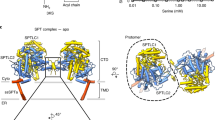
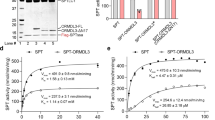
Abstract
Despite the essential roles of sphingolipids both as structural components of membranes and critical signalling molecules, we have a limited understanding of how cells sense and regulate their levels. Here we reveal the function in sphingolipid metabolism of the ORM genes (known as ORMDL genes in humans)—a conserved gene family that includes ORMDL3, which has recently been identified as a potential risk factor for childhood asthma. Starting from an unbiased functional genomic approach in Saccharomyces cerevisiae, we identify Orm proteins as negative regulators of sphingolipid synthesis that form a conserved complex with serine palmitoyltransferase, the first and rate-limiting enzyme in sphingolipid production. We also define a regulatory pathway in which phosphorylation of Orm proteins relieves their inhibitory activity when sphingolipid production is disrupted. Changes in ORM gene expression or mutations to their phosphorylation sites cause dysregulation of sphingolipid metabolism. Our work identifies the Orm proteins as critical mediators of sphingolipid homeostasis and raises the possibility that sphingolipid misregulation contributes to the development of childhood asthma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Brown, M. S. & Goldstein, J. L. Cholesterol feedback: from Schoenheimer’s bottle to Scap’s MELADL. J. Lipid Res. 50 (suppl.). S15–S27 (2009)
Hampton, R. Y. & Garza, R. M. Protein quality control as a strategy for cellular regulation: lessons from ubiquitin-mediated regulation of the sterol pathway. Chem. Rev. 109, 1561–1574 (2009)
Burg, J. S. et al. Insig regulates HMG-CoA reductase by controlling enzyme phosphorylation in fission yeast. Cell Metab. 8, 522–531 (2008)
Loewen, C. J. et al. Phospholipid metabolism regulated by a transcription factor sensing phosphatidic acid. Science 304, 1644–1647 (2004)
Martin, C. E., Oh, C. S. & Jiang, Y. Regulation of long chain unsaturated fatty acid synthesis in yeast. Biochim. Biophys. Acta 1771, 271–285 (2007)
Kurat, C. F. et al. Cdk1/Cdc28-dependent activation of the major triacylglycerol lipase Tgl4 in yeast links lipolysis to cell-cycle progression. Mol. Cell 33, 53–63 (2009)
Fischer, J. et al. The gene encoding adipose triglyceride lipase (PNPLA2) is mutated in neutral lipid storage disease with myopathy. Nature Genet. 39, 28–30 (2007)
Dickson, R. C., Sumanasekera, C. & Lester, R. L. Functions and metabolism of sphingolipids in Saccharomyces cerevisiae . Prog. Lipid Res. 45, 447–465 (2006)
Cowart, L. A. & Obeid, L. M. Yeast sphingolipids: recent developments in understanding biosynthesis, regulation, and function. Biochim. Biophys. Acta 1771, 421–431 (2007)
Hanada, K. et al. Molecular machinery for non-vesicular trafficking of ceramide. Nature 426, 803–809 (2003)
D’Angelo, G. et al. Glycosphingolipid synthesis requires FAPP2 transfer of glucosylceramide. Nature 449, 62–67 (2007)
Kumagai, K., Kawano, M., Shinkai-Ouchi, F., Nishijima, M. & Hanada, K. Interorganelle trafficking of ceramide is regulated by phosphorylation-dependent cooperativity between the PH and START domains of CERT. J. Biol. Chem. 282, 17758–17766 (2007)
Klemm, R. W. et al. Segregation of sphingolipids and sterols during formation of secretory vesicles at the trans-Golgi network. J. Cell Biol. 185, 601–612 (2009)
Aronova, S. et al. Regulation of ceramide biosynthesis by TOR complex 2. Cell Metab. 7, 148–158 (2008)
Vacaru, A. M. et al. Sphingomyelin synthase-related protein SMSr controls ceramide homeostasis in the ER. J. Cell Biol. 185, 1013–1027 (2009)
Hannun, Y. A. & Obeid, L. M. Principles of bioactive lipid signalling: lessons from sphingolipids. Nature Rev. Mol. Cell Biol. 9, 139–150 (2008)
Lebman, D. A. & Spiegel, S. Cross-talk at the crossroads of sphingosine-1-phosphate, growth factors, and cytokine signaling. J. Lipid Res. 49, 1388–1394 (2008)
Akinbami, L. J., Moorman, J. E., Garbe, P. L. & Sondik, E. J. Status of childhood asthma in the United States, 1980–2007. Pediatrics 123 (suppl. 3). S131–S145 (2009)
Vercelli, D. Discovering susceptibility genes for asthma and allergy. Nature Rev. Immunol. 8, 169–182 (2008)
Moffatt, M. F. et al. Genetic variants regulating ORMDL3 expression contribute to the risk of childhood asthma. Nature 448, 470–473 (2007)
