Abstract
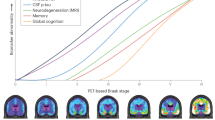
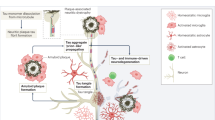
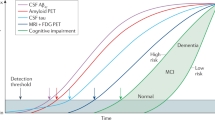
Alzheimer's disease affects millions of people around the world. Currently, there are no treatments that prevent or slow the disease. Like other neurodegenerative diseases, Alzheimer's disease is characterized by protein misfolding in the brain. This process and the associated brain damage begin years before the substantial neurodegeneration that accompanies dementia. Studies using new neuroimaging techniques and fluid biomarkers suggest that Alzheimer's disease pathology can be detected preclinically. These advances should allow the design of new clinical trials and early mechanism-based therapeutic intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Alzheimer, A. About a peculiar disease of the cerebral cortex [translation]. Alzheimer Dis. Assoc. Disord. 1, 7–8 (1987).
Khachaturian, Z. S. Diagnosis of Alzheimer's disease. Arch. Neurol. 42, 1097–1105 (1985).
Mirra, S. S. et al. The consortium to establish a registry for Alzheimer's disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer's disease. Neurology 41, 479–486 (1991).
Hyman, B. T. & Trojanowski, J. Q. Consensus recommendations for the postmortem diagnosis of Alzheimer disease from the National Institute on Aging and the Reagan Institute Working Group on diagnostic criteria for the neuropathological assessment of Alzheimer disease. J. Neuropathol. Exp. Neurol. 56, 1095–1097 (1997).
Crystal, H. et al. Clinico-pathologic studies in dementia: nondemented subjects with pathologically confirmed Alzheimer's disease. Neurology 38, 1682–1687 (1988).
Price, J. L. et al. Neuropathology of nondemented aging: presumptive evidence for preclinical Alzheimer disease. Neurobiol. Aging 30, 1026–1036 (2009).
Braak, H. & Braak, E. Frequency of stages of Alzheimer-related lesions in different age categories. Neurobiol. Aging 18, 351–357 (1997).
Evans, D. A. et al. Prevalence of Alzheimer's disease in a community population of older persons. Higher than previously reported. J. Am. Med. Assoc. 262, 2551–2556 (1989).
Morris, J. C. & Price, J. L. Pathologic correlates of nondemented aging, mild cognitive impairment, and early stage Alzheimer's disease. J. Mol. Neurosci. 17, 101–118 (2001).
Gomez-Isla, T. et al. Profound loss of layer II entorhinal cortex neurons occurs in very mild Alzheimer's disease. J. Neurosci. 16, 4491–4500 (1996).
Hardy, J. The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science 297, 353–356 (2002).
Morris, J. C. The clinical dementia rating (CDR). Current version and scoring rules. Neurology 43, 2412–2414 (1993).
Petersen, R. C. et al. Mild cognitive impairment: clinical characterization and outcome. Arch. Neurol. 56, 303–308 (1999).
Fox, N., Warrington, E. & Rossor, M. Serial magnetic resonance imaging of cerebral atrophy in preclinical Alzheimer's disease. Lancet 353, 2125 (1999). This paper reports how 5 of 28 cognitively normal individuals at risk of autosomal-dominant early-onset Alzheimer's disease developed “probable Alzheimer's disease” and could be distinguished by a mean rate of global cerebral atrophy of 1.5% per year from clinically stable subjects with a rate of 0.1% to 0.2% per year.
Carlson, N. E. et al. Trajectories of brain loss in aging and the development of cognitive impairment. Neurology 70, 828–833 (2008).
Devanand, D. P. et al. Hippocampal and entorhinal atrophy in mild cognitive impairment: prediction of Alzheimer disease. Neurology 68, 828–836 (2007).
Small, G. W. et al. PET of brain amyloid and tau in mild cognitive impairment. N. Engl. J. Med. 355, 2652–2663 (2006).
Klunk, W. E. et al. Imaging brain amyloid in Alzheimer's disease with Pittsburgh compound-B. Ann. Neurol. 55, 306–319 (2004). This first study of the tracer compound PIB in humans describes both the retention of PIB in areas of Alzheimer's-disease-affected brains known to contain amyloid and an inverse relationship between the PIB PET signal and cerebral glucose metabolism, measured by fluorodeoxyglucose PET.
Mintun, M. A. et al. [11C]PIB in a nondemented population: potential antecedent marker of Alzheimer disease. Neurology 67, 446–452 (2006).
Fagan, A. M. et al. Inverse relation between in vivo amyloid imaging load and CSF Aβ42 in humans. Ann. Neurol. 59, 512–519 (2006). This study illustrates an inverse relationship between mean cortical retention of PIB and cerebrospinal-fluid A β 42 concentrations among both demented and non-demented subjects, suggesting that brain amyloid deposition results in low cerebrospinal-fluid A β 42 and that amyloid imaging and cerebrospinal-fluid A β 42 might serve as antecedent biomarkers of preclinical Alzheimer's disease.
