Abstract
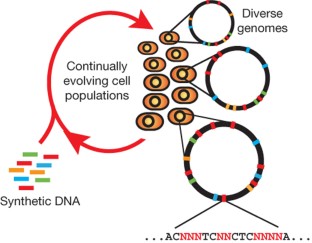
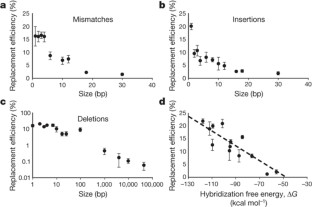
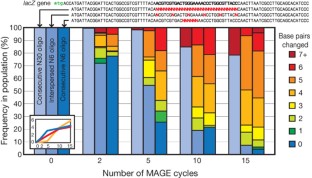
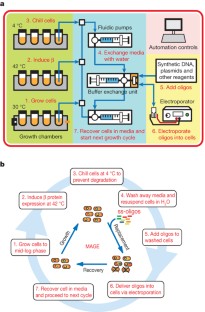
The breadth of genomic diversity found among organisms in nature allows populations to adapt to diverse environments1,2. However, genomic diversity is difficult to generate in the laboratory and new phenotypes do not easily arise on practical timescales3. Although in vitro and directed evolution methods4,5,6,7,8,9 have created genetic variants with usefully altered phenotypes, these methods are limited to laborious and serial manipulation of single genes and are not used for parallel and continuous directed evolution of gene networks or genomes. Here, we describe multiplex automated genome engineering (MAGE) for large-scale programming and evolution of cells. MAGE simultaneously targets many locations on the chromosome for modification in a single cell or across a population of cells, thus producing combinatorial genomic diversity. Because the process is cyclical and scalable, we constructed prototype devices that automate the MAGE technology to facilitate rapid and continuous generation of a diverse set of genetic changes (mismatches, insertions, deletions). We applied MAGE to optimize the 1-deoxy-d-xylulose-5-phosphate (DXP) biosynthesis pathway in Escherichia coli to overproduce the industrially important isoprenoid lycopene. Twenty-four genetic components in the DXP pathway were modified simultaneously using a complex pool of synthetic DNA, creating over 4.3 billion combinatorial genomic variants per day. We isolated variants with more than fivefold increase in lycopene production within 3 days, a significant improvement over existing metabolic engineering techniques. Our multiplex approach embraces engineering in the context of evolution by expediting the design and evolution of organisms with new and improved properties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Venter, J. C. et al. Environmental genome shotgun sequencing of the Sargasso Sea. Science 304, 66–74 (2004)
Tringe, S. G. et al. Comparative metagenomics of microbial communities. Science 308, 554–557 (2005)
Elena, S. F. & Lenski, R. E. Evolution experiments with microorganisms: the dynamics and genetic bases of adaptation. Nature Rev. Genet. 4, 457–469 (2003)
Ellington, A. D. & Szostak, J. W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990)
Crameri, A., Raillard, S.-A., Bermudez, E., Stemmer, W. P. & C DNA shuffling of a family of genes from diverse species accelerates directed evolution. Nature 391, 288–291 (1998)
Joo, H., Lin, Z. & Arnold, F. H. Laboratory evolution of peroxide-mediated cytochrome P450 hydroxylation. Nature 399, 670–673 (1999)
Zhang, Y. X. et al. Genome shuffling leads to rapid phenotypic improvement in bacteria. Nature 415, 644–646 (2002)
Pfleger, B. F., Pitera, D. J., Smolke, C. D. & Keasling, J. D. Combinatorial engineering of intergenic regions in operons tunes expression of multiple genes. Nature Biotechnol. 24, 1027–1032 (2006)
Cadwell, R. C. & Joyce, G. F. Randomization of genes by PCR mutagenesis. PCR Methods Appl. 2, 28–33 (1992)
Shendure, J. et al. Accurate multiplex polony sequencing of an evolved bacterial genome. Science 309, 1728–1732 (2005)
Zhang, Y., Buchholz, F., Muyrers, J. P. & Stewart, A. F. A new logic for DNA engineering using recombination in Escherichia coli . Nature Genet. 20, 123–128 (1998)
Costantino, N. & Court, D. L. Enhanced levels of λ Red-mediated recombinants in mismatch repair mutants. Proc. Natl Acad. Sci. USA 100, 15748–15753 (2003)
Sharan, S. K., Thomason, L. C., Kuznetsov, S. G. & Court, D. L. Recombineering: a homologous recombination-based method of genetic engineering. Nature Protocols 4, 206–223 (2009)
Ellis, H. M., Yu, D., DiTizio, T. & Court, D. L. High efficiency mutagenesis, repair, and engineering of chromosomal DNA using single-stranded oligonucleotides. Proc. Natl Acad. Sci. USA 98, 6742–6746 (2001)
Markham, N. R. & Zuker, M. DINAMelt web server for nucleic acid melting prediction. Nucleic Acids Res. 33, W577–W581 (2005)
Jin, Y. S. & Stephanopoulos, G. Multi-dimensional gene target search for improving lycopene biosynthesis in Escherichia coli . Metab. Eng. 9, 337–347 (2007)
Kang, M. J. et al. Identification of genes affecting lycopene accumulation in Escherichia coli using a shot-gun method. Biotechnol. Bioeng. 91, 636–642 (2005)
Chen, H., Bjerknes, M., Kumar, R. & Jay, E. Determination of the optimal aligned spacing between the Shine – Dalgarno sequence and the translation initiation codon of Escherichia coli mRNAs. Nucleic Acids Res. 22, 4953–4957 (1994)
