulkader, F., Braun, M. & MacDonald, P. E. KATP-channels and glucose-regulated glucagon secretion. Trends Endocrinol. Metab. 19, 277–284 (2008)
Heimberg, H., De Vos, A., Pipeleers, D., Thorens, B. & Schuit, F. Differences in glucose transporter gene expression between rat pancreatic α- and β-cells are correlated to differences in glucose transport but not in glucose utilization. J. Biol. Chem. 270, 8971–8975 (1995)
Heimberg, H. et al. The glucose sensor protein glucokinase is expressed in glucagon-producing α-cells. Proc. Natl Acad. Sci. USA 93, 7036–7041 (1996)
Burgoyne, R. D. & Morgan, A. Secretory granule exocytosis. Physiol. Rev. 83, 581–632 (2003)
Fausto, N. Liver regeneration and repair: hepatocytes, progenitor cells, and stem cells. Hepatology 39, 1477–1487 (2004)
Meier, J. J., Bhushan, A., Butler, A. E., Rizza, R. A. & Butler, P. C. Sustained beta cell apoptosis in patients with long-standing type 1 diabetes: indirect evidence for islet regeneration? Diabetologia 48, 2221–2228 (2005)
Meier, J. J. et al. Direct evidence of attempted beta cell regeneration in an 89-year-old patient with recent-onset type 1 diabetes. Diabetologia 49, 1838–1844 (2006)
Karges, B. et al. Complete long-term recovery of β-cell function in autoimmune type 1 diabetes after insulin treatment. Diabetes Care 27, 1207–1208 (2004)
Karges, B. et al. Immunological mechanisms associated with long-term remission of human type 1 diabetes. Diabetes Metab. Res. Rev. 22, 184–189 (2006)
Hogan, B., Beddington, R., Costantini, F. & Lacy, E. Manipulating the Mouse Embryo. A Laboratory Manual 2nd edn, Ch. 7 289–358 (Cold Spring Harbor Laboratory Press, 1994)
Nguyen, V. X., Nix, D. E., Gillikin, S. & Schentag, J. J. Effect of oral antacid administration on the pharmacokinetics of intravenous doxycycline. Antimicrob. Agents Chemother. 33, 434–436 (1989)
Acknowledgements
We are grateful to P. Vassalli and C. V. E. Wright for their comments and support. We also thank R. Stein, S. Kim and A. Ruiz i Altaba for discussions, and G. Flores, C. Gysler, O. Fazio, G. Philippin and C. Vesin for their technical help. We thank D. Melton for the RIP-CreER transgenic strain, and B. Thorens and C. V. E. Wright for anti-Glut2 and anti-Pdx1 antibodies, respectively. Work was funded with grants from the NIH/NIDDK (‘Beta Cell Biology Consortium’), JDRF (Juvenile Diabetes Research Foundation), Swiss National Science Foundation (and the NCCR ‘Frontiers in Genetics’), and 6FP EU Integrated Project ‘Beta Cell Therapy for Diabetes’ to P.L.H.
Author Contributions F.T., V.N. and I.A. contributed equally to this work. F.T. and V.N. prepared constructs for generating the transgenics, wrote the manuscript and together with I.A. performed most experiments and analyses. K.K. provided one plasmid. R.D. and S.C. conducted gene expression analyses, and S.C. performed immunofluorescence microscopy. P.L.H. conceived the experiments and wrote the manuscript with contributions from F.T., V.N., I.A. and S.C.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S12 with legends, Supplementary Methods and Supplementary References. (PDF 2623 kb)
Rights and permissions
About this article
Cite this article
Thorel, F., Népote, V., Avril, I. et al. Conversion of adult pancreatic α-cells to β-cells after extreme β-cell loss. Nature 464, 1149–1154 (2010). https://doi.org/10.1038/nature08894
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08894
This article is cited by
-
Genetic lineage tracing identifies adaptive mechanisms of pancreatic islet β cells in various mouse models of diabetes with distinct age of initiation
Science China Life Sciences (2024)
-
Novel time-resolved reporter mouse reveals spatial and transcriptional heterogeneity during alpha cell differentiation
Diabetologia (2024)
-
CDK8/19 inhibition plays an important role in pancreatic β-cell induction from human iPSCs
Stem Cell Research & Therapy (2023)
-
Reversing pancreatic β-cell dedifferentiation in the treatment of type 2 diabetes
Experimental & Molecular Medicine (2023)
-
Glucagon receptor antagonists might stimulate β-cell expansion
Nature Reviews Endocrinology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.