Abstract
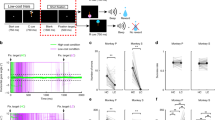
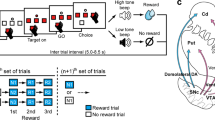
Midbrain dopamine neurons are activated by reward or sensory stimuli predicting reward1,2,3,4. These excitatory responses increase as the reward value increases5. This response property has led to a hypothesis that dopamine neurons encode value-related signals and are inhibited by aversive events. Here we show that this is true only for a subset of dopamine neurons. We recorded the activity of dopamine neurons in monkeys (Macaca mulatta) during a Pavlovian procedure with appetitive and aversive outcomes (liquid rewards and airpuffs directed at the face, respectively). We found that some dopamine neurons were excited by reward-predicting stimuli and inhibited by airpuff-predicting stimuli, as the value hypothesis predicts. However, a greater number of dopamine neurons were excited by both of these stimuli, inconsistent with the hypothesis. Some dopamine neurons were also excited by both rewards and airpuffs themselves, especially when they were unpredictable. Neurons excited by the airpuff-predicting stimuli were located more dorsolaterally in the substantia nigra pars compacta, whereas neurons inhibited by the stimuli were located more ventromedially, some in the ventral tegmental area. A similar anatomical difference was observed for their responses to actual airpuffs. These findings suggest that different groups of dopamine neurons convey motivational signals in distinct manners.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schultz, W. Predictive reward signal of dopamine neurons. J. Neurophysiol. 80, 1–27 (1998)
Satoh, T., Nakai, S., Sato, T. & Kimura, M. Correlated coding of motivation and outcome of decision by dopamine neurons. J. Neurosci. 23, 9913–9923 (2003)
Takikawa, Y., Kawagoe, R. & Hikosaka, O. A possible role of midbrain dopamine neurons in short- and long-term adaptation of saccades to position-reward mapping. J. Neurophysiol. 92, 2520–2529 (2004)
Morris, G., Arkadir, D., Nevet, A., Vaadia, E. & Bergman, H. Coincident but distinct messages of midbrain dopamine and striatal tonically active neurons. Neuron 43, 133–143 (2004)
Tobler, P. N., Fiorillo, C. D. & Schultz, W. Adaptive coding of reward value by dopamine neurons. Science 307, 1642–1645 (2005)
Ungless, M. A., Magill, P. J. & Bolam, J. P. Uniform inhibition of dopamine neurons in the ventral tegmental area by aversive stimuli. Science 303, 2040–2042 (2004)
Chiodo, L. A., Antelman, S. M., Caggiula, A. R. & Lineberry, C. G. Sensory stimuli alter the discharge rate of dopamine (DA) neurons: evidence for two functional types of DA cells in the substantia nigra. Brain Res. 189, 544–549 (1980)
Coizet, V., Dommett, E. J., Redgrave, P. & Overton, P. G. Nociceptive responses of midbrain dopaminergic neurones are modulated by the superior colliculus in the rat. Neuroscience 139, 1479–1493 (2006)
Schultz, W. & Romo, R. Responses of nigrostriatal dopamine neurons to high-intensity somatosensory stimulation in the anesthetized monkey. J. Neurophysiol. 57, 201–217 (1987)
Mantz, J., Thierry, A. M. & Glowinski, J. Effect of noxious tail pinch on the discharge rate of mesocortical and mesolimbic dopamine neurons: selective activation of the mesocortical system. Brain Res. 476, 377–381 (1989)
Guarraci, F. A. & Kapp, B. S. An electrophysiological characterization of ventral tegmental area dopaminergic neurons during differential Pavlovian fear conditioning in the awake rabbit. Behav. Brain Res. 99, 169–179 (1999)
Mirenowicz, J. & Schultz, W. Preferential activation of midbrain dopamine neurons by appetitive rather than aversive stimuli. Nature 379, 449–451 (1996)
Joshua, M., Adler, A., Mitelman, R., Vaadia, E. & Bergman, H. Midbrain dopaminergic neurons and striatal cholinergic interneurons encode the difference between reward and aversive events at different epochs of probabilistic classical conditioning trials. J. Neurosci. 28, 11673–11684 (2008)
Schultz, W., Dayan, P. & Montague, P. R. A neural substrate of prediction and reward. Science 275, 1593–1599 (1997)
