Abstract
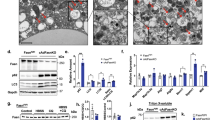
The intracellular storage and utilization of lipids are critical to maintain cellular energy homeostasis. During nutrient deprivation, cellular lipids stored as triglycerides in lipid droplets are hydrolysed into fatty acids for energy. A second cellular response to starvation is the induction of autophagy, which delivers intracellular proteins and organelles sequestered in double-membrane vesicles (autophagosomes) to lysosomes for degradation and use as an energy source. Lipolysis and autophagy share similarities in regulation and function but are not known to be interrelated. Here we show a previously unknown function for autophagy in regulating intracellular lipid stores (macrolipophagy). Lipid droplets and autophagic components associated during nutrient deprivation, and inhibition of autophagy in cultured hepatocytes and mouse liver increased triglyceride storage in lipid droplets. This study identifies a critical function for autophagy in lipid metabolism that could have important implications for human diseases with lipid over-accumulation such as those that comprise the metabolic syndrome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Martin, S. & Parton, R. G. Lipid droplets: a unified view of a dynamic organelle. Nature Rev. Mol. Cell Biol. 7, 373–378 (2006)
Zechner, R., Strauss, J. G., Haemmerle, G., Lass, A. & Zimmermann, R. Lipolysis: pathway under construction. Curr. Opin. Lipidol. 16, 333–340 (2005)
Finn, P. F. & Dice, J. F. Proteolytic and lipolytic responses to starvation. Nutrition 22, 830–844 (2006)
Komatsu, M. et al. Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. J. Cell Biol. 169, 425–434 (2005)
Mizushima, N., Levine, B., Cuervo, A. M. & Klionsky, D. J. Autophagy fights disease through cellular self-digestion. Nature 451, 1069–1075 (2008)
Mizushima, N. & Klionsky, D. J. Protein turnover via autophagy: implications for metabolism. Annu. Rev. Nutr. 27, 19–40 (2007)
Blommaart, E. F., Krause, U., Schellens, J. P., Vreeling-Sindelarova, H. & Meijer, A. J. The phosphatidylinositol 3-kinase inhibitors wortmannin and LY294002 inhibit autophagy in isolated rat hepatocytes. Eur. J. Biochem. 243, 240–246 (1997)
Leclercq, I. A. et al. CYP2E1 and CYP4A as microsomal catalysts of lipid peroxides in murine nonalcoholic steatohepatitis. J. Clin. Invest. 105, 1067–1075 (2000)
Sahai, A. et al. Roles of phosphatidylinositol 3-kinase and osteopontin in steatosis and aminotransferase release by hepatocytes treated with methionine-choline-deficient medium. Am. J. Physiol. Gastrointest. Liver Physiol. 291, G55–G62 (2006)
Owen, O. E., Reichard, G. A., Patel, M. S. & Boden, G. Energy metabolism in feasting and fasting. Adv. Exp. Med. Biol. 111, 169–188 (1979)
Kellner-Weibel, G., McHendry-Rinde, B., Haynes, M. P. & Adelman, S. Evidence that newly synthesized esterified cholesterol is deposited in existing cytoplasmic lipid inclusions. J. Lipid Res. 42, 768–777 (2001)
Brasaemle, D. L. et al. Perilipin A increases triacylglycerol storage by decreasing the rate of triacylglycerol hydrolysis. J. Biol. Chem. 275, 38486–38493 (2000)
Gocze, P. M. & Freeman, D. A. Factors underlying the variability of lipid droplet fluorescence in MA-10 Leydig tumor cells. Cytometry 17, 151–158 (1994)
Wiggins, D. & Gibbons, G. F. The lipolysis/esterification cycle of hepatic triacylglycerol. Its role in the secretion of very-low-density lipoprotein and its response to hormones and sulphonylureas. Biochem. J. 284, 457–462 (1992)
Bernales, S., McDonald, K. L. & Walter, P. Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response. PLoS Biol. 4, e423 (2006)
Roberts, P. et al. Piecemeal microautophagy of nucleus in Saccharomyces cerevisiae . Mol. Biol. Cell 14, 129–141 (2003)
Yorimitsu, T., Nair, U., Yang, Z. & Klionsky, D. J. Endoplasmic reticulum stress triggers autophagy. J. Biol. Chem. 281, 30299–30304 (2006)
Cuervo, A. M. et al. Autophagy and aging: the importance of maintaining “clean” cells. Autophagy 1, 131–140 (2005)
Ford, E. S., Giles, W. H. & Dietz, W. H. Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. J. Am. Med. Assoc. 287, 356–359 (2002)
Wang, Y., Schattenberg, J. M., Rigoli, R. M., Storz, P. & Czaja, M. J. Hepatocyte resistance to oxidative stress is dependent on protein kinase C-mediated down-regulation of c-Jun/AP-1. J. Biol. Chem. 279, 31089–31097 (2004)
Mizushima, N. et al. Dissection of autophagosome formation using Apg5-deficient mouse embryonic stem cells. J. Cell Biol. 152, 657–668 (2001)
Wang, Y. et al. Loss of macroautophagy promotes or prevents fibroblast apoptosis depending on the death stimulus. J. Biol. Chem. 283, 4766–4777 (2008)