Galanter, J. et al. ORMDL3 gene is associated with asthma in three ethnically diverse populations. Am. J. Respir. Crit. Care Med. 177, 1194–1200 (2008)
Wu, H. et al. Genetic variation in ORM1-like 3 (ORMDL3) and gasdermin-like (GSDML) and childhood asthma. Allergy 64, 629–635 (2009)
Bouzigon, E. et al. Effect of 17q21 variants and smoking exposure in early-onset asthma. N. Engl. J. Med. 359, 1985–1994 (2008)
Verlaan, D. J. et al. Allele-specific chromatin remodeling in the ZPBP2/GSDMB/ORMDL3 locus associated with the risk of asthma and autoimmune disease. Am. J. Hum. Genet. 85, 377–393 (2009)
Tavendale, R., Macgregor, D. F., Mukhopadhyay, S. & Palmer, C. N. A polymorphism controlling ORMDL3 expression is associated with asthma that is poorly controlled by current medications. J. Allergy Clin. Immunol. 121, 860–863 (2008)
Hjelmqvist, L. et al. ORMDL proteins are a conserved new family of endoplasmic reticulum membrane proteins. Genome Biol. 3 10.1186/gb-2002-3-6-research0027 (2002)
Schuldiner, M. et al. Exploration of the function and organization of the yeast early secretory pathway through an epistatic miniarray profile. Cell 123, 507–519 (2005)
Hanada, K. Serine palmitoyltransferase, a key enzyme of sphingolipid metabolism. Biochim. Biophys. Acta 1632, 16–30 (2003)
Beeler, T. et al. The Saccharomyces cerevisiae TSC10/YBR265w gene encoding 3-ketosphinganine reductase is identified in a screen for temperature-sensitive suppressors of the Ca2+-sensitive csg2Δ mutant. J. Biol. Chem. 273, 30688–30694 (1998)
Ejsing, C. S. et al. Global analysis of the yeast lipidome by quantitative shotgun mass spectrometry. Proc. Natl Acad. Sci. USA 106, 2136–2141 (2009)
Delgado, A., Casas, J., Llebaria, A., Abad, J. L. & Fabrias, G. Inhibitors of sphingolipid metabolism enzymes. Biochim. Biophys. Acta 1758, 1957–1977 (2006)
Strahl, T. & Thorner, J. Synthesis and function of membrane phosphoinositides in budding yeast, Saccharomyces cerevisiae . Biochim. Biophys. Acta 1771, 353–404 (2007)
Brice, S. E., Alford, C. W. & Cowart, L. A. Modulation of sphingolipid metabolism by the phosphatidylinositol-4-phosphate phosphatase Sac1p through regulation of phosphatidylinositol in Saccharomyces cerevisiae . J. Biol. Chem. 284, 7588–7596 (2009)
Kearns, B. G. et al. Essential role for diacylglycerol in protein transport from the yeast Golgi complex. Nature 387, 101–105 (1997)
Kinoshita, E., Kinoshita-Kikuta, E., Takiyama, K. & Koike, T. Phosphate-binding tag, a new tool to visualize phosphorylated proteins. Mol. Cell. Proteomics 5, 749–757 (2006)
Tabuchi, M., Audhya, A., Parsons, A. B., Boone, C. & Emr, S. D. The phosphatidylinositol 4,5-biphosphate and TORC2 binding proteins Slm1 and Slm2 function in sphingolipid regulation. Mol. Cell. Biol. 26, 5861–5875 (2006)
Blagoveshchenskaya, A. & Mayinger, P. SAC1 lipid phosphatase and growth control of the secretory pathway. Mol. Biosyst. 5, 36–42 (2009)
Hornemann, T., Wei, Y. & von Eckardstein, A. Is the mammalian serine palmitoyltransferase a high-molecular-mass complex? Biochem. J. 405, 157–164 (2007)
Rivera, J., Proia, R. L. & Olivera, A. The alliance of sphingosine-1-phosphate and its receptors in immunity. Nature Rev. Immunol. 8, 753–763 (2008)
Ammit, A. J. et al. Sphingosine 1-phosphate modulates human airway smooth muscle cell functions that promote inflammation and airway remodeling in asthma. FASEB J. 15, 1212–1214 (2001)
Masini, E. et al. Ceramide: a key signaling molecule in a guinea pig model of allergic asthmatic response and airway inflammation. J. Pharmacol. Exp. Ther. 324, 548–557 (2008)
Ryan, J. J. & Spiegel, S. The role of sphingosine-1-phosphate and its receptors in asthma. Drug News Perspect. 21, 89–96 (2008)
Idzko, M. et al. Local application of FTY720 to the lung abrogates experimental asthma by altering dendritic cell function. J. Clin. Invest. 116, 2935–2944 (2006)
Barrett, J. C. et al. Genome-wide association defines more than 30 distinct susceptibility loci for Crohn’s disease. Nature Genet. 40, 955–962 (2008)
Barrett, J. C. et al. Genome-wide association study and meta-analysis find that over 40 loci affect risk of type 1 diabetes. Nature Genet. 41, 703–707 (2009)