Montague, P. R., Dayan, P. & Sejnowski, T. J. A framework for mesencephalic dopamine systems based on predictive Hebbian learning. J. Neurosci. 16, 1936–1947 (1996)
Lynd-Balta, E. & Haber, S. N. The organization of midbrain projections to the striatum in the primate: sensorimotor-related striatum versus ventral striatum. Neuroscience 59, 625–640 (1994)
Ikemoto, S. Dopamine reward circuitry: two projection systems from the ventral midbrain to the nucleus accumbens–olfactory tubercle complex. Brain Res. Rev. 56, 27–78 (2007)
Knutson, B., Adams, C. M., Fong, G. W. & Hommer, D. Anticipation of increasing monetary reward selectively recruits nucleus accumbens. J. Neurosci. 21, RC159 (2001)
Cromwell, H. C. & Schultz, W. Effects of expectations for different reward magnitudes on neuronal activity in primate striatum. J. Neurophysiol. 89, 2823–2838 (2003)
Schultz, W., Apicella, P., Scarnati, E. & Ljungberg, T. Neuronal activity in monkey ventral striatum related to the expectation of reward. J. Neurosci. 12, 4595–4610 (1992)
Kitama, T., Ohno, T., Tanaka, M., Tsubokawa, H. & Yoshida, K. Stimulation of the caudate nucleus induces contraversive saccadic eye movements as well as head turning in the cat. Neurosci. Res. 12, 287–292 (1991)
Hikosaka, O., Takikawa, Y. & Kawagoe, R. Role of the basal ganglia in the control of purposive saccadic eye movements. Physiol. Rev. 80, 953–978 (2000)
Carli, M., Evenden, J. L. & Robbins, T. W. Depletion of unilateral striatal dopamine impairs initiation of contralateral actions and not sensory attention. Nature 313, 679–682 (1985)
Holland, P. C. & Gallagher, M. Amygdala circuitry in attentional and representational processes. Trends Cogn. Sci. 3, 65–73 (1999)
Lin, S. C. & Nicolelis, M. A. Neuronal ensemble bursting in the basal forebrain encodes salience irrespective of valence. Neuron 59, 138–149 (2008)
Richardson, R. T. & DeLong, M. R. Electrophysiological studies of the functions of the nucleus basalis in primates. Adv. Exp. Med. Biol. 295, 233–252 (1991)
Matsumoto, M. & Hikosaka, O. Representation of negative motivational value in the primate lateral habenula. Nature Neurosci. 12, 77–84 (2009)
Matsumoto, M. & Hikosaka, O. Lateral habenula as a source of negative reward signals in dopamine neurons. Nature 447, 1111–1115 (2007)
Davies, R. M., Gerstein, G. L. & Baker, S. N. Measurement of time-dependent changes in the irregularity of neural spiking. J. Neurophysiol. 96, 906–918 (2006)
Acknowledgements
We thank S. Hong, E. Bromberg-Martin, M. Yasuda, S. Yamamoto and Y. Tachibana for discussion, M. K. Smith for his histological expertise, J. W. McClurkin, A. M. Nichols, T. W. Ruffner, A. V. Hays and L. P. Jensen for technical assistance, and G. Tansey, D. Parker and B. Nagy for animal care. This research was supported by the Intramural Research Program at the National Institutes of Health, National Eye Institute.
Author Contributions M.M. designed the Pavlovian procedure, performed the experiments and analysed the data. O.H. supported all of these processes. M.M. and O.H. discussed the results and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Notes, Supplementary References, Supplementary Tables 1-4 and Supplementary Figures 1-13 with Legends. (PDF 691 kb)
Rights and permissions
About this article
Cite this article
Matsumoto, M., Hikosaka, O. Two types of dopamine neuron distinctly convey positive and negative motivational signals. Nature 459, 837–841 (2009). https://doi.org/10.1038/nature08028
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08028
This article is cited by
-
Neural inhibition as implemented by an actor-critic model involves the human dorsal striatum and ventral tegmental area
Scientific Reports (2024)
-
Dopamine release in human associative striatum during reversal learning
Nature Communications (2024)
-
Curiosity: primate neural circuits for novelty and information seeking
Nature Reviews Neuroscience (2024)
-
Blocking D2/D3 dopamine receptors in male participants increases volatility of beliefs when learning to trust others
Nature Communications (2023)
-
A glutamatergic DRN–VTA pathway modulates neuropathic pain and comorbid anhedonia-like behavior in mice
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.