Abstract
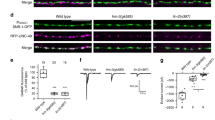
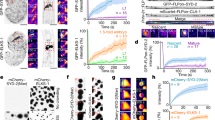
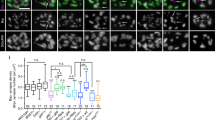
Efficient neurotransmission at chemical synapses relies on spatial congruence between the presynaptic active zone, where synaptic vesicles fuse, and the postsynaptic differentiation, where neurotransmitter receptors concentrate. Diverse molecular systems have evolved to localize receptors at synapses, but in most cases, they rely on scaffolding proteins localized below the plasma membrane1,2,3. A few systems have been suggested to control the synaptic localization of neurotransmitter receptors through extracellular interactions, such as the pentraxins that bind AMPA receptors and trigger their aggregation4. However, it is not yet clear whether these systems have a central role in the organization of postsynaptic domains in vivo or rather provide modulatory functions5. Here we describe an extracellular scaffold that is necessary to cluster acetylcholine receptors at neuromuscular junctions in the nematode Caenorhabditis elegans. It involves the ectodomain of the previously identified transmembrane protein LEV-10 (ref. 6) and a novel extracellular protein, LEV-9. LEV-9 is secreted by the muscle cells and localizes at cholinergic neuromuscular junctions. Acetylcholine receptors, LEV-9 and LEV-10 are interdependent for proper synaptic localization and physically interact based on biochemical evidence. Notably, the function of LEV-9 relies on eight complement control protein (CCP) domains. These domains, also called ‘sushi domains’, are usually found in proteins regulating complement activity in the vertebrate immune system7. Because the complement system does not exist in protostomes, our results suggest that some of the numerous uncharacterized CCP proteins expressed in the mammalian brain might be directly involved in the organization of the synapse, independently from immune functions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Elias, G. M. & Nicoll, R. A. Synaptic trafficking of glutamate receptors by MAGUK scaffolding proteins. Trends Cell Biol. 17, 343–352 (2007)
Kneussel, M. & Loebrich, S. Trafficking and synaptic anchoring of ionotropic inhibitory neurotransmitter receptors. Biol. Cell 99, 297–309 (2007)
Sanes, J. R. & Lichtman, J. W. Induction, assembly, maturation and maintenance of a postsynaptic apparatus. Nature Rev. Neurosci. 2, 791–805 (2001)
O’Brien, R. J. et al. Synaptic clustering of AMPA receptors by the extracellular immediate-early gene product Narp. Neuron 23, 309–323 (1999)
Bjartmar, L. et al. Neuronal pentraxins mediate synaptic refinement in the developing visual system. J. Neurosci. 26, 6269–6281 (2006)
Gally, C., Eimer, S., Richmond, J. E. & Bessereau, J. L. A transmembrane protein required for acetylcholine receptor clustering in Caenorhabditis elegans . Nature 431, 578–582 (2004)
Kirkitadze, M. D. & Barlow, P. N. Structure and flexibility of the multiple domain proteins that regulate complement activation. Immunol. Rev. 180, 146–161 (2001)
Lewis, J. A., Wu, C. H., Berg, H. & Levine, J. H. The genetics of levamisole resistance in the nematode Caenorhabditis elegans . Genetics 95, 905–928 (1980)
Fleming, J. T. et al. Caenorhabditis elegans levamisole resistance genes lev-1, unc-29, and unc-38 encode functional nicotinic acetylcholine receptor subunits. J. Neurosci. 17, 5843–5857 (1997)
Boulin, T. et al. Eight genes are required for functional reconstitution of the Caenorhabditis elegans levamisole-sensitive acetylcholine receptor. Proc. Natl Acad. Sci. USA 105, 18590–18595 (2008)