Hirschfield, G. M. et al. Primary biliary cirrhosis associated with HLA, IL12A, and IL12RB2 variants. N. Engl. J. Med. 360, 2544–2555 (2009)
Gable, K., Slife, H., Bacikova, D., Monaghan, E. & Dunn, T. M. Tsc3p is an 80-amino acid protein associated with serine palmitoyltransferase and required for optimal enzyme activity. J. Biol. Chem. 275, 7597–7603 (2000)
Lester, R. L. & Dickson, R. C. High-performance liquid chromatography analysis of molecular species of sphingolipid-related long chain bases and long chain base phosphates in Saccharomyces cerevisiae after derivatization with 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate. Anal. Biochem. 298, 283–292 (2001)
Jiménez, C. R., Huang, L., Qiu, Y. & Burlingame, A. L. in Current Protocols in Protein Science (eds Coligan, J. E., Dunn, B. M., Ploegh, H. L., Speicher, D. W. & Wingfield, P. T.) 16.4.1–16.4.5 (John Wiley, 1998)
Huber, A. et al. Characterization of the rapamycin-sensitive phosphoproteome reveals that Sch9 is a central coordinator of protein synthesis. Genes Dev. 23, 1929–1943 (2009)
Acknowledgements
We acknowledge A. Falick, S. Zhou and D. King for identification of proteins by mass spectrometry; M. Schuldiner and S. Wang for E-MAP data collection; D. Fiedler and K. Shokat for providing the phosphate-binding acrylamide reagent; E. Griffis and M. D’Ambrosio for assistance with fluorescence microscopy; N. Ingolia for a codon-optimized mCherry-tagging plasmid; E. Burchard and J. Galanter for discussions of asthma genetics; I. Poser and A. Hyman for advice and providing the HeLa-Kyoto cell line; and M. Bassik, G. Brar, V. Denic, A. Frost, N. Ingolia, E. Quan, B. Toyama and other members of the Weissman laboratory for discussions. This work was supported by funding from Deutsche Forschungsgemeinschaft SFB/TR 13 projects A1 (K.S.) and D1 (A.S.), EUFP6 PRISM (K.S.), ETH Zurich (R.A.), the National Heart, Lung, and Blood Institute, National Institute of Health (N01-HV-28179) (R.A.), SystemsX.ch, the Swiss initiative for systems biology (R.A.), the Boehringer Ingelheim Fonds and the Swiss National Science Foundation (B.B.), the Howard Hughes Medical Institute (J.S.W.), the University of California, San Francisco Strategic Asthma Basic Research Center (J.S.W.), the National Science Foundation Graduate Research Fellowship Program (D.K.B.) and the Fannie and John Hertz Foundation (D.K.B.).
Author Contributions D.K.B. designed, performed and interpreted experiments. S.R.C. oversaw E-MAP data collection and analysis. B.B. performed and analysed protein mass spectrometry experiments to identify sites of phosphorylation under the supervision of R.A. C.S.E. performed and analysed lipidomic measurements with the support of A.S. and K.S. J.S.W. designed and interpreted experiments. D.K.B. and J.S.W. prepared the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
This file contains Supplementary Figures 1-7 with Legends, Supplementary Methods, Supplementary Table 1 and Supplementary References. (PDF 16508 kb)
Supplementary Data 1
This file contains genetic interaction (E-MAP) data including the genetic interaction scores obtained for select mutant strains with all other strains in our E-MAP dataset. Correlation values obtained for pair-wise comparisons of interaction profiles are also included. (XLS 425 kb)
Supplementary Data 2
This file contains additional information on lipid species quantified by mass spectrometry for lipidomic experiments performed on yeast and Hela cell samples. (XLS 158 kb)
Rights and permissions
About this article
Cite this article
Breslow, D., Collins, S., Bodenmiller, B. et al. Orm family proteins mediate sphingolipid homeostasis. Nature 463, 1048–1053 (2010). https://doi.org/10.1038/nature08787
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08787
This article is cited by
-
IL-10 constrains sphingolipid metabolism to limit inflammation
Nature (2024)
-
Simultaneous reduction of all ORMDL proteins decreases the threshold of mast cell activation
Scientific Reports (2023)
-
Ceramide sensing by human SPT-ORMDL complex for establishing sphingolipid homeostasis
Nature Communications (2023)
-
Lipid remodeling of adipose tissue in metabolic health and disease
Experimental & Molecular Medicine (2023)
-
A set of gene knockouts as a resource for global lipidomic changes
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.