Abstract
Arising from: S. K. Singh, M. N. Kagalwala, J. Parker-Thornburg, H. Adams & S. Majumder Nature 453, 223–227 (2008)10.1038/nature06863; Singh et al. reply
The DNA-binding protein REST (also called NRSF) is a transcriptional repressor that targets many neuronal genes1,2 and is abundant in human and mouse pluripotent embryonic stem cells (ESCs)3,4,5. In a recent Letter to Nature, Singh et al.6 suggested that REST controls the self-renewal and pluripotency of ESCs, because they found that ESCs in which a single REST allele was disrupted (Fig. 1a, β-geo-stop insertion) had reduced alkaline phosphatase activity and expressed lower levels of several pluripotency-associated genes6. Here we show that partial or complete loss of functional REST protein does not abrogate ESC potential as reflected by marker gene expression. These data are consistent with earlier reports7,8, and argue that REST is not required for maintaining ESC pluripotency.

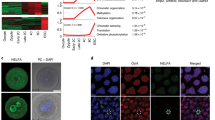
a, Genetic alterations to the Rest locus analysed here (phosphoglycerate kinase 1-neomycin resistance gene (Pgk-Neo) insertion7) and by Singh et al.6 (β-geo-stop insertions in the gene trap ESC lines RRC and YHC). Rectangles represent exons; coding regions are in black. b, REST, Oct4 and lamin B protein levels in wild-type (+/+), homozygous (-/-) and heterozygous (+/-) Rest ESC lysates, and (marked by an asterisk) in wild-type ESCs transfected with siRNA or shRNA constructs targeting Rest (siRest and shRest) or a control sequence (siCtrl and shCtrl). c, Chromatin immunoprecipitation of Rest+/+ (black), Rest+/- (grey) and Rest-/- (white) ESCs using anti-REST (left panel) and anti-Ezh2 (right panel) versus control antibody (immunoglobulin G, IgG). REST binds SG38457 (also known as Fam70b) and Syt4 (which both contain a RE1 motif) but not Math1 (also known as Atoh1; RE1-negative). No significant binding of REST was detected in Rest-/- ESCs. Ezh2-binding at Math1 (ref. 10) confirmed that chromatin fragments were intact. Error bars represent the standard deviation of three experiments. d, Alkaline phosphatase (AP) activity of mutant ESCs (percentage AP-positive colonies ± standard deviation). e, REST, Oct4 and lamin B protein levels in two additional Rest+/- (D05 and F12) and three wild-type ESC lysates. f, Transcript levels in D05 (dark grey), F12 (light grey) and wild-type (black) ESCs are shown (and, for comparison, retinoic-acid-treated embryoid bodies (white)) relative to wild type. Values were normalized to housekeeping genes and error bars show standard deviation of 4–6 experiments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Chong, J. A. et al. REST: a mammalian silencer protein that restricts sodium channel gene expression to neurons. Cell 80, 949–957 (1995)
Schoenherr, C. J. & Anderson, D. J. The neuron-restrictive silencer factor (NRSF): a coordinate repressor of multiple neuron-specific genes. Science 267, 1360–1363 (1995)
Boyer, L. A. et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122, 947–956 (2005)
Westbrook, T. F. et al. SCFβ-TRCP controls oncogenic transformation and neural differentiation through REST degradation. Nature 452, 370–374 (2008)
Kim, J., Chu, J., Shen, X., Wang, J. & Orkin, S. H. An extended transcriptional network for pluripotency of embryonic stem cells. Cell 132, 1049–1061 (2008)
Singh, S. K., Kagalwala, M. N., Parker-Thornburg, J., Adams, H. & Majumder, S. REST maintains self-renewal and pluripotency of embryonic stem cells. Nature 453, 223–227 (2008)
Chen, Z. F., Paquette, A. J. & Anderson, D. J. NRSF/REST is required in vivo for repression of multiple neuronal target genes during embryogenesis. Nature Genet. 20, 136–142 (1998)
Loh, Y. H. et al. The Oct4 and Nanog transcription network regulates pluripotency in mouse embryonic stem cells. Nature Genet. 38, 431–440 (2006)
Chen, Z. F. & Behringer, R. R. twist is required in head mesenchyme for cranial neural tube morphogenesis. Genes Dev. 9, 686–699 (1995)
Azuara, V. et al. Chromatin signatures of pluripotent cell lines. Nature Cell Biol. 8, 532–538 (2006)
Jorgensen, H. F. et al. The impact of chromatin modifiers on the timing of locus replication in mouse embryonic stem cells. Genome Biol. 8, R169 (2007)
Conti, L. et al. Niche-independent symmetrical self-renewal of a mammalian tissue stem cell. PLoS Biol. 3, e283 (2005)
Billon, N., Jolicoeur, C., Ying, Q. L., Smith, A. & Raff, M. Normal timing of oligodendrocyte development from genetically engineered, lineage-selectable mouse ES cells. J. Cell Sci. 115, 3657–3665 (2002)
Author information
Authors and Affiliations
PowerPoint slides
Rights and permissions
About this article
Cite this article
Jørgensen, H., Chen, ZF., Merkenschlager, M. et al. Is REST required for ESC pluripotency?. Nature 457, E4–E5 (2009). https://doi.org/10.1038/nature07783
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature07783
This article is cited by
-
Novel Rest functions revealed by conditional gene ablation
Medical Molecular Morphology (2018)
-
REST is a crucial regulator for acquiring EMT-like and stemness phenotypes in hormone-refractory prostate cancer
Scientific Reports (2017)
-
RE-1 silencing transcription factor (REST): a regulator of neuronal development and neuronal/endocrine function
Cell and Tissue Research (2015)
-
NRSF: an Angel or a Devil in Neurogenesis and Neurological Diseases
Journal of Molecular Neuroscience (2015)
-
Regulation of gene expression during early neuronal differentiation: evidence for patterns conserved across neuron populations and vertebrate classes
Cell and Tissue Research (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.