Touroutine, D. et al. acr-16 encodes an essential subunit of the levamisole-resistant nicotinic receptor at the Caenorhabditis elegans neuromuscular junction. J. Biol. Chem. 280, 27013–27021 (2005)
Francis, M. M. et al. The Ror receptor tyrosine kinase CAM-1 is required for ACR-16-mediated synaptic transmission at the C. elegans neuromuscular junction. Neuron 46, 581–594 (2005)
Bessereau, J. L. et al. Mobilization of a Drosophila transposon in the Caenorhabditis elegans germ line. Nature 413, 70–74 (2001)
Williams, D. C., Boulin, T., Ruaud, A. F., Jorgensen, E. M. & Bessereau, J. L. Characterization of Mos1-mediated mutagenesis in Caenorhabditis elegans: a method for the rapid identification of mutated genes. Genetics 169, 1779–1785 (2005)
Bingle, C. D. & Vyakarnam, A. Novel innate immune functions of the whey acidic protein family. Trends Immunol. 29, 444–453 (2008)
Soares, D. C. et al. Large-scale modelling as a route to multiple surface comparisons of the CCP module family. Protein Eng. Des. Sel. 18, 379–388 (2005)
Fares, H. & Greenwald, I. Genetic analysis of endocytosis in Caenorhabditis elegans: coelomocyte uptake defective mutants. Genetics 159, 133–145 (2001)
Robert, V. & Bessereau, J. L. Targeted engineering of the Caenorhabditis elegans genome following Mos1-triggered chromosomal breaks. EMBO J. 26, 170–183 (2007)
Qian, H., Robertson, A. P., Powell-Coffman, J. A. & Martin, R. J. Levamisole resistance resolved at the single-channel level in Caenorhabditis elegans . FASEB J. 22, 3247–3254 (2008)
Zheng, Y., Mellem, J. E., Brockie, P. J., Madsen, D. M. & Maricq, A. V. SOL-1 is a CUB-domain protein required for GLR-1 glutamate receptor function in C. elegans . Nature 427, 451–457 (2004)
Ng, D. et al. Neto1 is a novel CUB-domain NMDA receptor-interacting protein required for synaptic plasticity and learning. PLoS Biol. 7, e41 (2009)
Zhang, W. et al. A transmembrane accessory subunit that modulates kainate-type glutamate receptors. Neuron 61, 385–396 (2009)
Arlaud, G. J., Barlow, P. N., Gaboriaud, C., Gros, P. & Narayana, S. V. Deciphering complement mechanisms: the contributions of structural biology. Mol. Immunol. 44, 3809–3822 (2007)
Hoshino, M., Suzuki, E., Nabeshima, Y. & Hama, C. Hikaru genki protein is secreted into synaptic clefts from an early stage of synapse formation in Drosophila . Development 122, 589–597 (1996)
Hoshino, M. et al. Neural expression of hikaru genki protein during embryonic and larval development of Drosophila melanogaster . Dev. Genes Evol. 209, 1–9 (1999)
Roll, P. et al. SRPX2 mutations in disorders of language cortex and cognition. Hum. Mol. Genet. 15, 1195–1207 (2006)
Nonaka, M. & Yoshizaki, F. Primitive complement system of invertebrates. Immunol. Rev. 198, 203–215 (2004)
Robert, V. J., Katic, I. & Bessereau, J. L. Mos1 transposition as a tool to engineer the Caenorhabditis elegans genome by homologous recombination. Methods 10.1016/j.ymeth.2009.02.013 (in the press)
Boulin, T., Etchberger, J. F. & Hobert, O. Reporter gene fusions. WormBook doi/10.1895/wormbook.1.106.1. 1–23 (2006)
Liegeois, S., Benedetto, A., Garnier, J. M., Schwab, Y. & Labouesse, M. The V0-ATPase mediates apical secretion of exosomes containing Hedgehog-related proteins in Caenorhabditis elegans . J. Cell Biol. 173, 949–961 (2006)
Brenner, S. The genetics of Caenorhabditis elegans . Genetics 77, 71–94 (1974)
Ruaud, A. F. & Bessereau, J. L. Activation of nicotinic receptors uncouples a developmental timer from the molting timer in C. elegans . Development 133, 2211–2222 (2006)
Coudreuse, D. Y., Roel, G., Betist, M. C., Destree, O. & Korswagen, H. C. Wnt gradient formation requires retromer function in Wnt-producing cells. Science 312, 921–924 (2006)
