Abstract
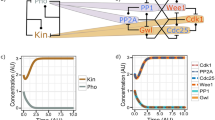
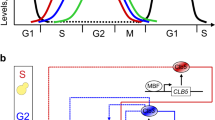
The eukaryotic cell cycle comprises an ordered series of events, orchestrated by the activity of cyclin-dependent kinases (Cdks), leading from chromosome replication during S phase to their segregation in mitosis. The unidirectionality of cell-cycle transitions is fundamental for the successful completion of this cycle. It is thought that irrevocable proteolytic degradation of key cell-cycle regulators makes cell-cycle transitions irreversible, thereby enforcing directionality1,2,3. Here we have experimentally examined the contribution of cyclin proteolysis to the irreversibility of mitotic exit, the transition from high mitotic Cdk activity back to low activity in G1. We show that forced cyclin destruction in mitotic budding yeast cells efficiently drives mitotic exit events. However, these remain reversible after termination of cyclin proteolysis, with recovery of the mitotic state and cyclin levels. Mitotic exit becomes irreversible only after longer periods of cyclin degradation, owing to activation of a double-negative feedback loop involving the Cdk inhibitor Sic1 (refs 4, 5). Quantitative modelling suggests that feedback is required to maintain low Cdk activity and to prevent cyclin resynthesis. Our findings demonstrate that the unidirectionality of mitotic exit is not the consequence of proteolysis but of systems-level feedback required to maintain the cell cycle in a new stable state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
King, R. W., Deshaies, R. J., Peters, J.-M. & Kirschner, M. W. How proteolysis drives the cell cycle. Science 274, 1652–1659 (1996)
Reed, S. I. Ratchets and clocks: the cell cycle, ubiquitylation and protein turnover. Nature Rev. Mol. Cell Biol. 4, 855–864 (2003)
Potapova, T. A. et al. The reversibility of mitotic exit in vertebrate cells. Nature 440, 954–958 (2006)
Donovan, J. D., Toyn, J. H., Johnson, A. L. & Johnston, L. H. P40 SDB25, a putative CDK inhibitor, has a role in the M/G1 transition in Saccharomyces cerevisiae . Genes Dev. 8, 1640–1653 (1994)
Visintin, R. et al. The phosphatase Cdc14 triggers mitotic exit by reversal of Cdk-dependent phosphorylation. Mol. Cell 2, 709–718 (1998)
Zachariae, W., Schwab, M., Nasmyth, K. & Seufert, W. Control of cyclin ubiquitination by CDK-regulated binding of Hct1 to the anaphase promoting complex. Science 282, 1721–1724 (1998)
Jaspersen, S. L., Charles, J. F. & Morgan, D. O. Inhibitory phosphorylation of the APC regulator Hct1 is controlled by the kinase Cdc28 and the phosphatase Cdc14. Curr. Biol. 9, 227–236 (1999)
Yeong, F. M., Lim, H. H., Padmashree, C. G. & Surana, U. Exit from mitosis in budding yeast: Biphasic inactivation of the Cdc28-Clb2 mitotic kinase and the role of Cdc20. Mol. Cell 5, 501–511 (2000)
Novak, B., Tyson, J. J., Gyorffy, B. & Csikasz-Nagy, A. Irreversible cell-cycle transitions are due to systems-level feedback. Nature Cell Biol. 9, 724–728 (2007)
Shirayama, M., Zachariae, W., Ciosk, R. & Nasmyth, K. The Polo-like kinase Cdc5p and the WD-repeat protein Cdc20p/fizzy are regulators and substrates of the anaphase promoting complex in Saccharomyces cerevisiae . EMBO J. 17, 1336–1349 (1998)
Shirayama, M., Toth, A., Galova, M. & Nasmyth, K. APCCdc20 promotes exit from mitosis by destroying the anaphase inhibitor Pds1 and cyclin Clb5. Nature 402, 203–207 (1999)
Pereira, G. & Schiebel, E. Separase regulates INCENP-Aurora B anaphase spindle function through Cdc14. Science 302, 2120–2124 (2003)
Higuchi, T. & Uhlmann, F. Stabilization of microtubule dynamics at anaphase onset promotes chromosome segregation. Nature 433, 171–176 (2005)
Khmelinskii, A., Lawrence, C., Roostalu, J. & Schiebel, E. Cdc14-regulated midzone assembly controls anaphase B. J. Cell Biol. 177, 981–993 (2007)
Juang, Y.-L. et al. APC-mediated proteolysis of Ase1 and the morphogenesis of the mitotic spindle. Science 275, 1311–1314 (1997)
Irniger, S., Piatti, S., Michaelis, C. & Nasmyth, K. Genes involved in sister chromatid separation are needed for B-type cyclin proteolysis in budding yeast. Cell 81, 269–277 (1995)
