Abstract
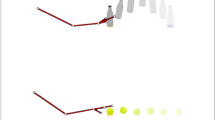
Typically developing human infants preferentially attend to biological motion within the first days of life1. This ability is highly conserved across species2,3 and is believed to be critical for filial attachment and for detection of predators4. The neural underpinnings of biological motion perception are overlapping with brain regions involved in perception of basic social signals such as facial expression and gaze direction5, and preferential attention to biological motion is seen as a precursor to the capacity for attributing intentions to others6. However, in a serendipitous observation7, we recently found that an infant with autism failed to recognize point-light displays of biological motion, but was instead highly sensitive to the presence of a non-social, physical contingency that occurred within the stimuli by chance. This observation raised the possibility that perception of biological motion may be altered in children with autism from a very early age, with cascading consequences for both social development and the lifelong impairments in social interaction that are a hallmark of autism spectrum disorders8. Here we show that two-year-olds with autism fail to orient towards point-light displays of biological motion, and their viewing behaviour when watching these point-light displays can be explained instead as a response to non-social, physical contingencies—physical contingencies that are disregarded by control children. This observation has far-reaching implications for understanding the altered neurodevelopmental trajectory of brain specialization in autism9.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
14 May 2009
The Y axis label on Fig. 3c was changed on 14 May 2009.
References
Simion, F., Regolin, L. & Bulf, H. A predisposition for biological motion in the newborn baby. Proc. Natl Acad. Sci. USA 105, 809–813 (2008)
Regolin, L., Tommasi, L. & Vallortigara, G. Visual perception of biological motion in newly hatched chicks as revealed by an imprinting procedure. Anim. Cogn. 3, 53–60 (2000)
Blake, R. Cats perceive biological motion. Psychol. Sci. 4, 54–57 (1993)
Johnson, M. H. Biological motion: a perceptual life detector? Curr. Biol. 16, R376–R377 (2006)
Pelphrey, K. A., Morris, J. P., Michelich, C. R., Allison, T. & McCarthy, G. Functional anatomy of biological motion perception in posterior temporal cortex: an fMRI study of eye, mouth and hand movements. Cereb. Cortex 15, 1866–1876 (2005)
Frith, C. D. & Frith, U. Interacting minds: a biological basis. Science 286, 1692–1695 (1999)
Klin, A. & Jones, W. Altered face scanning and impaired recognition of biological motion in a 15-month-old infant with autism. Dev. Sci. 11, 40–46 (2008)
Klin, A., Jones, W., Schultz, R. T. & Volkmar, F. The enactive mind – from actions to cognition: lessons from autism. Phil. Trans. R. Soc. Lond. B Biol. Sci. 358, 345–360 (2003)
Johnson, M. H. Functional brain development in humans. Nature Rev. Neurosci. 2, 475–483 (2001)
Johansson, G. Visual perception of biological motion and a model for its analysis. Percept. Psychophys. 14, 201–211 (1973)
Fox, R. & McDaniel, C. The perception of biological motion by human infants. Science 218, 486–487 (1982)
Oram, M. W. & Perrett, D. I. Integration of form and motion in the anterior superior temporal polysensory area (STPa) of the macaque monkey. J. Neurophysiol. 76, 109–129 (1996)
Omori, E. & Watanabe, S. Discrimination of Johansson’s stimuli in pigeons. Int. J. Comp. Psychol. 9, 92 (1996)
Vallortigara, G., Regolin, L. & Marconato, F. Visually inexperienced chicks exhibit spontaneous preference for biological motion patterns. PLoS Biol. 3, e208 (2005)
Thompson, J. C., Clarke, M., Stewart, T. & Puce, A. Configural processing of biological motion in human superior temporal sulcus. J. Neurosci. 25, 9059–9066 (2005)
Neri, P., Morrone, M. C. & Burr, D. C. Seeing biological motion. Nature 395, 894–896 (1998)
Jordan, H., Reiss, J. E., Hoffman, J. E. & Landau, B. Intact perception of biological motion in the face of profound spatial deficits: Williams syndrome. Psychol. Sci. 13, 162–167 (2002)
Jokisch, D., Troje, N. F., Koch, B., Schwarz, M. & Daum, I. Differential involvement of the cerebellum in biological and coherent motion perception. Eur. J. Neurosci. 21, 3439–3446 (2005)
Bidet-Caulet, A., Voisin, J., Bertrand, O. & Folumpt, P. Listening to a walking human activates the temporal biological motion area. Neuroimage 28, 132–139 (2005)
Chawarska, K., Klin, A. & Volkmar, F. R. Autism Spectrum Disorders in Infants and Toddlers: Diagnosis, Assessment and Treatment (Guilford Press, 2008)
Volkmar, F. R., Lord, C., Bailey, A., Schultz, R. T. & Klin, A. Autism and pervasive developmental disorders. J. Child Psychol. Psychiatry 45, 135–170 (2004)
Klin, A., Jones, W., Schultz, R., Volkmar, F. & Cohen, D. Visual fixation patterns during viewing of naturalistic social situations as predictors of social competence in individuals with autism. Arch. Gen. Psychiatry 59, 809–816 (2002)
