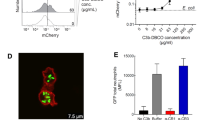
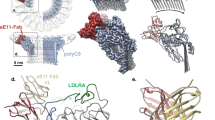
Abstract
The complement system is an essential component of the innate and acquired immune system1, and consists of a series of proteolytic cascades that are initiated by the presence of microorganisms. In health, activation of complement is precisely controlled through membrane-bound and soluble plasma-regulatory proteins including complement factor H (fH; ref. 2), a 155 kDa protein composed of 20 domains (termed complement control protein repeats). Many pathogens have evolved the ability to avoid immune-killing by recruiting host complement regulators3 and several pathogens have adapted to avoid complement-mediated killing by sequestering fH to their surface4. Here we present the structure of a complement regulator in complex with its pathogen surface-protein ligand. This reveals how the important human pathogen Neisseria meningitidis subverts immune responses by mimicking the host, using protein instead of charged-carbohydrate chemistry to recruit the host complement regulator, fH. The structure also indicates the molecular basis of the host-specificity of the interaction between fH and the meningococcus, and informs attempts to develop novel therapeutics and vaccines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lachmann, P. J. Biological functions of the complement system. Biochem. Soc. Trans. 18, 1143–1145 (1990)
Zipfel, P. F., Jokiranta, T. S., Hellwage, J., Koistinen, V. & Meri, S. The factor H protein family. Immunopharmacology 42, 53–60 (1999)
Lambris, J. D., Ricklin, D. & Geisbrecht, B. V. Complement evasion by human pathogens. Nature Rev. Microbiol. 6, 132–142 (2008)
Jozsi, M. & Zipfel, P. F. Factor H family proteins and human diseases. Trends Immunol. 29, 380–387 (2008)
Stephens, D. S., Greenwood, B. & Brandtzaeg, P. Epidemic meningitis, meningococcaemia, and Neisseria meningitidis . Lancet 369, 2196–2210 (2007)
Feavers, I. M. ABC of meningococcal diversity. Nature 404, 451–452 (2000)
Gray, S. J. et al. Epidemiology of meningococcal disease in England and Wales 1993/94 to 2003/04: Contribution and experiences of the Meningococcal Reference Unit. J. Med. Microbiol. 55, 887–896 (2006)
Harrison, L. H. Prospects for vaccine prevention of meningococcal infection. Clin. Microbiol. Rev. 19, 142–164 (2006)
Fletcher, L. D. et al. Vaccine potential of the Neisseria meningitidis 2086 lipoprotein. Infect. Immun. 72, 2088–2100 (2004)
Masignani, V. et al. Vaccination against Neisseria meningitidis using three variants of the lipoprotein GNA1870. J. Exp. Med. 197, 789–799 (2003)
Cantini, F. et al. Solution structure of the immunodominant domain of protective antigen GNA1870 of Neisseria meningitidis . J. Biol. Chem. 281, 7220–7227 (2006)
Giuliani, M. M. et al. The region comprising amino acids 100 to 255 of Neisseria meningitidis lipoprotein GNA 1870 elicits bactericidal antibodies. Infect. Immun. 73, 1151–1160 (2005)
Madico, G. et al. The meningococcal vaccine candidate GNA1870 binds the complement regulatory protein factor H and enhances serum resistance. J. Immunol. 177, 501–510 (2006)
Schneider, M. C. et al. Functional significance of factor H binding to Neisseria meningitidis . J. Immunol. 176, 7566–7575 (2006)
Haralambous, E. et al. Factor H, a regulator of complement activity, is a major determinant of meningococcal disease susceptibility in UK Caucasian patients. Scand. J. Infect. Dis. 38, 764–771 (2006)
Davies, D. R., Padlan, E. A. & Sheriff, S. Antibody-antigen complexes. Annu. Rev. Biochem. 59, 439–473 (1990)
Edwards, A. O. et al. Complement factor H polymorphism and age-related macular degeneration. Science 308, 421–424 (2005)
Hageman, G. S. et al. A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proc. Natl Acad. Sci. USA 102, 7227–7232 (2005)
Haines, J. L. et al. Complement factor H variant increases the risk of age-related macular degeneration. Science 308, 419–421 (2005)
Beernink, P. T. & Granoff, D. M. Bactericidal antibody responses induced by meningococcal recombinant chimeric factor H-binding protein vaccines. Infect. Immun. 76, 2568–2575 (2008)
Prosser, B. E. et al. Structural basis for complement factor H linked age-related macular degeneration. J. Exp. Med. 204, 2277–2283 (2007)
Schmidt, C. Q. et al. A new map of glycosaminoglycan and C3b binding sites on factor H. J. Immunol. 181, 2610–2619 (2008)
Meri, S. & Pangburn, M. K. Discrimination between activators and nonactivators of the alternative pathway of complement: Regulation via a sialic acid/polyanion binding site on factor H. Proc. Natl Acad. Sci. USA 87, 3982–3986 (1990)
Fedtke, I., Gotz, F. & Peschel, A. Bacterial evasion of innate host defenses–the Staphylococcus aureus lesson. Int. J. Med. Microbiol. 294, 189–194 (2004)