Morris J. C. et al. PIB imaging predicts progression to symptomatic Alzheimer's disease. Arch. Neurol. (in the press).
Zhang, M. R. et al. Development of a new radioligand, N-(5-fluoro-2-phenoxyphenyl)-N-(2-[18F]fluoroethyl-5-methoxybenzyl)acetamide, for PET imaging of peripheral benzodiazepine receptor in primate brain. J. Med. Chem. 47, 2228–2235 (2004).
Vas, A. et al. Functional neuroimaging in multiple sclerosis with radiolabelled glia markers: preliminary comparative PET studies with [11C]vinpocetine and [11C]PK11195 in patients. J. Neurol. Sci. 264, 9–17 (2008).
Edison, P. et al. Microglia, amyloid, and cognition in Alzheimer's disease: an [11C](R)PK11195-PET and [11C]PIB-PET study. Neurobiol. Dis. 32, 412–419 (2008).
Nakamura, S. et al. Expression of monoamine oxidase B activity in astrocytes of senile plaques. Acta Neuropathol. 80, 419–425 (1990).
Hirvonen, J. et al. Assessment of MAO-B occupancy in the brain with PET and [11C]-L-deprenyl-D2: a dose-finding study with a novel MAO-B inhibitor, EVT 301. Clin. Pharmacol. Ther. 85, 506–512 (2009).
Dickerson, B. C. & Sperling, R. A. Functional abnormalities of the medial temporal lobe memory system in mild cognitive impairment and Alzheimer's disease: insights from functional MRI studies. Neuropsychologia 46, 1624–1635 (2008).
Buckner, R. L. et al. Molecular, structural, and functional characterization of Alzheimer's disease: Evidence for a relationship between default activity, amyloid, and memory. J. Neurosci. 25, 7709–7717 (2005). In this paper, five in vivo neuroimaging methods are used to illustrate the similarities among the anatomical distributions of atrophy, reduced glucose metabolism and amyloid deposits seen in Alzheimer's disease, and of the default network, suggesting that young-adult brain activity and metabolism patterns may be conducive to cortical amyloid deposition.
Hirao, K. et al. The prediction of rapid conversion to Alzheimer's disease in mild cognitive impairment using regional cerebral blood flow SPECT. Neuroimage 28, 1014–1021 (2005).
de Leon, M. J. et al. Prediction of cognitive decline in normal elderly subjects with 2-[18F]fluoro-2-deoxy-D-glucose/positron-emission tomography (FDG/PET). Proc. Natl Acad. Sci. USA 98, 10966–10971 (2001).
Chetelat, G. et al. Mild cognitive impairment — can FDG-PET predict who is to rapidly convert to Alzheimer's disease? Neurology 60, 1374–1377 (2003).
Kadir, A. et al. Effect of phenserine treatment on brain functional activity and amyloid in Alzheimer's disease. Ann. Neurol. 63, 621–631 (2008).
Gusnard, D. A. & Raichle, M. E. Searching for a baseline: functional imaging and the resting human brain. Nature Rev. Neurosci. 2, 685–694 (2001).
Buckner, R. L., Andrews-Hanna, J. R. & Schacter, D. L. The brain's default network: anatomy, function, and relevance to disease. Ann. NY Acad. Sci. 1124, 1–38 (2008).
Kamenetz, F. et al. APP processing and synaptic function. Neuron 37, 925–937 (2003).
Cirrito, J. R. et al. Synaptic activity regulates interstitial fluid amyloid-beta levels in vivo . Neuron 48, 913–922 (2005).
Brody, D. L. et al. Amyloid-beta dynamics correlate with neurological status in the injured human brain. Science 321, 1221–1224 (2008).
Motter, R. et al. Reduction of β-amyloid peptide42 in the cerebrospinal fluid of patients with Alzheimer's disease. Ann. Neurol. 38, 643–648 (1995).
Fagan, A. M. et al. Decreased cerebrospinal fluid Aβ42 correlates with brain atrophy in cognitively normal elderly. Ann. Neurol. 65, 176–183 (2009).
Tapiola, T. et al. Cerebrospinal fluid β-amyloid 42 and tau proteins as biomarkers of Alzheimer-type pathologic changes in the brain. Arch. Neurol. 66, 382–389 (2009).
Fagan, A. M. et al. Cerebrospinal fluid tau/β-amyloid42 ratio as a prediction of cognitive decline in nondemented older adults. Arch. Neurol. 64, 343–349 (2007).
