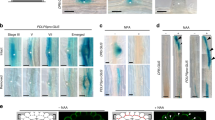
Abstract
Acquisition of cell identity in plants relies strongly on positional information1, hence cell–cell communication and inductive signalling are instrumental for developmental patterning. During Arabidopsis embryogenesis, an extra-embryonic cell is specified to become the founder cell of the primary root meristem, hypophysis, in response to signals from adjacent embryonic cells2. The auxin-dependent transcription factor MONOPTEROS (MP) drives hypophysis specification by promoting transport of the hormone auxin from the embryo to the hypophysis precursor. However, auxin accumulation is not sufficient for hypophysis specification, indicating that additional MP-dependent signals are required3. Here we describe the microarray-based isolation of MP target genes that mediate signalling from embryo to hypophysis. Of three direct transcriptional target genes, TARGET OF MP 5 (TMO5) and TMO7 encode basic helix–loop–helix (bHLH) transcription factors that are expressed in the hypophysis-adjacent embryo cells, and are required and partially sufficient for MP-dependent root initiation. Importantly, the small TMO7 transcription factor moves from its site of synthesis in the embryo to the hypophysis precursor, thus representing a novel MP-dependent intercellular signal in embryonic root specification.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Scheres, B. Plant cell identity. The role of position and lineage. Plant Physiol. 125, 112–114 (2001)
Hamann, T., Mayer, U. & Jürgens, G. The auxin-insensitive bodenlos mutation affects primary root formation and apical-basal patterning in the Arabidopsis embryo. Development 126, 1387–1395 (1999)
Weijers, D. et al. Auxin triggers transient local signaling for cell specification in Arabidopsis embryogenesis. Dev. Cell 10, 265–270 (2006)
Weigel, D. & Jürgens, G. Stem cells that make stems. Nature 415, 751–754 (2002)
Hardtke, C. S. & Berleth, T. The Arabidopsis gene MONOPTEROS encodes a transcription factor mediating embryo axis formation and vascular development. EMBO J. 17, 1405–1411 (1998)
Ulmasov, T., Hagen, G. & Guilfoyle, T. J. Activation and repression of transcription by auxin-response factors. Proc. Natl Acad. Sci. USA 96, 5844–5849 (1999)
Hamann, T., Benkova, E., Bäurle, I., Kientz, M. & Jürgens, G. The Arabidopsis BODENLOS gene encodes an auxin response protein inhibiting MONOPTEROS-mediated embryo patterning. Genes Dev. 16, 1610–1615 (2002)
Dharmasiri, N. et al. Plant development is regulated by a family of auxin receptor F box proteins. Dev. Cell 9, 109–119 (2005)
Weijers, D. et al. Developmental specificity of auxin response by pairs of ARF and Aux/IAA transcriptional regulators. EMBO J. 24, 1874–1885 (2005)
Tian, Q., Uhlir, N. J. & Reed, J. W. Arabidopsis SHY2/IAA3 inhibits auxin-regulated gene expression. Plant Cell 14, 301–319 (2002)
Okushima, Y. et al. Functional genomic analysis of the AUXIN RESPONSE FACTOR gene family members in Arabidopsis thaliana: unique and overlapping functions of ARF7 and ARF19. Plant Cell 17, 444–463 (2005)
Riechmann, J. L. et al. Arabidopsis transcription factors: genome-wide comparative analysis among eukaryotes. Science 290, 2105–2110 (2000)
Okushima, Y., Fukaki, H., Onoda, M., Theologis, A. & Tasaka, M. ARF7 and ARF19 regulate lateral root formation via direct activation of LBD/ASL genes in Arabidopsis . Plant Cell 19, 118–130 (2007)
Donner, T. J., Sherr, I. & Scarpella, E. Regulation of preprocambial cell state acquisition by auxin signalling in Arabidopsis leaves. Development 136, 3235–3246 (2009)
Galinha, C. et al. PLETHORA proteins as dose-dependent master regulators of Arabidopsis root development. Nature 449, 1053–1057 (2007)
Rashotte, A. M. et al. A subset of Arabidopsis AP2 transcription factors mediates cytokinin responses in concert with a two-component pathway. Proc. Natl Acad. Sci. USA 103, 11081–11085 (2006)
Lee, S. et al. Overexpression of PRE1 and its homologous genes activates gibberellin-dependent responses in Arabidopsis thaliana . Plant Cell Physiol. 47, 591–600 (2006)
Wang, H. et al. Regulation of Arabidopsis brassinosteroid signaling by atypical basic helix-loop-helix proteins. Plant Cell 21, 3781–3791 (2009)
Ulmasov, T., Hagen, G. & Guilfoyle, T. J. Dimerization and DNA binding of auxin response factors. Plant J. 19, 309–319 (1999)
Cole, M. et al. DORNRÖSSCHEN is a direct target of the auxin response factor MONOPTEROS in the Arabidopsis embryo. Development 136, 1643–1651 (2009)
Toledo-Ortiz, G., Huq, E. & Quail, P. H. The Arabidopsis basic/helix-loop-helix transcription factor family. Plant Cell 15, 1749–1770 (2003)
Nakajima, K., Sena, G., Nawy, T. & Benfey, P. N. Intercellular movement of the putative transcription factor SHR in root patterning. Nature 413, 307–311 (2001)