Hoppel, C., DiMarco, J. P. & Tandler, B. Riboflavin and rat hepatic cell structure and function. Mitochondrial oxidative metabolism in deficiency states. J. Biol. Chem. 254, 4164–4170 (1979)
Piva, R. et al. Ablation of oncogenic ALK is a viable therapeutic approach for anaplastic large-cell lymphomas. Blood 107, 689–697 (2006)
Schattenberg, J. M., Wang, Y., Singh, R., Rigoli, R. M. & Czaja, M. J. Hepatocyte CYP2E1 overexpression and steatohepatitis lead to impaired hepatic insulin signaling. J. Biol. Chem. 280, 9887–9894 (2005)
Kaushik, S., Massey, A. C. & Cuervo, A. M. Lysosome membrane lipid microdomains: novel regulators of chaperone-mediated autophagy. EMBO J. 25, 3921–3933 (2006)
Postic, C. et al. Dual roles for glucokinase in glucose homeostasis as determined by liver and pancreatic beta cell-specific gene knock-outs using Cre recombinase. J. Biol. Chem. 274, 305–315 (1999)
Brasaemle, D. L. & Wolins, N. E. Isolation of lipid droplets from cells by density gradient centrifugation. Curr. Protoc. Cell Biol. Chapter 3, unit 3.15 (2006)
Cuervo, A. M., Palmer, A., Rivett, A. J. & Knecht, E. Degradation of proteasomes by lysosomes in rat liver. Eur. J. Biochem. 227, 792–800 (1995)
Goldstein, J. L., Basu, S. K. & Brown, M. S. Receptor-mediated endocytosis of low-density lipoprotein in cultured cells. Methods Enzymol. 98, 241–260 (1983)
Andersson, L. et al. PLD1 and ERK2 regulate cytosolic lipid droplet formation. J. Cell Sci. 119, 2246–2257 (2006)
Dunn, W. A. Studies on the mechanisms of autophagy: maturation of the autophagic vacuole. J. Cell Biol. 110, 1935–1945 (1990)
Nixon, R. A. et al. Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study. J. Neuropathol. Exp. Neurol. 64, 113–122 (2005)
Marzella, L., Ahlberg, J. & Glaumann, H. Isolation of autophagic vacuoles from rat liver: morphological and biochemical characterization. J. Cell Biol. 93, 144–154 (1982)
Millar, J. S., Cromley, D. A., McCoy, M. G., Rader, D. J. & Billheimer, J. T. Determining hepatic triglyceride production in mice: comparison of poloxamer 407 with Triton WR-1339. J. Lipid Res. 46, 2023–2028 (2005)
Acknowledgements
We thank D. Silver for his discussions, N. Mizushima for providing the Atg5-/- mouse embryonic fibroblasts, R. Stockert for the protein disulphide isomerase antibody and the personnel at the Analytical Imaging Facility for their technical assistance. This work was supported by National Institutes of Health grants from the National Institute of Diabetes and Digestive and Kidney Diseases and National Institute on Aging, a Glenn Award and an American Liver Foundation Postdoctoral Research Fellowship Award (R.S.).
Author Contributions R.S. performed biochemical analyses and immunoblots. S.K. performed the imaging studies and subcellular fractionations. Y.W. generated the shRNAs and performed immunoblotting. Y.X. performed biochemical analyses. R.S., Y.W., Y.X. and I.N. all contributed to the in vivo studies. M.K. and K.T. provided the knockout mice. A.M.C. and M.J.C. conceived and planned the study, analysed data and wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-19 with Legends. (PDF 13944 kb)
Supplementary Movie 1
This movie shows the dynamic association of lipid droplets (stained with BODIPY 493/503; green) with lysosomes (stained with Lysotracker; red). Cells were imaged at 30 sec intervals. Arrows point to colocalization event (top) and transient association/dissociation (bottom). (AVI 7245 kb)
Supplementary Movie 2
This movie shoes the dynamic association of lipid droplets (stained with BODIPY 493/503; green) with lysosomes (stained with Lysotracker; red). Cells were imaged at 30 sec intervals. Arrow points to colocalization event with lysosomes leading to a reduced size of the lipid droplet. (AVI 1187 kb)
Rights and permissions
About this article
Cite this article
Singh, R., Kaushik, S., Wang, Y. et al. Autophagy regulates lipid metabolism. Nature 458, 1131–1135 (2009). https://doi.org/10.1038/nature07976
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07976
This article is cited by
-
TBC1D5 reverses the capability of HIF-2α in tumor progression and lipid metabolism in clear cell renal cell carcinoma by regulating the autophagy
Journal of Translational Medicine (2024)
-
Lipid droplet accumulation in Wdr45-deficient cells caused by impairment of chaperone-mediated autophagic degradation of Fasn
Lipids in Health and Disease (2024)
-
Targeting DGAT1 inhibits prostate cancer cells growth by inducing autophagy flux blockage via oxidative stress
Oncogene (2024)
-
Viruses and autophagy: bend, but don’t break
Nature Reviews Microbiology (2024)
-
Control of NAD+ homeostasis by autophagic flux modulates mitochondrial and cardiac function
The EMBO Journal (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.