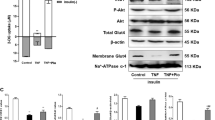
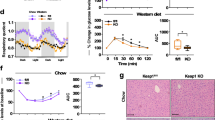
Abstract
Sirtuins are NAD+-dependent protein deacetylases. They mediate adaptive responses to a variety of stresses, including calorie restriction and metabolic stress. Sirtuin 3 (SIRT3) is localized in the mitochondrial matrix, where it regulates the acetylation levels of metabolic enzymes, including acetyl coenzyme A synthetase 2 (refs 1, 2). Mice lacking both Sirt3 alleles appear phenotypically normal under basal conditions, but show marked hyperacetylation of several mitochondrial proteins3. Here we report that SIRT3 expression is upregulated during fasting in liver and brown adipose tissues. During fasting, livers from mice lacking SIRT3 had higher levels of fatty-acid oxidation intermediate products and triglycerides, associated with decreased levels of fatty-acid oxidation, compared to livers from wild-type mice. Mass spectrometry of mitochondrial proteins shows that long-chain acyl coenzyme A dehydrogenase (LCAD) is hyperacetylated at lysine 42 in the absence of SIRT3. LCAD is deacetylated in wild-type mice under fasted conditions and by SIRT3 in vitro and in vivo; and hyperacetylation of LCAD reduces its enzymatic activity. Mice lacking SIRT3 exhibit hallmarks of fatty-acid oxidation disorders during fasting, including reduced ATP levels and intolerance to cold exposure. These findings identify acetylation as a novel regulatory mechanism for mitochondrial fatty-acid oxidation and demonstrate that SIRT3 modulates mitochondrial intermediary metabolism and fatty-acid use during fasting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Schwer, B., Bunkenborg, J., Verdin, R. O., Andersen, J. S. & Verdin, E. Reversible lysine acetylation controls the activity of the mitochondrial enzyme acetyl-CoA synthetase 2. Proc. Natl Acad. Sci. USA 103, 10224–10229 (2006)
Hallows, W. C., Lee, S. & Denu, J. M. Sirtuins deacetylate and activate mammalian acetyl-CoA synthetases. Proc. Natl Acad. Sci. USA 103, 10230–10235 (2006)
Lombard, D. B. et al. Mammalian Sir2 homolog SIRT3 regulates global mitochondrial lysine acetylation. Mol. Cell. Biol. 27, 8807–8814 (2007)
Kim, S. C. et al. Substrate and functional diversity of lysine acetylation revealed by a proteomics survey. Mol. Cell 23, 607–618 (2006)
Guarente, L. Sirtuins as potential targets for metabolic syndrome. Nature 444, 868–874 (2006)
Schwer, B. & Verdin, E. Conserved metabolic regulatory functions of sirtuins. Cell Metab. 7, 104–112 (2008)
Schwer, B. et al. Calorie restriction alters mitochondrial protein acetylation. Aging Cell 8, 604–606 (2009)
Cox, K. B. et al. Gestational, pathologic and biochemical differences between very long-chain acyl-CoA dehydrogenase deficiency and long-chain acyl-CoA dehydrogenase deficiency in the mouse. Hum. Mol. Genet. 10, 2069–2077 (2001)
Kurtz, D. M. et al. Targeted disruption of mouse long-chain acyl-CoA dehydrogenase gene reveals crucial roles for fatty acid oxidation. Proc. Natl Acad. Sci. USA 95, 15592–15597 (1998)
Zhang, D. et al. Mitochondrial dysfunction due to long-chain Acyl-CoA dehydrogenase deficiency causes hepatic steatosis and hepatic insulin resistance. Proc. Natl Acad. Sci. USA 104, 17075–17080 (2007)
Son, C. G. et al. Database of mRNA gene expression profiles of multiple human organs. Genome Res. 15, 443–450 (2005)
Ahn, B. H. et al. A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc. Natl Acad. Sci. USA 105, 14447–14452 (2008)
Schuler, A. M. & Wood, P. A. Mouse models for disorders of mitochondrial fatty acid beta-oxidation. ILAR J 43, 57–65 (2002)