Massari, M. E. & Murre, C. Helix-loop-helix proteins: regulators of transcription in eucaryotic organisms. Mol. Cell. Biol. 20, 429–440 (2000)
Takada, S. & Jürgens, G. Transcriptional regulation of epidermal cell fate in the Arabidopsis embryo. Development 134, 1141–1150 (2007)
Wesley, S. V. et al. Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J. 27, 581–590 (2001)
Weijers, D. et al. An Arabidopsis Minute-like phenotype caused by a semi-dominant mutation in a RIBOSOMAL PROTEIN S5 gene. Development 128, 4289–4299 (2001)
Schwab, R., Ossowski, S., Riester, M., Warthmann, N. & Weigel, D. Highly specific gene silencing by artificial microRNAs in Arabidopsis . Plant Cell 18, 1121–1133 (2006)
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana . Plant J. 16, 735–743 (1998)
van den Bosch, H. M. et al. Gene expression of transporters and phase I/II metabolic enzymes in murine small intestine during fasting. BMC Genomics 8, 267 (2007)
Wu, Z., Irizarry, R. A., Gentleman, R., Martinez-Murillo, F. & Spencer, F. A model-based background adjustment for oligonucleotide expression arrays. J. Am. Stat. Assoc. 99, 909–917 (2004)
Dai, M. et al. Evolving gene/transcript definitions significantly alter the interpretation of GeneChip data. Nucleic Acids Res. 33, e175 (2005)
Smyth, G. K. Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat. Appl. Genet. Mol. Biol. 3, Article–3 (2004)
Sartor, M. A. et al. Intensity-based hierarchical Bayes method improves testing for differentially expressed genes in microarray experiments. BMC Bioinformatics 7, 538 (2006)
Leibfried, A. et al. WUSCHEL controls meristem function by direct regulation of cytokinin-inducible response regulators. Nature 438, 1172–1175 (2005)
Aker, J., Borst, J. W., Karlova, R. & de Vries, S. C. The Arabidopsis AAA protein CDC48A interacts in vivo with the somatic embryogenesis receptor-like kinase 1 receptor at the plasma membrane. J. Struct. Biol. 156, 62–71 (2006)
Lauber, M. H. et al. The Arabidopsis KNOLLE protein is a cytokinesis-specific syntaxin. J. Cell Biol. 139, 1485–1493 (1997)
Acknowledgements
We acknowledge P. van Oorschot, A. van Haperen and S. Heilbronner for technical assistance, C. Arsene for plant care, S. de Folter and O. Mathieu for advice on ChIP, M. Wunderlich and G. Bijl for help with qPCR, G. Hooiveld for help with statistical analysis of the microarray data and the Nottingham Arabidopsis Stock Centre for mutant and transgenic seeds. We thank P. Maier, S. de Vries, A. Koltunow, A. Lokerse, S. Saiga, V. Willemsen and M. Tsiantis for discussions and comments on the manuscript. This work was supported by grants from the Netherlands Organization for Scientific Research (NWO; VIDI 864.06.012 to D.W.), from the Deutsche Forschungsgemeinschaft (DFG; SFB446 to G.J.) and the Netherlands Proteomics Centre (NPC; D.W., grant awarded to S. de Vries).
Author Contributions A.S. established conditions for the microarray experiment, which was performed together with M.S., validated the array data by qPCR, generated most transgenic lines for expression analysis and functional studies of TMO genes, which was done together with B.M. M.K. performed in situ hybridizations, J.F. and D.W. performed ChIP, and W.L. generated and analysed TMO7 downregulation and TMO7-LIKE1 reporter lines. B.M. performed MP overexpression, and suspensor-specific TMO7 expression experiments, and generated pMP–GFP lines. E.H.R. identified the suspensor-specific At1g34170 gene, and generated pSUSP-GFP lines. D.W. and G.J. conceived and supervised the study. D.W. wrote the paper with input from G.J., A.S. and B.M.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S7 with legends, and Supplementary Tables S1-S3. (PDF 9058 kb)
Rights and permissions
About this article
Cite this article
Schlereth, A., Möller, B., Liu, W. et al. MONOPTEROS controls embryonic root initiation by regulating a mobile transcription factor. Nature 464, 913–916 (2010). https://doi.org/10.1038/nature08836
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08836
This article is cited by
-
ARGONAUTE10 controls cell fate specification and formative cell divisions in the Arabidopsis root
The EMBO Journal (2024)
-
Lateral Root versus Nodule: The Auxin-Cytokinin Interplay
Journal of Plant Growth Regulation (2023)
-
Abiotic factors and endophytes co-regulate flavone and terpenoid glycoside metabolism in Glycyrrhiza uralensis
Applied Microbiology and Biotechnology (2023)
-
Overexpression of SlPRE3 alters the plant morphologies in Solanum lycopersicum
Plant Cell Reports (2023)
-
Specification of female germline by microRNA orchestrated auxin signaling in Arabidopsis
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.