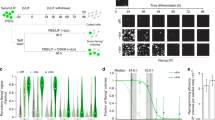
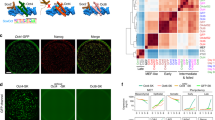
Abstract
Pluripotency of embryonic stem (ES) cells is controlled by defined transcription factors1,2. During differentiation, mouse ES cells undergo global epigenetic reprogramming, as exemplified by X-chromosome inactivation (XCI) in which one female X chromosome is silenced to achieve gene dosage parity between the sexes3,4,5. Somatic XCI is regulated by homologous X-chromosome pairing6,7 and counting8,9,10, and by the random choice of future active and inactive X chromosomes. XCI and cell differentiation are tightly coupled11, as blocking one process compromises the other8,12 and dedifferentiation of somatic cells to induced pluripotent stem cells is accompanied by X chromosome reactivation2. Recent evidence suggests coupling of Xist expression to pluripotency factors occurs13, but how the two are interconnected remains unknown. Here we show that Oct4 (also known as Pou5f1)14 lies at the top of the XCI hierarchy, and regulates XCI by triggering X-chromosome pairing and counting. Oct4 directly binds Tsix and Xite, two regulatory noncoding RNA genes of the X-inactivation centre15,16, and also complexes with XCI trans-factors, Ctcf and Yy1 (ref. 17), through protein–protein interactions. Depletion of Oct4 blocks homologous X-chromosome pairing and results in the inactivation of both X chromosomes in female cells. Thus, we have identified the first trans-factor that regulates counting, and ascribed new functions to Oct4 during X-chromosome reprogramming.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006)
Maherali, N. et al. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell 1, 55–70 (2007)
Wutz, A. & Gribnau, J. X inactivation Xplained. Curr. Opin. Genet. Dev. 17, 387–393 (2007)
Lucchesi, J. C., Kelly, W. G. & Panning, B. Chromatin remodeling in dosage compensation. Annu. Rev. Genet. 39, 615–651 (2005)
Payer, B. & Lee, J. T. X chromosome dosage compensation: how mammals keep the balance. Annu. Rev. Genet. 42, 733–772 (2008)
Xu, N., Tsai, C. L. & Lee, J. T. Transient homologous chromosome pairing marks the onset of X inactivation. Science 311, 1149–1152 (2006)
Bacher, C. P. et al. Transient colocalization of X-inactivation centres accompanies the initiation of X inactivation. Nature Cell Biol. 8, 293–299 (2006)
Lee, J. T. Regulation of X-chromosome counting by Tsix and Xite sequences. Science 309, 768–771 (2005)
Morey, C. et al. The region 3′ to Xist mediates X chromosome counting and H3 Lys-4 dimethylation within the Xist gene. EMBO J. 23, 594–604 (2004)
Monkhorst, K., Jonkers, I., Rentmeester, E., Grosveld, F. & Gribnau, J. X inactivation counting and choice is a stochastic process: evidence for involvement of an X-linked activator. Cell 132, 410–421 (2008)
Monk, M. & Harper, M. I. Sequential X chromosome inactivation coupled with cellular differentiation in early mouse embryos. Nature 281, 311–313 (1979)
Silva, S. S., Rowntree, R. K., Mekhoubad, S. & Lee, J. T. X-chromosome inactivation and epigenetic fluidity in human embryonic stem cells. Proc. Natl Acad. Sci. USA 105, 4820–4825 (2008)
Navarro, P. et al. Molecular coupling of Xist regulation and pluripotency. Science 321, 1693–1695 (2008)
Nichols, J. et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell 95, 379–391 (1998)
Lee, J. T. & Lu, N. Targeted mutagenesis of Tsix leads to nonrandom X inactivation. Cell 99, 47–57 (1999)
Ogawa, Y. & Lee, J. T. Xite, X-inactivation intergenic transcription elements that regulate the probability of choice. Mol. Cell 11, 731–743 (2003)
Donohoe, M. E., Zhang, L. F., Xu, N., Shi, Y. & Lee, J. T. Identification of a Ctcf cofactor, Yy1, for the X chromosome binary switch. Mol. Cell 25, 43–56 (2007)
Stavropoulos, N., Rowntree, R. K. & Lee, J. T. Identification of developmentally specific enhancers for Tsix in the regulation of X chromosome inactivation. Mol. Cell. Biol. 25, 2757–2769 (2005)