Alper, H., Jin, Y. S., Moxley, J. F. & Stephanopoulos, G. Identifying gene targets for the metabolic engineering of lycopene biosynthesis in Escherichia coli . Metab. Eng. 7, 155–164 (2005)
Alper, H., Miyaoku, K. & Stephanopoulos, G. Construction of lycopene-overproducing E. coli strains by combining systematic and combinatorial gene knockout targets. Nature Biotechnol. 23, 612–616 (2005)
Farmer, W. R. & Liao, J. C. Precursor balancing for metabolic engineering of lycopene production in Escherichia coli . Biotechnol. Prog. 17, 57–61 (2001)
Kim, S. W. & Keasling, J. D. Metabolic engineering of the nonmevalonate isopentenyl diphosphate synthesis pathway in Escherichia coli enhances lycopene production. Biotechnol. Bioeng. 72, 408–415 (2001)
Yuan, L. Z., Rouviere, P. E., Larossa, R. A. & Suh, W. Chromosomal promoter replacement of the isoprenoid pathway for enhancing carotenoid production in E. coli . Metab. Eng. 8, 79–90 (2006)
Khosla, C. & Keasling, J. D. Metabolic engineering for drug discovery and development. Nature Rev. Drug Discov. 2, 1019–1025 (2003)
Cropp, T. A. & Schultz, P. G. An expanding genetic code. Trends Genet. 20, 625–630 (2004)
Gibson, D. G. et al. Complete chemical synthesis, assembly, and cloning of a Mycoplasma genitalium genome. Science 319, 1215–1220 (2008)
Metzgar, D. et al. Acinetobacter sp. ADP1: an ideal model organism for genetic analysis and genome engineering. Nucleic Acids Res. 32, 5780–5790 (2004)
Nakayama, M. & Ohara, O. Improvement of recombination efficiency by mutation of Red proteins. Biotechniques 38, 917–924 (2005)
Datta, S., Costantino, N., Zhou, X. & Court, D. L. Identification and analysis of recombineering functions from Gram-negative and Gram-positive bacteria and their phages. Proc. Natl Acad. Sci. USA 105, 1626–1631 (2008)
Tian, J. et al. Accurate multiplex gene synthesis from programmable DNA microchips. Nature 432, 1050–1054 (2004)
Yu, D. et al. An efficient recombination system for chromosome engineering in Escherichia coli . Proc. Natl Acad. Sci. USA 97, 5978–5983 (2000)
Cunningham, F. X., Sun, Z., Chamovitz, D., Hirschberg, J. & Gantt, E. Molecular structure and enzymatic function of lycopene cyclase from the cyanobacterium Synechococcus sp strain PCC7942. Plant Cell 6, 1107–1121 (1994)
Acknowledgements
We are grateful to J. Jacobson for his insights and advice throughout this work. We thank D. Court for his insights and sharing strain DY330, N. Reppas for advice and sharing strain EcNR2, F. X. Cunningham for sharing pAC-LYC, and B. H. Sterling for assistance in constructing the EcFI5 strain. We also thank M. Jewett, J. Aach, D. Bang, S. Kosuri and members of the Church laboratory for advice and discussions. We thank the NSF, DOE, DARPA, the Wyss Institute for Biologically Inspired Engineering and training fellowships from the NIH and NDSEG (H.H.W.) for supporting this research.
Author Contributions H.H.W., F.J.I. and G.M.C. conceived the study jointly with P.A.C.; H.H.W. and F.J.I. designed and performed experiments with assistance from P.A.C., Z.Z.S., G.X. and C.R.F.; H.H.W. and F.J.I. wrote the manuscript; G.M.C. supervised all aspects of the study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
we wish to disclose that three authors (G.M.C., H.H.W, F.J.I.) have a pending patent application whose value may be affected by the publication of this paper. G.M.C. also discloses various associations with companies as outlined at http://arep.med.harvard.edu/gmc/tech.html.
Supplementary information
Supplementary Information
This file contains Supplementary Data, Supplementary References, Supplementary Figures 1-3 with Legends and Supplementary Tables 1-3. (PDF 311 kb)
Rights and permissions
About this article
Cite this article
Wang, H., Isaacs, F., Carr, P. et al. Programming cells by multiplex genome engineering and accelerated evolution. Nature 460, 894–898 (2009). https://doi.org/10.1038/nature08187
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08187
This article is cited by
-
Competition-driven eco-evolutionary feedback reshapes bacteriophage lambda’s fitness landscape and enables speciation
Nature Communications (2024)
-
Bacterial genome engineering using CRISPR-associated transposases
Nature Protocols (2024)
-
The non-mevalonate pathway requires a delicate balance of intermediates to maximize terpene production
Applied Microbiology and Biotechnology (2024)
-
Biosynthesis pathways of expanding carbon chains for producing advanced biofuels
Biotechnology for Biofuels and Bioproducts (2023)
-
Precise cut-and-paste DNA insertion using engineered type V-K CRISPR-associated transposases
Nature Biotechnology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.



