Abstract
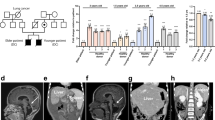
Patients with dyskeratosis congenita (DC), a disorder of telomere maintenance, suffer degeneration of multiple tissues1,2,3. Patient-specific induced pluripotent stem (iPS) cells4 represent invaluable in vitro models for human degenerative disorders like DC. A cardinal feature of iPS cells is acquisition of indefinite self-renewal capacity, which is accompanied by induction of the telomerase reverse transcriptase gene (TERT)5,6,7. We investigated whether defects in telomerase function would limit derivation and maintenance of iPS cells from patients with DC. Here we show that reprogrammed DC cells overcome a critical limitation in telomerase RNA component (TERC) levels to restore telomere maintenance and self-renewal. We discovered that TERC upregulation is a feature of the pluripotent state, that several telomerase components are targeted by pluripotency-associated transcription factors, and that in autosomal dominant DC, transcriptional silencing accompanies a 3′ deletion at the TERC locus. Our results demonstrate that reprogramming restores telomere elongation in DC cells despite genetic lesions affecting telomerase, and show that strategies to increase TERC expression may be therapeutically beneficial in DC patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kirwan, M. & Dokal, I. Dyskeratosis congenita: a genetic disorder of many faces. Clin. Genet. 73, 103–112 (2008)
Mason, P. J., Wilson, D. B. & Bessler, M. Dyskeratosis congenita–a disease of dysfunctional telomere maintenance. Curr. Mol. Med. 5, 159–170 (2005)
Calado, R. T. & Young, N. S. Telomere maintenance and human bone marrow failure. Blood 111, 4446–4455 (2008)
Park, I. H. et al. Disease-specific induced pluripotent stem cells. Cell 134, 877–886 (2008)
Park, I. H. et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature 451, 141–146 (2008)
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007)
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007)
Marion, R. M. et al. Telomeres acquire embryonic stem cell characteristics in induced pluripotent stem cells. Cell Stem Cell 4, 141–154 (2009)
Heiss, N. S. et al. X-linked dyskeratosis congenita is caused by mutations in a highly conserved gene with putative nucleolar functions. Nature Genet. 19, 32–38 (1998)
Mitchell, J. R., Wood, E. & Collins, K. A telomerase component is defective in the human disease dyskeratosis congenita. Nature 402, 551–555 (1999)
Wong, J. M. & Collins, K. Telomerase RNA level limits telomere maintenance in X-linked dyskeratosis congenita. Genes Dev. 20, 2848–2858 (2006)
Cawthon, R. M. Telomere measurement by quantitative PCR. Nucleic Acids Res. 30, e47 (2002)
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998)
Vaziri, H. & Benchimol, S. Reconstitution of telomerase activity in normal human cells leads to elongation of telomeres and extended replicative life span. Curr. Biol. 8, 279–282 (1998)
Vulliamy, T. et al. The RNA component of telomerase is mutated in autosomal dominant dyskeratosis congenita. Nature 413, 432–435 (2001)
Westin, E. R. et al. Telomere restoration and extension of proliferative lifespan in dyskeratosis congenita fibroblasts. Aging Cell 6, 383–394 (2007)
Cairney, C. J. & Keith, W. N. Telomerase redefined: integrated regulation of hTR and hTERT for telomere maintenance and telomerase activity. Biochimie 90, 13–23 (2008)
Yi, X., Tesmer, V. M., Savre-Train, I., Shay, J. W. & Wright, W. E. Both transcriptional and posttranscriptional mechanisms regulate human telomerase template RNA levels. Mol. Cell. Biol. 19, 3989–3997 (1999)
Montanaro, L. et al. Dyskerin expression influences the level of ribosomal RNA pseudo-uridylation and telomerase RNA component in human breast cancer. J. Pathol. 210, 10–18 (2006)
Fu, D. & Collins, K. Distinct biogenesis pathways for human telomerase RNA and H/ACA small nucleolar RNAs. Mol. Cell 11, 1361–1372 (2003)
Marrone, A., Stevens, D., Vulliamy, T., Dokal, I. & Mason, P. J. Heterozygous telomerase RNA mutations found in dyskeratosis congenita and aplastic anemia reduce telomerase activity via haploinsufficiency. Blood 104, 3936–3942 (2004)
Trahan, C. & Dragon, F. Dyskeratosis congenita mutations in the H/ACA domain of human telomerase RNA affect its assembly into a pre-RNP. RNA 15, 235–243 (2009)
Broccoli, D., Young, J. W. & de Lange, T. Telomerase activity in normal and malignant hematopoietic cells. Proc. Natl Acad. Sci. USA 92, 9082–9086 (1995)