Hansson, O. et al. Association between CSF biomarkers and incipient Alzheimer's disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurol. 5, 228–234 (2006). This report illustrates the utility of cerebrospinal-fluid biomarkers A β 42 , tau and phosphorylated tau in predicting the progression of patients from mild cognitive impairment to dementia attributed clinically to Alzheimer's disease.
Li, G. et al. CSF tau/Aβ42 ratio for increased risk of mild cognitive impairment: a follow-up study. Neurology 69, 631–639 (2007).
Cairns, N. J. et al. PiB-PET detection of cerebral Aβ may lag clinical, cognitive, and CSF markers of Alzheimer's disease: a case report. Arch. Neurol. (in the press).
Mehta, P. D., Pirttila, T., Patrick, B., Barshatzky, M. & Mehta, S. Amyloid β protein 1–40 and 1–42 levels in matched cerebrospinal fluid and plasma from patients with Alzheimer disease. Neurosci. Lett. 304, 102–106 (2001).
Vanderstichele, H. et al. Standardization of measurement of β-amyloid(1-42) in cerebrospinal fluid and plasma. Amyloid 7, 245–258 (2000).
Graff-Radford, N. R. et al. Association of low plasma Aβ42/Aβ40 ratios with increased imminent risk for mild cognitive impairment and Alzheimer disease. Arch. Neurol. 64, 354–362 (2007).
Mayeux, R. et al. Plasma amyloid β-peptide 1–42 and incipient Alzheimer's disease. Ann. Neurol. 46, 412–416 (1999).
Portelius, E. et al. Characterization of tau in cerebrospinal fluid using mass spectrometry. J. Proteome Res. 7, 2114–2120 (2008).
Vandermeeren, M. et al. Detection of tau proteins in normal and Alzheimer's disease cerebrospinal fluid with a sensitive sandwich enzyme-linked immunosorbant assay. J. Neurochem. 61, 1828–1834 (1993).
Meyer-Luehmann, M. et al. Rapid appearance and local toxicity of amyloid-β plaques in a mouse model of Alzheimer's disease. Nature 451, 720–724 (2008).
Montine, T. J. et al. The magnitude of brain lipid peroxidation correlates with the extent of degeneration but not with density of neuritic plaques or neurofibrillary tangles or with APOE genotype in Alzheimer's disease patients. Am. J. Pathol. 155, 863–868 (1999).
Praticò, D., Lee, M.-Y.V., Trojanowski, J. Q., Rokach, J. & Fitzgerald, G. A. Increased F2-isoprostanes in Alzheimer's disease: evidence for enhanced lipid peroxidation in vivo . FASEB J. 12, 1777–1783 (1998).
de Leon, M. J. et al. Longitudinal CSF isoprostane and MRI atrophy in the progression to Alzheimer's disease. J. Neurol. 254, 1666–1675 (2007).
Pratico, D. et al. Increased 8,12-iso-iPF2α-VI in Alzheimer's disease: correlation of a noninvasive index of lipid peroxidation with disease severity. Ann. Neurol. 48, 809–812 (2000).
Montine, T. J. et al. No difference in plasma or urinary F2-isoprostanes among patients with Huntington's disease or Alzheimer's disease and controls. Ann. Neurol. 48, 950 (2000).
Abe, T., Tohgi, H., Isobe, C., Murata, T. & Sato, C. Remarkable increase in the concentration of 8-hydroxyguanosine in cerebrospinal fluid from patients with Alzheimer's disease. J. Neurosci. Res. 70, 447–450 (2002).
Abraham, C. R., Selkoe, D. J. & Potter, H. Immunochemical identification of the serine protease inhibitor α1-antichymotrypsin in the brain amyloid deposits of Alzheimer's disease. Cell 52, 487–501 (1988).
Thal, D. R., Schober, R. & Birkenmeier, G. The subunits of α2-macroglobulin receptor/low density lipoprotein receptor-related protein, native and transformed α2-macroglobulin and interleukin 6 in Alzheimer's disease. Brain Res. 777, 223–227 (1997).
Zanjani, H. et al. Complement activation in very early Alzheimer disease. Alzheimer Dis. Assoc. Disord. 19, 55–66 (2005).
DeKosky, S. T. et al. Plasma and cerebrospinal fluid α1-antichymotrypsin levels in Alzheimer's disease: correlation with cognitive impairment. Ann. Neurol. 53, 81–90 (2003).
Hye, A. et al. Proteome-based plasma biomarkers for Alzheimer's disease. Brain 129, 3042–3050 (2006).
Hu, Y. et al. Identification and validation of novel CSF biomarkers for early stages of Alzheimer's disease. Proteomics Clin. Appl. 1, 1373–1384 (2007).
Thambisetty, M. et al. Proteome-based identification of plasma proteins associated with hippocampal metabolism in early Alzheimer's disease. J. Neurol. 255, 1712–1720 (2008).