Jones, W., Carr, K. & Klin, A. Absence of preferential looking to the eyes of approaching adults predicts level of social disability in 2-year-olds with autism spectrum disorder. Arch. Gen. Psychiatry 65, 946–954 (2008)
Schultz, R. T. Developmental deficits in social perception in autism: the role of the amygdala and fusiform face area. Int. J. Dev. Neurosci. 23, 125–141 (2005)
Dalton, K. M. et al. Gaze fixation and the neural circuitry of face processing in autism. Nature Neurosci. 8, 519–526 (2005)
Blake, R., Turner, L. M., Smoski, M. J., Pozdol, S. L. & Stone, W. L. Visual recognition of biological motion is impaired in children with autism. Psychol. Sci. 14, 151–157 (2003)
Pavlova, M. & Sokolov, A. Orientation specificity in biological motion perception. Percept. Psychophys. 62, 889–899 (2000)
Reid, V. M., Hoehl, S. & Striano, T. The perception of biological motion by infants: an event-related potential study. Neurosci. Lett. 395, 211–214 (2006)
Driver, J. & Spence, C. Multisensory perception: beyond modularity and convergence. Curr. Biol. 10, R731–R735 (2000)
Chen, P. Y. & Popovich, P. M. Correlation: Parametric and Nonparametric Measures 20–23 (Sage Publications, 2002)
Von Collani, E. & Dräger, K. Binomial Distribution Handbook for Scientists and Engineers 182–187 (Birkhäuser, 2001)
Leigh, R. J. & Zee, D. S. The Neurology of Eye Movements 3rd edn 94 (Oxford Univ. Press, 1999)
Hershey, J. & Movellan, J. R. in Advances in Neural Information Processing Systems 12 (eds Solla, S. A., Leen, T. K. & Muller, K. R.) 813–819 (MIT Press, 2000)
Bredin, H. & Chollet, G. Measuring audio and visual speech synchrony: methods and applications. IET International Conference on Visual Information Engineering 255–260 (2006)
Rabiner, L. R. & Schafer, R. W. Digital Processing of Speech Signals (Prentice-Hall, 1978)
Tchorz, J. & Kollmeier, B. Estimation of the signal-to-noise ratio with amplitude modulation spectrograms. Speech Commun. 38, 1–17 (2002)
Acknowledgements
This work was supported by a grant from the National Institutes of Mental Health (U54-MH66494), by Autism Speaks (piloting of methods), and by the Simons Foundation (current methodological developments). We wish to thank the families of the children included in this study for their time and participation. We would also like to thank F. Shic for conceptual discussions and thoughts on methods. We would like to thank L. Dickholtz, A. Kord, L. Else and M. Campisi for their assistance in creating the biological motion stimuli. We also wish to thank K. Carr, A. Blank, A. Bhatt, S. Shultz and K. Knoch for their help in this research project, and our colleagues K. Chawarska, R. Paul and F. Volkmar for conceptual discussions and for their contributions to the clinical characterization of the samples.
Author Contributions A.K. and W.J. developed the initial idea and design of the study, interpreted data, wrote the final manuscript, and take full responsibility for the integrity of data and the accuracy of data analysis. A.K. supervised participant characterization. W.J. supervised all technical aspects of experimental procedure, data acquisition and analysis. P.G. contributed to initial development of AVS methods and data analysis. D.J.L., with W.J. and G.R., developed the final AVS methods. G.R., with W.J. and A.K., helped develop new animations for the second experiment. W.J. created the figures. A.K. and W.J. performed the final revision of the manuscript for intellectual content.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Notes for Supplementary Movies 1-2 and Supplementary Video 3, Supplementary Tables 1-2, Supplementary Data and Supplementary References. (PDF 81 kb)
Supplementary Movie 1
This movie complements Figure 1 in the main text (see file s1 for full Legend). (MOV 7132 kb)
Supplementary Movie 2
This movie complements Figure 2 in the main text (see file s1 for full Legend). (MOV 8950 kb)
Supplementary Video 3
This video complements Figure 3 in the main text (see file s1 for full Legend). (MOV 14620 kb)
Rights and permissions
About this article
Cite this article
Klin, A., Lin, D., Gorrindo, P. et al. Two-year-olds with autism orient to non-social contingencies rather than biological motion. Nature 459, 257–261 (2009). https://doi.org/10.1038/nature07868
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07868
This article is cited by
-
Feasibility of a 2-minute eye-tracking protocol to support the early identification of autism
Scientific Reports (2024)
-
Salience network connectivity is altered in 6-week-old infants at heightened likelihood for developing autism
Communications Biology (2024)
-
Spatiotemporal patterns of spontaneous movement in neonates are significantly linked to risk of autism spectrum disorders at 18 months old
Scientific Reports (2023)
-
The Role of Play in Developing Social Skills Among Children Diagnosed with Autism Spectrum Disorder in Palestine
Journal of Psychosocial Rehabilitation and Mental Health (2023)
-
Attention Allocation During Exploration of Visual Arrays in ASD: Results from the ABC-CT Feasibility Study
Journal of Autism and Developmental Disorders (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.