Schwab, M., Neutzner, M., Möcker, D. & Seufert, W. Yeast Hct1 recognizes the mitotic cyclin Clb2 and other substrates of the ubiquitin ligase APC. EMBO J. 20, 5165–5175 (2001)
Reynolds, D. et al. Recruitment of Thr 319-phosphorylated Ndd1p to the FHA domain of Fkh2p requires Clb kinase activity: a mechanism for CLB cluster gene activation. Genes Dev. 17, 1789–1802 (2003)
Pic-Taylor, A., Darieva, Z., Morgan, B. A. & Sharrocks, A. D. Regulation of cell cycle-specific gene expression through cyclin-dependent kinase-mediated phosphorylation of the forkhead transcription factor Fkh2p. Mol. Cell. Biol. 24, 10036–10046 (2004)
Moll, T., Tebb, G., Surana, U., Robitsch, H. & Nasmyth, K. The role of phosphorylation and the CDC28 protein kinase in cell cycle-regulated nuclear import of the S. cerevisiae transcription factor SWI5. Cell 66, 743–758 (1991)
Verma, R. et al. Phosphoregulation of Sic1p by G1 Cdk required for its degradation and entry into S phase. Science 278, 455–460 (1997)
Bishop, A. C. et al. A chemical switch for inhibitor-sensitive alleles of any protein kinase. Nature 407, 395–401 (2000)
Nguyen, V. Q., Co, C. & Li, J. J. Cyclin-dependent kinases prevent DNA re-replication through multiple mechanisms. Nature 411, 1068–1073 (2001)
Queralt, E., Lehane, C., Novak, B. & Uhlmann, F. Downregulation of PP2ACdc55 phosphatase by separase initiates mitotic exit in budding yeast. Cell 125, 719–732 (2006)
Kramer, E. R., Scheuringer, N., Podtelejnikov, A. V., Mann, M. & Peters, J.-M. Mitotic regulation of the APC activator proteins CDC20 and CDH1. Mol. Biol. Cell 11, 1555–1569 (2000)
Pomerening, J. R., Ubersax, J. A. & Ferrell, J. E. Rapid cycling and precocious termination of G1 phase in cells expressing CDK1AF. Mol. Biol. Cell 19, 3426–3441 (2008)
Cross, F. R., Archambault, V., Miller, M. & Kolvstad, M. Testing a mathematical model of the yeast cell cycle. Mol. Biol. Cell 13, 52–70 (2002)
Novak, B. & Tyson, J. J. Numerical analysis of a comprehensive model of M-phase control in Xenopus oocyte extracts and intact embryos. J. Cell Sci. 106, 1153–1168 (1993)
Pomerening, J. R., Sontag, E. D. & Ferrell, J. E. Building a cell cycle oscillator: hysteresis and bistability in the activation of Cdc2. Nature Cell Biol. 5, 346–351 (2003)
Foiani, M., Marini, F., Gamba, D., Lucchini, G. & Plevani, P. The B subunit of the DNA polymerase α-primase complex in Saccharomyces cerevisiae executes an essential function at the initial stage of DNA replication. Mol. Cell. Biol. 14, 923–933 (1994)
Acknowledgements
We thank C. Bouchoux, J. Kilmartin, E. Schwob and W. Zachariae for antibodies and constructs, and members of our laboratory for discussion and critical reading of the manuscript. This work was supported by a European Commission Marie Curie Individual Fellowship to S.L.-A., and the BBSRC and EC FP7 (O.K.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary information
This file contains Supplementary Figures 1-6 with Legends, Supplementary Data, Supplementary References and Supplementary Table 1. (PDF 1656 kb)
Rights and permissions
About this article
Cite this article
López-Avilés, S., Kapuy, O., Novák, B. et al. Irreversibility of mitotic exit is the consequence of systems-level feedback. Nature 459, 592–595 (2009). https://doi.org/10.1038/nature07984
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07984
This article is cited by
-
Quantitative model of eukaryotic Cdk control through the Forkhead CONTROLLER
npj Systems Biology and Applications (2021)
-
A PxL motif promotes timely cell cycle substrate dephosphorylation by the Cdc14 phosphatase
Nature Structural & Molecular Biology (2018)
-
Bistability analysis in mathematical model of M-phase control in Xenopus oocyte extracts
Computational and Applied Mathematics (2018)
-
Models in biology: lessons from modeling regulation of the eukaryotic cell cycle
BMC Biology (2015)
-
Robustness of cell cycle control and flexible orders of signaling events
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.