Smyth, M. D. et al. Decreased levels of C1q in cerebrospinal fluid of living Alzheimer patients correlate with disease state. Neurobiol. Aging 15, 609–614 (1994).
Loeffler, D. A. et al. Cerebrospinal fluid C3a increases with age, but does not increase further in Alzheimer's disease. Neurobiol. Aging 18, 555–557 (1997).
Tan, Z. S. et al. Inflammatory markers and the risk of Alzheimer disease: the Framingham study. Neurology 68, 1902–1908 (2007).
Simard, A. R., Soulet, D., Gowing, G., Julien, J. P. & Rivest, S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer's disease. Neuron 49, 489–502 (2006).
Guerreiro, R. J. et al. Peripheral inflammatory cytokines as biomarkers in Alzheimer's disease and mild cognitive impairment. Neurodegener. Dis. 4, 406–412 (2007).
Ray, S. et al. Classification and prediction of clinical Alzheimer's diagnosis based on plasma signaling proteins. Nature Med. 13, 1359–1362 (2007). This paper identified a panel of 18 signalling proteins in blood plasma that might be used to predict and diagnose early Alzheimer's disease. This work illustrates the potential of 'unbiased' screening of multiple analytes in biological fluids to identify early diagnostic and prognostic biomarkers for Alzheimer's disease.
Davidsson, P. et al. Proteome analysis of cerebrospinal fluid proteins in Alzheimer patients. Neuroreport 13, 611–615 (2002).
Zhang, J. et al. Quantitative proteomics of cerebrospinal fluid from patients with Alzheimer disease. J. Alzheimer's Dis. 7, 125–133; discussion 173–180 (2005).
Finehout, E. J., Franck, Z., Choe, L. H., Relkin, N. & Lee, K. H. Cerebrospinal fluid proteomic biomarkers for Alzheimer's disease. Ann. Neurol. 61, 120–129 (2007).
Bateman, R. J. et al. Human amyloid-β synthesis and clearance rates as measured in cerebrospinal fluid in vivo . Nature Med. 12, 856–861 (2006). This report uses amyloid- β peptide species as an example to describe a novel technique that will facilitate the evaluation of disease-modifying treatments for Alzheimer's disease by measuring the production and clearance rates of proteins in the human central nervous system, following administration of a non-radioactive isotopically labelled amino acid (in this case leucine).
Bateman, R. J. et al. A γ-secretase inhibitor decreases amyloid-β production in the central nervous system. Ann. Neurol. 66, 48–54 (2009).
Kauwe, J. S. et al. Alzheimer's disease risk variants show association with cerebrospinal fluid amyloid beta. Neurogenetics 10, 13–17 (2009).
Craig-Schapiro, R., Fagan, A. M. & Holtzman, D. M. Biomarkers of Alzheimer's disease. Neurobiol. Dis. 35, 128–140 (2009).
Sunderland, T. et al. Decreased β-amyloid1–42 and increased tau levels in cerebrospinal fluid of patients with Alzheimer disease. J. Am. Med. Assoc. 289, 2094–2103 (2003).
Welsh-Bohmer, K. A. & Johnson, S. in Handbook of Dementing Illnesses 2nd edn (eds Morris, J. C., Galvin, J. E. & Holtzman, D. M.) 59–87 (Taylor & Francis, 2006).
Acknowledgements
We thank J. Cirrito and MedPIC, the art and design centre at Washington University School of Medicine, for assistance with graphic design. Work in the authors' laboratories was supported by US National Institutes of Health grants P01–AG026276, PO1–AG03991, P50AG00568125 and T32NS007205.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
Competing interests: D.M.H. is a co-founder of C2N Diagnostics, which focuses on biomarkers for neurodegenerative disease.
Additional information
Reprints and permissions information is available at http://www.nature.com/reprints.
Correspondence should be addressed to D.M.H. (holtzman@neuro.wustl.edu).
Rights and permissions
About this article
Cite this article
Perrin, R., Fagan, A. & Holtzman, D. Multimodal techniques for diagnosis and prognosis of Alzheimer's disease. Nature 461, 916–922 (2009). https://doi.org/10.1038/nature08538
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08538
This article is cited by
-
An MRI brain tumour detection using logistic regression-based machine learning model
International Journal of System Assurance Engineering and Management (2024)
-
Evaluating the feasibility of cognitive impairment detection in Alzheimer’s disease screening using a computerized visual dynamic test
Journal of NeuroEngineering and Rehabilitation (2023)
-
Decoding the heterogeneity of Alzheimer’s disease diagnosis and progression using multilayer networks
Molecular Psychiatry (2023)
-
CDiP technology for reverse engineering of sporadic Alzheimer’s disease
Journal of Human Genetics (2023)
-
Resting-state global brain activity affects early β-amyloid accumulation in default mode network
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.