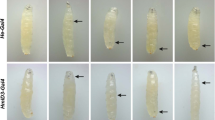
Abstract
Reactive oxygen species (ROS), produced during various electron transfer reactions in vivo, are generally considered to be deleterious to cells1. In the mammalian haematopoietic system, haematopoietic stem cells contain low levels of ROS. However, unexpectedly, the common myeloid progenitors (CMPs) produce significantly increased levels of ROS2. The functional significance of this difference in ROS level in the two progenitor types remains unresolved2,3. Here we show that Drosophila multipotent haematopoietic progenitors, which are largely akin to the mammalian myeloid progenitors4, display increased levels of ROS under in vivo physiological conditions, which are downregulated on differentiation. Scavenging the ROS from these haematopoietic progenitors by using in vivo genetic tools retards their differentiation into mature blood cells. Conversely, increasing the haematopoietic progenitor ROS beyond their basal level triggers precocious differentiation into all three mature blood cell types found in Drosophila, through a signalling pathway that involves JNK and FoxO activation as well as Polycomb downregulation. We conclude that the developmentally regulated, moderately high ROS level in the progenitor population sensitizes them to differentiation, and establishes a signalling role for ROS in the regulation of haematopoietic cell fate. Our results lead to a model that could be extended to reveal a probable signalling role for ROS in the differentiation of CMPs in mammalian haematopoietic development and oxidative stress response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Finkel, T. & Holbrook, N. J. Oxidants, oxidative stress and the biology of ageing. Nature 408, 239–247 (2000)
Tothova, Z. et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell 128, 325–339 (2007)
Coffer, P. J. & Burgering, B. M. Stressed marrow: FoxOs stem tumour growth. Nature Cell Biol. 9, 251–253 (2007)
Evans, C. J., Hartenstein, V. & Banerjee, U. Thicker than blood: conserved mechanisms in Drosophila and vertebrate hematopoiesis. Dev. Cell 5, 673–690 (2003)
Jung, S. H., Evans, C. J., Uemura, C. & Banerjee, U. The Drosophila lymph gland as a developmental model of hematopoiesis. Development 132, 2521–2533 (2005)
Wood, W. & Jacinto, A. Drosophila melanogaster embryonic haemocytes: masters of multitasking. Nature Rev. Mol. Cell Biol. 8, 542–551 (2007)
Lebestky, T., Jung, S. H. & Banerjee, U. A. Serrate-expressing signaling center controls Drosophila hematopoiesis. Genes Dev. 17, 348–353 (2003)
Mandal, L., Martinez-Agosto, J. A., Evans, C. J., Hartenstein, V. & Banerjee, U. A. Hedgehog- and Antennapedia-dependent niche maintains Drosophila haematopoietic precursors. Nature 446, 320–324 (2007)
Krzemien, J. et al. Control of blood cell homeostasis in Drosophila larvae by the posterior signalling centre. Nature 446, 325–328 (2007)
Lebestky, T., Chang, T., Hartenstein, V. & Banerjee, U. Specification of Drosophila hematopoietic lineage by conserved transcription factors. Science 288, 146–149 (2000)
Bruckner, K. et al. The PDGF/VEGF receptor controls blood cell survival in Drosophila . Dev. Cell 7, 73–84 (2004)
Missirlis, F. et al. A putative glutathione peroxidase of Drosophila encodes a thioredoxin peroxidase that provides resistance against oxidative stress but fails to complement a lack of catalase activity. Biol. Chem. 384, 463–472 (2003)
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993)
Owusu-Ansah, E., Yavari, A., Mandal, S. & Banerjee, U. Distinct mitochondrial retrograde signals control the G1-S cell cycle checkpoint. Nature Genet. 40, 356–361 (2008)
Owusu-Ansah, E., Yavari, A. . & Banerjee, U. . A protocol for in vivo detection of reactive oxygen species. Nature Protocols 10.1038/nprot.2008.23 (2008)
