Abstract
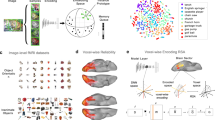
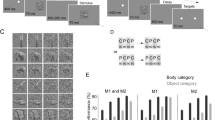
The visual system has an extraordinary capability to extract categorical information from complex natural scenes. For example, subjects are able to rapidly detect the presence of object categories such as animals or vehicles in new scenes that are presented very briefly1,2. This is even true when subjects do not pay attention to the scenes and simultaneously perform an unrelated attentionally demanding task3, a stark contrast to the capacity limitations predicted by most theories of visual attention4,5. Here we show a neural basis for rapid natural scene categorization in the visual cortex, using functional magnetic resonance imaging and an object categorization task in which subjects detected the presence of people or cars in briefly presented natural scenes. The multi-voxel pattern of neural activity in the object-selective cortex evoked by the natural scenes contained information about the presence of the target category, even when the scenes were task-irrelevant and presented outside the focus of spatial attention. These findings indicate that the rapid detection of categorical information in natural scenes is mediated by a category-specific biasing mechanism in object-selective cortex that operates in parallel across the visual field, and biases information processing in favour of objects belonging to the target object category.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Potter, M. C. Meaning in visual search. Science 187, 965–966 (1975)
Thorpe, S., Fize, D. & Marlot, C. Speed of processing in the human visual system. Nature 381, 520–522 (1996)
Li, F. F., VanRullen, R., Koch, C. & Perona, P. Rapid natural scene categorization in the near absence of attention. Proc. Natl Acad. Sci. USA 99, 9596–9601 (2002)
Broadbent, D. E. Perception and Communication (Oxford Univ. Press, 1958)
Treisman, A. M. & Gelade, G. A feature-integration theory of attention. Cognit. Psychol. 12, 97–136 (1980)
Fei-Fei, L., VanRullen, R., Koch, C. & Perona, P. Why does natural scene categorization require little attention? Exploring attentional requirements for natural and synthetic stimuli. Vis. Cogn. 12, 893–924 (2005)
Rousselet, G. A., Fabre-Thorpe, M. & Thorpe, S. J. Parallel processing in high-level categorization of natural images. Nature Neurosci. 5, 629–630 (2002)
Wojciulik, E., Kanwisher, N. & Driver, J. Covert visual attention modulates face-specific activity in the human fusiform gyrus: fMRI study. J. Neurophysiol. 79, 1574–1578 (1998)
Cox, D. D. & Savoy, R. L. Functional magnetic resonance imaging (fMRI) “brain reading”: detecting and classifying distributed patterns of fMRI activity in human visual cortex. Neuroimage 19, 261–270 (2003)
Haxby, J. V. et al. Distributed and overlapping representations of faces and objects in ventral temporal cortex. Science 293, 2425–2430 (2001)
Malach, R. et al. Object-related activity revealed by functional magnetic resonance imaging in human occipital cortex. Proc. Natl Acad. Sci. USA 92, 8135–8139 (1995)
Kriegeskorte, N., Goebel, R. & Bandettini, P. Information-based functional brain mapping. Proc. Natl Acad. Sci. USA 103, 3863–3868 (2006)
Maunsell, J. H. & Treue, S. Feature-based attention in visual cortex. Trends Neurosci. 29, 317–322 (2006)
Motter, B. C. Neural correlates of attentive selection for color or luminance in extrastriate area V4. J. Neurosci. 14, 2178–2189 (1994)
Saenz, M., Buracas, G. T. & Boynton, G. M. Global effects of feature-based attention in human visual cortex. Nature Neurosci. 5, 631–632 (2002)
Chelazzi, L., Miller, E. K., Duncan, J. & Desimone, R. A neural basis for visual search in inferior temporal cortex. Nature 363, 345–347 (1993)
Duncan, J. EPS mid-career award 2004: brain mechanisms of attention. Q. J. Exp. Psychol. (Colchester) 59, 2–27 (2006)
Duncan, J. & Humphreys, G. W. Visual search and stimulus similarity. Psychol. Rev. 96, 433–458 (1989)
O’Regan, J. K., Rensink, R. A. & Clark, J. J. Change-blindness as a result of ‘mudsplashes’. Nature 398, 34 (1999)
Rensink, R. A., O’Regan, J. K. & Clark, J. J. To see or not to see: the need for attention to perceive changes in scenes. Psychol. Sci. 8, 368–373 (1997)
Simons, D. J. & Rensink, R. A. Change blindness: past, present, and future. Trends Cogn. Sci. 9, 16–20 (2005)
O’Regan, J. K. Solving the “real” mysteries of visual perception: the world as an outside memory. Can. J. Psychol. 46, 461–488 (1992)
Russell, B. C., Torralba, A., Murphy, K. P. & Freeman, W. T. LabelMe: a database and web-based tool for image annotation. Int. J. Comput. Vis. 77, 157–173 (2008)
Walther, D. B., Caddigan, E., Fei-Fei, L. & Beck, D. M. Natural scene categories revealed in distributed patterns of activity in the human brain. J. Neurosci. (in the press)
Cox, R. W. & Hyde, J. S. AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. 29, 162–173 (1996)
Acknowledgements
We thank P. Downing, U. Hasson, S. McMains, K. Norman, T. Stein and A. Treisman for suggestions on earlier versions of the manuscript, members of the Kastner laboratory for help with retinotopic mapping, and N. Oosterhof for help with data analysis. This work was supported by grants from the National Institutes of Health (2RO1 MH64043, 1RO1 EY017699, 2P50 MH–62196) to S.K., and a Microsoft Research New Faculty Fellowship and the F. Moss gift to L.F.-F.
Author Contributions M.V.P., in collaboration with L.F.-F. and S.K., conceived the experiment. M.V.P. acquired and analysed the data. M.V.P., L.F.-F. and S.K. interpreted the results and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-2 with Legends, Supplementary Table 1, Supplementary Methods, a Supplementary Discussion and Supplementary References. (PDF 1690 kb)
Rights and permissions
About this article
Cite this article
Peelen, M., Fei-Fei, L. & Kastner, S. Neural mechanisms of rapid natural scene categorization in human visual cortex. Nature 460, 94–97 (2009). https://doi.org/10.1038/nature08103
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08103
This article is cited by
-
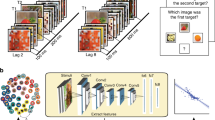
Brain-inspired models for visual object recognition: an overview
Artificial Intelligence Review (2022)
-
Attention expedites target selection by prioritizing the neural processing of distractor features
Communications Biology (2021)
-
Identifying task-relevant spectral signatures of perceptual categorization in the human cortex
Scientific Reports (2020)
-
Psychological potential field and human eye fixation on binary line-drawing images: A comparative experimental study
Computational Visual Media (2020)
-
Machine vision benefits from human contextual expectations
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.