Chao, W., Huynh, K. D., Spencer, R. J., Davidow, L. S. & Lee, J. T. CTCF, a candidate trans-acting factor for X-inactivation choice. Science 295, 345–347 (2002)
Tsai, C. L., Rowntree, R. K., Cohen, D. E. & Lee, J. T. Higher order chromatin structure at the X-inactivation center via looping DNA. Dev. Biol. 319, 416–425 (2008)
Ohlsson, R., Renkawitz, R. & Lobanenkov, V. V. CTCF is a uniquely versatile transcription regulator linked to epigenetics and disease. Trends Genet. 17, 520–527 (2001)
West, A. G., Gaszner, M. & Felsenfeld, G. Insulators: many functions, many mechanisms. Genes Dev. 16, 271–288 (2002)
Park, K. & Atchison, M. L. Isolation of a candidate repressor/activator, NF-E1 (YY-1, δ), that binds to the immunoglobulin κ 3′ enhancer and the immunoglobulin heavy-chain μE1 site. Proc. Natl Acad. Sci. USA 88, 9804–9808 (1991)
Shi, Y., Lee, J. S. & Galvin, K. M. Everything you have ever wanted to know about Yin Yang 1. Biochim. Biophys. Acta 1332, F49–F66 (1997)
Xu, N., Donohoe, M. E., Silva, S. S. & Lee, J. T. Evidence that homologous X-chromosome pairing requires transcription and Ctcf protein. Nature Genet. 39, 1390–1396 (2007)
Avner, P. & Heard, E. X-chromosome inactivation: counting, choice and initiation. Nature Rev. Genet. 2, 59–67 (2001)
Lee, T. I., Johnstone, S. E. & Young, R. A. Chromatin immunoprecipitation and microarray-based analysis of protein location. Nature Protocols 1, 729–748 (2006)
Chen, X. et al. Integration of external signaling pathways with the core transcriptional network in embryonic stem cells. Cell 133, 1106–1117 (2008)
Stavropoulos, N., Lu, N. & Lee, J. T. A functional role for Tsix transcription in blocking Xist RNA accumulation but not in X-chromosome choice. Proc. Natl Acad. Sci. USA 98, 10232–10237 (2001)
Toyooka, Y., Shimosato, Y., Murakami, K., Takahashi. K & Niwa, H. Identification and characterization of subpopulations in undifferentiated ES cell culture. Development 135, 909–913 (2008)
Acknowledgements
We thank N. Stavropoulos and J. Lopilato for their contributions to the early phase of this work. We also thank J. Zhao, J. A. Erwin and J. Ahn for critical reading of the manuscript and all laboratory members for stimulating discussion. This work was funded by a National Institutes of Health grant to J.T.L. (GM58839). J.T.L. is an Investigator of the Howard Hughes Medical Institute.
Author Contributions M.E.D. designed, performed and analysed the Xite/Tsix bioinformatics screen, EMSA, tests of protein–protein interactions, luciferase assays, knockdowns and qRT–PCR; S.S.S. designed, performed and analysed the pairing and counting experiments; and S.F.P. designed, performed and analysed the Xist bioinformatics screen and the quantitative ChIP assays. N.X. participated in initial pairing analyses. J.T.L. formulated, analysed and directed the study and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
This file contains Supplementary Figures S1-S4 with Legends. (PDF 642 kb)
Rights and permissions
About this article
Cite this article
Donohoe, M., Silva, S., Pinter, S. et al. The pluripotency factor Oct4 interacts with Ctcf and also controls X-chromosome pairing and counting. Nature 460, 128–132 (2009). https://doi.org/10.1038/nature08098
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08098
This article is cited by
-
GATA transcription factors drive initial Xist upregulation after fertilization through direct activation of long-range enhancers
Nature Cell Biology (2023)
-
Gene regulation in time and space during X-chromosome inactivation
Nature Reviews Molecular Cell Biology (2022)
-
Integrated analysis of Xist upregulation and X-chromosome inactivation with single-cell and single-allele resolution
Nature Communications (2021)
-
SPEN is required for Xist upregulation during initiation of X chromosome inactivation
Nature Communications (2021)
-
Functions and properties of nuclear lncRNAs—from systematically mapping the interactomes of lncRNAs
Journal of Biomedical Science (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.