Tolwani, R. J. et al. Medium-chain acyl-CoA dehydrogenase deficiency in gene-targeted mice. PLoS Genet. 1, e23 (2005)
Herrema, H. et al. Disturbed hepatic carbohydrate management during high metabolic demand in medium-chain acyl-CoA dehydrogenase (MCAD)-deficient mice. Hepatology 47, 1894–1904 (2008)
Spiekerkoetter, U. et al. Evidence for impaired gluconeogenesis in very long-chain acyl-CoA dehydrogenase-deficient mice. Horm. Metab. Res. 38, 625–630 (2006)
Zhang, W., Della-Fera, M. A., Hartzell, D., Hausman, D. & Baile, C. Adipose tissue gene expression profiles in ob/ob mice treated with leptin. Life Sci. 83, 35–42 (2008)
Yechoor, V. K. et al. Distinct pathways of insulin-regulated versus diabetes-regulated gene expression: an in vivo analysis in MIRKO mice. Proc. Natl Acad. Sci. USA 101, 16525–16530 (2004)
Schwer, B., North, B. J., Frye, R. A., Ott, M. & Verdin, E. The human silent information regulator (Sir)2 homologue hSIRT3 is a mitochondrial nicotinamide adenine dinucleotide-dependent deacetylase. J. Cell Biol. 158, 647–657 (2002)
Yang, H. et al. Nutrient-sensitive mitochondrial NAD+ levels dictate cell survival. Cell 130, 1095–1107 (2007)
McGarry, J. D. & Foster, D. W. Regulation of hepatic fatty acid oxidation and ketone body production. Annu. Rev. Biochem. 49, 395–420 (1980)
Eaton, S., Bartlett, K. & Pourfarzam, M. Mammalian mitochondrial β-oxidation. Biochem. J. 320, 345–357 (1996)
Shibata, M., Kihara, Y., Taguchi, M., Tashiro, M. & Otsuki, M. Nonalcoholic fatty liver disease is a risk factor for type 2 diabetes in middle-aged Japanese men. Diabetes Care 30, 2940–2944 (2007)
Targher, G. et al. Nonalcoholic fatty liver disease and risk of future cardiovascular events among type 2 diabetic patients. Diabetes 54, 3541–3546 (2005)
Hsiao, P. J. et al. Significant correlations between severe fatty liver and risk factors for metabolic syndrome. J. Gastroenterol. Hepatol. 22, 2118–2123 (2007)
Kim, J. Y., Hickner, R. C., Cortright, R. L., Dohm, G. L. & Houmard, J. A. Lipid oxidation is reduced in obese human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 279, E1039–E1044 (2000)
Frerman, F. E. & Goodman, S. I. Fluorometric assay of acyl-CoA dehydrogenases in normal and mutant human fibroblasts. Biochem. Med. 33, 38–44 (1985)
Bennett, M. J. Assays of fatty acid β-oxidation activity. Methods Cell Biol. 80, 179–197 (2007)
Srere, P. A. Citrate synthase. Methods Enzymol. 13, 3–11 (1969)
Wu, J. Y. et al. ENU mutagenesis identifies mice with mitochondrial branched-chain aminotransferase deficiency resembling human maple syrup urine disease. J. Clin. Invest. 113, 434–440 (2004)
Jensen, M. V. et al. Compensatory responses to pyruvate carboxylase suppression in islet β-cells. Preservation of glucose-stimulated insulin secretion. J. Biol. Chem. 281, 22342–22351 (2006)
Folch, J., Lees, M. & Sloane Stanley, G. H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 226, 497–509 (1957)
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959)
Morrison, W. R. & Smith, L. M. Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride-methanol. J. Lipid Res. 5, 600–608 (1964)
Oliver, H. & Lowry, J. V. P. A Flexible System of Enzymatic Analysis (Academic Press, 1972)
Saha, A. K. et al. Pioglitazone treatment activates AMP-activated protein kinase in rat liver and adipose tissue in vivo. Biochem. Biophys. Res. Commun. 314, 580–585 (2004)
Stremmel, W. & Berk, P. D. Hepatocellular influx of [14C]oleate reflects membrane transport rather than intracellular metabolism or binding. Proc. Natl Acad. Sci. USA 83, 3086–3090 (1986)
Rikova, K. et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell 131, 1190–1203 (2007)