Gotoh, Y. & Cooper, J. A. Reactive oxygen species- and dimerization-induced activation of apoptosis signal-regulating kinase 1 in tumor necrosis factor-α signal transduction. J. Biol. Chem. 273, 17477–17482 (1998)
Essers, M. A. et al. FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J. 23, 4802–4812 (2004)
Martin-Blanco, E. et al. puckered encodes a phosphatase that mediates a feedback loop regulating JNK activity during dorsal closure in Drosophila . Genes Dev. 12, 557–570 (1998)
Harvey, K. F. et al. FOXO-regulated transcription restricts overgrowth of Tsc mutant organs. J. Cell Biol. 180, 691–696 (2008)
Wang, M. C., Bohmann, D. & Jasper, H. JNK extends life span and limits growth by antagonizing cellular and organism-wide responses to insulin signaling. Cell 121, 115–125 (2005)
Lee, N., Maurange, C., Ringrose, L. & Paro, R. Suppression of Polycomb group proteins by JNK signalling induces transdetermination in Drosophila imaginal discs. Nature 438, 234–237 (2005)
Puig, O., Marr, M. T., Ruhf, M. L. & Tjian, R. Control of cell number by Drosophila FOXO: downstream and feedback regulation of the insulin receptor pathway. Genes Dev. 17, 2006–2020 (2003)
Teleman, A. A., Hietakangas, V., Sayadian, A. C. & Cohen, S. M. Nutritional control of protein biosynthetic capacity by insulin via Myc in Drosophila . Cell Metab. 7, 21–32 (2008)
Junger, M. A. et al. The Drosophila forkhead transcription factor FOXO mediates the reduction in cell number associated with reduced insulin signaling. J. Biol. 2, 20 (2003)
Takeda, S. et al. Local positive feedback regulation determines cell shape in root hair cells. Science 319, 1241–1244 (2008)
Foreman, J. et al. Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422, 442–446 (2003)
Acknowledgements
We thank I. Ando and H. Muller for antibodies, and E. Hafen, A. Martinez-Arias, F. Missirlis, S. Noselli, R. Paro, S Sinenko, the National Institute of Genetics Fly Stock Center (Japan) and the Bloomington Stock Center for fly stocks. We acknowledge M. Kulkarni and C. Pitsouli of the Perrimon laboratory for technical assistance. Owing to space limitations, we apologize to our colleagues whose work is not referenced. This study was supported by US National Institutes of Health grant R01HL067395 to U.B. and a T32 institutional postdoctoral fellowship T32-HL069766 to E.O.-A.
Author Contributions U.B. supervised the project. E.O.-A. conceived, designed and performed all experiments. E.O.-A. and U.B. discussed results and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S6 and Supplementary Table 1. (PDF 7364 kb)
Rights and permissions
About this article
Cite this article
Owusu-Ansah, E., Banerjee, U. Reactive oxygen species prime Drosophila haematopoietic progenitors for differentiation. Nature 461, 537–541 (2009). https://doi.org/10.1038/nature08313
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08313
This article is cited by
-
Mitochondrial perturbation in immune cells enhances cell-mediated innate immunity in Drosophila
BMC Biology (2024)
-
Oleoylethanolamide protects mesenchymal stem/stromal cells (MSCs) from oxidative stress and reduces adipogenic related genes expression in adipose-derived MSCs undergoing adipocyte differentiation
Molecular Biology Reports (2024)
-
Emerging roles of mitochondrial functions and epigenetic changes in the modulation of stem cell fate
Cellular and Molecular Life Sciences (2024)
-
Monoamine oxidase A-dependent ROS formation modulates human cardiomyocyte differentiation through AKT and WNT activation
Basic Research in Cardiology (2023)
-
Dysregulation of SOX17/NRF2 axis confers chemoradiotherapy resistance and emerges as a novel therapeutic target in esophageal squamous cell carcinoma
Journal of Biomedical Science (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.