Abstract
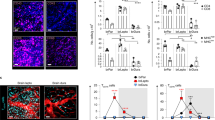
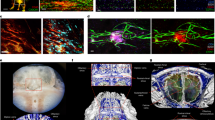
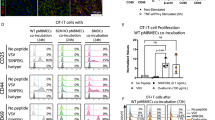
The tissues of the central nervous system are effectively shielded from the blood circulation by specialized vessels that are impermeable not only to cells, but also to most macromolecules circulating in the blood. Despite this seemingly absolute seclusion, central nervous system tissues are subject to immune surveillance and are vulnerable to autoimmune attacks1. Using intravital two-photon imaging in a Lewis rat model of experimental autoimmune encephalomyelitis, here we present in real-time the interactive processes between effector T cells and cerebral structures from their first arrival to manifest autoimmune disease. We observed that incoming effector T cells successively scanned three planes. The T cells got arrested to leptomeningeal vessels and immediately monitored the luminal surface, crawling preferentially against the blood flow. After diapedesis, the cells continued their scan on the abluminal vascular surface and the underlying leptomeningeal (pial) membrane. There, the T cells encountered phagocytes that effectively present antigens, foreign as well as myelin proteins. These contacts stimulated the effector T cells to produce pro-inflammatory mediators, and provided a trigger to tissue invasion and the formation of inflammatory infiltrations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wekerle, H., Linington, C., Lassmann, H. & Meyermann, R. Cellular immune reactivity within the CNS. Trends Neurosci. 9, 271–277 (1986)
Ben-Nun, A., Wekerle, H. & Cohen, I. R. The rapid isolation of clonable antigen-specific T lymphocyte lines capable of mediating autoimmune encephalomyelitis. Eur. J. Immunol. 11, 195–199 (1981)
Flügel, A., Willem, M., Berkowicz, T. & Wekerle, H. Gene transfer into CD4+ T lymphocytes: Green fluorescent protein engineered, encephalitogenic T cells used to illuminate immune responses in the brain. Nature Med. 5, 843–847 (1999)
Odoardi, F., Kawakami, N., Klinkert, W. E. F., Wekerle, H. & Flügel, A. Blood-borne soluble protein antigen intensifies T cell activation in autoimmune CNS lesions and exacerbates clinical disease. Proc. Natl Acad. Sci. USA 104, 18625–18630 (2007)
Traugott, U. & Raine, C. S. Acute experimental allergic encephalomyelitis. Myelin basic protein-reactive T cells in the circulation and in meningeal infiltrates. J. Neurol. Sci. 42, 331–336 (1979)
Tsuchida, M. et al. Identification of CD4-CD8- αβ T cells in the subarachnoid space of rats with experimental autoimmune encephalomyelitis. A possible route by which effector cells invade the lesions. Immunology 81, 420–427 (1994)
Auffray, C. et al. Monitoring of blood vessels and tissues by a population of monocytes with patrolling behavior. Science 317, 666–670 (2007)
Phillipson, M. et al. Intraluminal crawling of neutrophils to emigration sites: a molecularly distinct process from adhesion in the recruitment cascade. J. Exp. Med. 203, 2569–2575 (2006)
Geissmann, F. et al. Intravascular immune surveillance by CXCR6+ NKT cells patrolling liver sinusoids. PLoS Biol. 3, e113 (2005)
Flügel, A. et al. Migratory activity and functional changes of green fluorescent effector T cells before and during experimental autoimmune encephalomyelitis. Immunity 14, 547–560 (2001)
Vajkoczy, P., Laschinger, M. & Engelhardt, B. α4-integrin-VCAM binding mediates G protein independent capture of encephalitogenic T cell blasts to CNS white matter microvessels. J. Clin. Invest. 108, 557–565 (2001)
Kawakami, N. et al. Autoimmune CD4+ T cell memory: Lifelong persistence of encephalitogenic T cell clones in healthy immune repertoires. J. Immunol. 175, 69–81 (2005)
Wolf, K. et al. Compensation mechanism in tumor cell migration: mesenchymal–amoeboid transition after blocking of pericellular proteolysis. J. Cell Biol. 160, 267–277 (2003)
Yednock, T. A. et al. Prevention of experimental autoimmune encephalomyelitis by antibodies against α4β1 integrin. Nature 356, 63–66 (1992)
Schenkel, A. R., Mamdouh, Z. & Muller, W. A. Locomotion of monocytes on endothelium is a critical step during extravasation. Nature Immunol. 5, 393–400 (2004)
Carman, C. V. et al. Transcellular diapedesis is initiated by invasive podosomes. Immunity 26, 784–797 (2007)