Rush, J. et al. Immunoaffinity profiling of tyrosine phosphorylation in cancer cells. Nature Biotechnol. 23, 94–101 (2005)
Graham, J. M. Isolation of mitochondria from tissues and cells by differential centrifugation. Curr. Protocols Cell Biol. (suppl. 4), unit 3.3 (1999)
Graham, J. M. Purification of a crude mitochondrial fraction by density-gradient centrifugation. Curr. Protocols Cell Biol. (suppl. 4), unit 3.4 (1999)
Hirschey, M. D., Shimazu, T., Huang, J. & Verdin, E. Acetylation of mitochondrial proteins. Methods Enzymol. 457, 137–147 (2009)
Taniguchi, C. M., Ueki, K. & Kahn, R. Complementary roles of IRS-1 and IRS-2 in the hepatic regulation of metabolism. J. Clin. Invest. 115, 718–727 (2005)
Ueki, K., Kondo, T., Tseng, Y. H. & Kahn, C. R. Central role of suppressors of cytokine signaling proteins in hepatic steatosis, insulin resistance, and the metabolic syndrome in the mouse. Proc. Natl Acad. Sci. USA 101, 10422–10427 (2004)
Acknowledgements
We thank T. Canavan and J. J. Maher for primary hepatocyte preparation (P30 DK026743); C. Harris, L. Swift and the Mouse Metabolic Phenotyping Center (DK59637) for analysis of plasma and tissue lipid analysis; J. Wong for electron microscopy studies, S. Mihalik and D. Cuebas for synthesis of the 2,6-dimethylheptanoyl-CoA; A.-W. Mohsen for purification of recombinant pig ETF; Y.-C. Si for assistance with adenoviral studies; A. Wilson and J. Carroll for preparation of figures; and G. Howard and S. Ordway for editorial review. This work was supported in part by a Senior Scholarship in Aging from the Elison Medical Foundation to E.V. and by institutional support from the J. David Gladstone Institutes. F.W.A. is an Investigator of the Howard Hughes Medical Institute and recipient of an Ellison Medical Foundation Senior Scholar Award. D.B.L. is supported by a K08 award from the National Institute on Aging and the National Institutes of Health. B.S. is supported by an Ellison Medical Foundation/AFAR Senior Postdoctoral Research Grant. N.B.R. and A.K.S. are supported by National Institutes of Health (NIH) grants PO1 HL068758-06A1, DK019514-29 and R01 DK067509-04.
Author Contributions M.D.H., T.S., E.G., E.J., B.S., C.A.G., C.H., S.B. and A.K.S. performed in vitro, in vivo and biochemical studies; O.R.I., R.D.S. and J.R.B. performed metabolomic studies; D.B.L. and Y.L. performed mass spectrometry studies; M.D.H. and E.V. designed the studies, analysed the data and wrote the manuscript; all other authors reviewed and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S10 with Legends and Supplementary Table (PDF 1117 kb)
Rights and permissions
About this article
Cite this article
Hirschey, M., Shimazu, T., Goetzman, E. et al. SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 464, 121–125 (2010). https://doi.org/10.1038/nature08778
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08778
This article is cited by
-
Sirtuins in kidney health and disease
Nature Reviews Nephrology (2024)
-
Protein lysine acetylation does not contribute to the high rates of fatty acid oxidation seen in the post-ischemic heart
Scientific Reports (2024)
-
Mitochondrial impairment, decreased sirtuin activity and protein acetylation in dorsal root ganglia in Friedreich Ataxia models
Cellular and Molecular Life Sciences (2024)
-
Arsenic and adipose tissue: an unexplored pathway for toxicity and metabolic dysfunction
Environmental Science and Pollution Research (2024)
-
Impact of NAD+ metabolism on ovarian aging
Immunity & Ageing (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.