Chiu, C. P. et al. Differential expression of telomerase activity in hematopoietic progenitors from adult human bone marrow. Stem Cells 14, 239–248 (1996)
Hiyama, K. et al. Activation of telomerase in human lymphocytes and hematopoietic progenitor cells. J. Immunol. 155, 3711–3715 (1995)
Allsopp, R. C., Morin, G. B., DePinho, R., Harley, C. B. & Weissman, I. L. Telomerase is required to slow telomere shortening and extend replicative lifespan of HSCs during serial transplantation. Blood 102, 517–520 (2003)
Kirwan, M. et al. Exogenous TERC alone can enhance proliferative potential, telomerase activity and telomere length in lymphocytes from dyskeratosis congenita patients. Br. J. Haematol. 144, 771–781 (2009)
Liu, L. et al. Telomere lengthening early in development. Nature Cell Biol. 9, 1436–1441 (2007)
Loh, Y. H. et al. The Oct4 and Nanog transcription network regulates pluripotency in mouse embryonic stem cells. Nature Genet. 38, 431–440 (2006)
Agarwal, S. & Rao, A. Modulation of chromatin structure regulates cytokine gene expression during T cell differentiation. Immunity 9, 765–775 (1998)
Park, I. H., Lerou, P. H., Zhao, R., Huo, H. & Daley, G. Q. Generation of human-induced pluripotent stem cells. Nature Protocols 3, 1180–1186 (2008)
Bryan, T. M., Englezou, A., Gupta, J., Bacchetti, S. & Reddel, R. R. Telomere elongation in immortal human cells without detectable telomerase activity. EMBO J. 14, 4240–4248 (1995)
Yam, P. Y. et al. Design of HIV vectors for efficient gene delivery into human hematopoietic cells. Mol. Ther. 5, 479–484 (2002)
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990)
Atkinson, S. P., Hoare, S. F., Glasspool, R. M. & Keith, W. N. Lack of telomerase gene expression in alternative lengthening of telomere cells is associated with chromatin remodeling of the hTR and hTERT gene promoters. Cancer Res. 65, 7585–7590 (2005)
Li, S. et al. Rapid inhibition of cancer cell growth induced by lentiviral delivery and expression of mutant-template telomerase RNA and anti-telomerase short-interfering RNA. Cancer Res. 64, 4833–4840 (2004)
Watanabe, K. et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nature Biotechnol. 25, 681–686 (2007)
Greenberg, M. E. & Bender, T. P. in Current Protocols in Molecular Biology Ch. 4, Unit 4.10 (Wiley, 2007)
Acknowledgements
This work was funded by grants from the National Institutes of Health (NIH) and the Manton Center for Orphan Disease Research (G.Q.D.); NIH K08HL089150, Amy Clare Potter Fellowship and Manton Center for Orphan Disease Research (S.A.); the Agency of Science, Technology and Research and the Institute of Medical Biology, Singapore (Y.-H.L.); NIH R01AG0227388 (F.D.G. and A.J.K.); and the James and Esther King Biomedical Research Program and MOST 973 project (2009CB941000) (D.L.K and L.L).
Author Contributions S.A. performed project planning, experimental work, data interpretation and preparation of the manuscript. Y.-H.L., I.-H.P., J.H., E.M.M., J.D.M., R.M.R., M.O., H.H. and S.L. performed experimental work. H.-H.N., F.D.G., D.L.K., A.J.K., L.L. and G.Q.D. participated in project planning, data interpretation and preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
George Q. Daley is a member of the scientific advisory boards of MPM Capital, Epizyme, Inc., and iPierian, Inc.
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S14 with Legends, Supplementary Table 1, a Supplementary Note and Supplementary References. (PDF 3197 kb)
Rights and permissions
About this article
Cite this article
Agarwal, S., Loh, YH., McLoughlin, E. et al. Telomere elongation in induced pluripotent stem cells from dyskeratosis congenita patients. Nature 464, 292–296 (2010). https://doi.org/10.1038/nature08792
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08792
This article is cited by
-
Thymidine nucleotide metabolism controls human telomere length
Nature Genetics (2023)
-
New insights into the epitranscriptomic control of pluripotent stem cell fate
Experimental & Molecular Medicine (2022)
-
Three-dimensional modeling of human neurodegeneration: brain organoids coming of age
Molecular Psychiatry (2020)
-
The role of telomere-binding modulators in pluripotent stem cells
Protein & Cell (2020)
-
Recent technological advancements in stem cell research for targeted therapeutics
Drug Delivery and Translational Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.