Walther, M. et al. Exogenous antigen containing perivascular phagocytes induce a non-encephalitogenic extravasation of primed lymphocytes. J. Neuroimmunol. 117, 30–42 (2001)
Hickey, W. F., Vass, K. & Lassmann, H. Bone marrow derived elements in the central nervous system: an immunohistochemical and ultrastructural survey of rat chimeras. J. Neuropathol. Exp. Neurol. 51, 246–256 (1992)
Matyszak, M. K. & Perry, V. H. The potential role of dendritic cells in immune-mediated inflammatory diseases in the central nervous system. Neuroscience 74, 599–608 (1996)
McMenamin, P. G., Wealthall, R. J., Deverall, M., Cooper, S. J. & Griffin, B. Macrophages and dendritic cells in the rat meninges and choroid plexus: three-dimensional localisation by environmental scanning electron microscopy and confocal microscopy. Cell Tissue Res. 313, 259–269 (2003)
Kivisäkk, P. et al. Localizing central nervous system immune surveillance: Meningeal antigen-presenting cells activate T cells during experimental autoimmune encephalomyelitis. Ann. Neurol. 65, 457–469 (2009)
Reboldi, A. et al. C-C chemokine receptor 6–regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nature Immunol. 10, 514–523 (2009)
Becher, B., Bechmann, I. & Greter, M. Antigen presentation in autoimmunity and CNS inflammation: how T lymphocytes recognize the brain. J. Mol. Med. 84, 532–543 (2006)
Bechmann, I., Galea, I. & Perry, V. H. What is the blood-brain barrier (not)? Trends Immunol. 28, 5–11 (2007)
Hinrichs, D. J., Wegmann, K. W. & Dietsch, G. N. Transfer of experimental allergic encephalomyelitis to bone marrow chimeras: endothelial cells are not the restricting element. J. Exp. Med. 166, 1906–1911 (1987)
Hickey, W. F. & Kimura, H. Perivascular microglial cells of the CNS are bone-marrow derived and present antigen in vivo . Science 239, 290–292 (1988)
Lois, C., Hong, I. J., Pease, S., Brown, E. J. & Baltimore, D. Germline transmission and tissue-specific expression of transgenes delivered by lentiviral vectors. Science 295, 868–872 (2002)
Kawakami, N. et al. Live imaging of effector cell trafficking and autoantigen recognition within the unfolding autoimmune encephalomyelitis lesion. J. Exp. Med. 201, 1805–1814 (2005)
Kawakami, N. et al. The activation status of neuroantigen-specific T cells in the target organ determines the clinical outcome of autoimmune encephalomyelitis. J. Exp. Med. 199, 185–197 (2004)
Hadjantonakis, A.-K., Gertsenstein, M., Ikawa, M., Okabe, M. & Nagy, A. Generating green fluorescent mice by germline transmission of green fluorescent ES cells. Mech. Dev. 76, 79–90 (1998)
Eylar, E. H., Kniskern, P. J. & Jackson, J. J. Myelin basic proteins. Methods Enzymol. 32, 323–341 (1974)
Odoardi, F. et al. Instant effect of soluble antigen on effector T cells in peripheral immune organs during immunotherapy of autoimmune encephalomyelitis. Proc. Natl Acad. Sci. USA 104, 920–925 (2007)
Kuchroo, V. K. et al. Experimental allergic encephalomyelitis mediated by cloned T cells specific for a synthetic peptide of myelin proteolipid protein. Fine specificity and T cell receptor Vβ usage. J. Immunol. 148, 3776–3782 (1992)
Tamatani, T., Kotani, M. & Miyasaka, M. Characterization of the rat leukocyte integrin, CD11/CD18, by the use of LFA-1 subunit specific monoclonal antibodies. Eur. J. Immunol. 21, 627–633 (1991)
Issekutz, T. B. Dual inhibition of VLA-4 and LFA-1 maximally inhibits cutaneous delayed-type hypersensitivity-induced inflammation. Am. J. Pathol. 143, 1286–1293 (1993)
Kornack, D. R. & Rakic, P. Changes in cell-cycle kinetics during the development and evolution of primate neocortex. Proc. Natl Acad. Sci. USA 95, 1242–1246 (1998)
Acknowledgements
We thank S. Kosin, I. Haarmann, C. Federle, T. H. Ngugen and K. Nispel for technical assistance. We thank M. Krumbholz for help in statistical analysis, M. Hübener for support in 3D analysis software, and K. Dornmair, E. Meinl and E. Lütjen-Drecoll for critical comments on the paper. We are grateful to V. Staiger for providing software and R. Schorner for help with the artwork. This work was supported by the Deutsche Forschungsgemeinschaft (SFB455 project A8 and SFB571 project C6) and the Hertie foundation (Hertie no.1.01.1/04/010).