Flanagan, J. G. et al. Alkaline phosphatase fusions of ligands or receptors as in situ probes for staining of cells, tissues, and embryos. Methods Enzymol. 327, 19–35 (2000)
Mello, C. C., Kramer, J. M., Stinchcomb, D. & Ambros, V. Efficient gene transfer in C. elegans: extrachromosomal maintenance and integration of transforming sequences. EMBO J. 10, 3959–3970 (1991)
Miller, K. G., Emerson, M. D., McManus, J. R. & Rand, J. B. RIC-8 (Synembryn): a novel conserved protein that is required for Gqα signaling in the C. elegans nervous system. Neuron 27, 289–299 (2000)
Gally, C. & Bessereau, J. L. GABA is dispensable for the formation of junctional GABA receptor clusters in Caenorhabditis elegans . J. Neurosci. 23, 2591–2599 (2003)
Duerr, J. S. et al. The cat-1 gene of Caenorhabditis elegans encodes a vesicular monoamine transporter required for specific monoamine-dependent behaviors. J. Neurosci. 19, 72–84 (1999)
Duerr, J. S., Gaskin, J. & Rand, J. B. Identified neurons in C. elegans coexpress vesicular transporters for acetylcholine and monoamines. Am. J. Physiol. Cell Physiol. 280, C1616–C1622 (2001)
Richmond, J. E., Davis, W. S. & Jorgensen, E. M. UNC-13 is required for synaptic vesicle fusion in C. elegans . Nature Neurosci. 2, 959–964 (1999)
Richmond, J. E. Electrophysiological recordings from the neuromuscular junction of C. elegans . WormBook 10.1895/wormbook.1.112.1, 1–8 (2006)
Acknowledgements
We thank E. M. Jorgensen and D. Williams for the lev-9(ox171::Mos1) strain, M. Labouesse for the anti-VHA-5 antibodies, J. Rand for the anti-UNC-17 antibodies, A. Fire for the GFP vectors, the Caenorhabditis Genetic Center and W. R. Schafer for strains, I. Katic, M. Zhen and S. Marty for critical reading of the manuscript, and H. Gendrot and B. Mathieu for technical help. M.G. was supported by a fellowship from the Ministère de la Recherche and by the Association Française contre les Myopathies. G.R. is a Ministère de la Recherche fellow. This work was funded by an INSERM Avenir grant, the Agence Nationale de la Recherche (ANR-07-NEURO-032-01) and the Association Française contre les Myopathies. J.E.R. was supported by NIH RO1 MH073156.
Author Contributions M.G. performed most of the experiments. J.E.R. performed all the electrophysiology experiments (Fig. 3j, k and Supplementary Fig. 9d). G.R. generated and characterized the unc-63::YFP knock-in strain. M.G. and J.-L.B. wrote the manuscript. J.-L.B. supervised the project.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
This file contains Supplementary Figures S1-S9 with Legends. (PDF 4433 kb)
Rights and permissions
About this article
Cite this article
Gendrel, M., Rapti, G., Richmond, J. et al. A secreted complement-control-related protein ensures acetylcholine receptor clustering. Nature 461, 992–996 (2009). https://doi.org/10.1038/nature08430
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08430
This article is cited by
-
Differential regulation of degradation and immune pathways underlies adaptation of the ectosymbiotic nematode Laxus oneistus to oxic-anoxic interfaces
Scientific Reports (2022)
-
The signaling pathway of levamisole-sensitive-acetylcholine receptors involved in short-term forgetting of Caenorhabditis elegans
Molecular Genetics and Genomics (2022)
-
A C. elegans model of C9orf72-associated ALS/FTD uncovers a conserved role for eIF2D in RAN translation
Nature Communications (2021)
-
Intrinsic and extrinsic mechanisms of synapse formation and specificity in C. elegans
Cellular and Molecular Life Sciences (2019)
-
Complement receptor activity of recombinant porcine CR1-like protein expressed in a eukaryotic system
Immunologic Research (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.