Author Contributions I.B., N.K. and C.S. performed most of the imaging experiments. N.K., F.O. and D.M. performed the experiments testing the antigen presentation capacity of meningeal/perivascular macrophages. F.O. performed the transcriptome analyses, EAE studies and cytofluorometric characterizations. W.E.F.K. and J.W.E. performed cell sorting. C.F.-K. contributed to the immunohistochemical studies. T.B.I. provided a monoclonal antibody. A.F. coordinated the experimental work. A.F. and H.W. designed the study and wrote the manuscript with inputs from co-authors.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-12 with Legends and Legends for Supplementary Movies 1-9. (PDF 15004 kb)
Supplementary Movie 1
This movie file shows TMBP-GFP cell motility within leptomeningeal vessels during early EAE - see file s1 for full Legend. (MOV 7815 kb)
Supplementary Movie 2
This movie file shows rolling of TMBP-GFP cells in vascular beds of peripheral organs - see file s1 for full Legend. (MOV 7734 kb)
Supplementary Movie 3
This movie file shows intraluminal crawling and detachment of TMBP-GFP cells in vessels of the CNS - see file s1 for full Legend. (MOV 11037 kb)
Supplementary Movie 4
This movie file shows diapedesis of TMBP-GFP cells - see file s1 for full Legend. (MOV 7458 kb)
Supplementary Movie 5
This movie file shows intravascular crawling is abolished by integrin neutralization - see file s1 for full Legend. (MOV 8599 kb)
Supplementary Movie 6
This movie file shows meningeal infiltration by encephalitogenic effector T cells - see file s1 for full Legend. (MOV 10134 kb)
Supplementary Movie 7
This movie shows perivascular movement and subsequent diffuse distribution of TMBP-GFP cells in the Meninges - see file s1 for full Legend. (MOV 14018 kb)
Supplementary Movie 8
This movie file shows Location and locomotion of perivascular and meningeal phagocytes - see file s1 for full Legend. (MOV 4165 kb)
Supplementary Movie 9
This movie file shows TMBP cells in contact to perivascular /meningeal phagocytes - see file s1 for full Legend. (MOV 25825 kb)
Rights and permissions
About this article
Cite this article
Bartholomäus, I., Kawakami, N., Odoardi, F. et al. Effector T cell interactions with meningeal vascular structures in nascent autoimmune CNS lesions. Nature 462, 94–98 (2009). https://doi.org/10.1038/nature08478
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08478
This article is cited by
-
Endothelial cells and macrophages as allies in the healthy and diseased brain
Acta Neuropathologica (2024)
-
Imaging of brain barrier inflammation and brain fluid drainage in human neurological diseases
Cellular and Molecular Life Sciences (2024)
-
Slc43a2+ T cell metastasis from spleen to brain in RGNNV infected teleost
Science China Life Sciences (2024)
-
The choroid plexus acts as an immune cell reservoir and brain entry site in experimental autoimmune encephalomyelitis
Fluids and Barriers of the CNS (2023)
-
Antigen recognition detains CD8+ T cells at the blood-brain barrier and contributes to its breakdown
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.