Granoff, D. M., Welsch, J. A. & Ram, S. Binding of complement factor H to Neisseria meningitidis is specific for human fH and inhibits complement activation by rat and rabbit sera. Infect. Immun. 77, 764–769 (2009)
Gibbs, R. A. et al. Evolutionary and biomedical insights from the rhesus macaque genome. Science 316, 222–234 (2007)
Heckenberg, S. G. et al. Clinical features, outcome, and meningococcal genotype in 258 adults with meningococcal meningitis: A prospective cohort study. Medicine (Baltimore) 87, 185–192 (2008)
Jokiranta, T. S. et al. Analysis of the recognition mechanism of the alternative pathway of complement by monoclonal anti-factor H antibodies: Evidence for multiple interactions between H and surface bound C3b. FEBS Lett. 393, 297–302 (1996)
Sim, E., Palmer, M. S., Puklavec, M. & Sim, R. B. Monoclonal antibodies against the complement control protein factor H (beta 1 H). Biosci. Rep. 3, 1119–1131 (1983)
Prosser, B. E. et al. Expression, purification, cocrystallization and preliminary crystallographic analysis of sucrose octasulfate/human complement regulator factor H SCRs 6-8. Acta Crystallogr. F 63, 480–483 (2007)
Bahar, M. et al. SPINE workshop on automated X-ray analysis: a progress report. Acta Crystallogr. D 62, 1170–1183 (2006)
Kabsch, W. Automatic processing of rotation diffraction data from crystals of initially unknown symmetry and cell constants. J. Appl. Crystallogr. 26, 795–800 (1993)
Evans, P. R. Data reduction. In Proceedings of CCP4 Study Weekend (eds Sawyer, L., Isaacs, N. & Bailey, S.) 114–122 (SERC, Daresbury Laboratory, 1993)
Prosser, B. E. et al. Structural basis for complement factor H linked age-related macular degeneration. J. Exp. Med. 204, 2277–2283 (2007)
Vagin, A. A. & Isupov, M. N. Spherically averaged phased translation function and its application to the search for molecules and fragments in electron-density maps. Acta Crystallogr. D 57, 1451–1456 (2001)
Winn, M. D. An overview of the CCP4 project in protein crystallography: An example of a collaborative project. J. Synchrotron Radiat. 10, 23–25 (2003)
Blanc, E. et al. Refinement of severely incomplete structures with maximum likelihood in BUSTER-TNT. Acta Crystallogr. D 60, 2210–2221 (2004)
De La Fortelle, E. & Bricogne, G. Maximum-likelihood heavy-atom parameter refinement for the multiple isomorphous replacement and multiwavelength-anomalous dispersion methods. Methods Enzymol. 276, 472–494 (1997)
Cowtan, K. & Main, P. Miscellaneous algorithms for density modification. Acta Crystallogr. D 54, 487–493 (1998)
Emsley, P. & Cowtan, K. Coot: Model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Davis, I. W. et al. MolProbity: All-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res. 35, W375–383 (2007)
Acknowledgements
Thanks are due to G. Bricogne and the Global Phasing Consortium for access to a β version of autoSHARP and autoBUSTER, to the beamline staff of the European Synchrotron Radiation Source (particularly G. Leonard) and DIAMOND (UK), and to many members of the Laboratory of Molecular Biophysics, Oxford, and the Lea group for help with X-ray data collection. B.E.P. was funded by a Wellcome Trust studentship; P.R. and S.J. by Medical Research Council grants to S.M.L.; J.E.D. by a Wellcome Trust grant to SM.L.; and J.E.L. by an Engineering and Physical Sciences Research Grant to S.M.L. Work in C.M.T.’s laboratory is funded by the Wellcome Trust and the Medical Research Council. E.K. is an EMBO fellow.
Author Contributions B.E.P., J.E.D. and J.J.E.C. performed SPR experiments; B.E.P., J.J.E.C., J.E.L. and S.Q. expressed proteins and crystallized the complex; M.C.S., E.K. and Q.Z. performed all assays with Neisseria and expressed fHbp; S.J., P.R. and S.M.L. collected and analysed X ray data; S.M.L. phased the X-ray data and built/refined the models; and S.M.L. and C.M.T. designed the research and wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Methods and Supplementary Data, Supplementary Tables 1-2, Supplementary Figures 1-11 with Legends and Supplementary References (PDF 2346 kb)
Rights and permissions
About this article
Cite this article
Schneider, M., Prosser, B., Caesar, J. et al. Neisseria meningitidis recruits factor H using protein mimicry of host carbohydrates. Nature 458, 890–893 (2009). https://doi.org/10.1038/nature07769
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07769
This article is cited by
-
Immunological fingerprint of 4CMenB recombinant antigens via protein microarray reveals key immunosignatures correlating with bactericidal activity
Nature Communications (2020)
-
A receptor for the complement regulator factor H increases transmission of trypanosomes to tsetse flies
Nature Communications (2020)
-
Complement Evasion Strategies of Human Pathogenic Bacteria
Indian Journal of Microbiology (2020)
-
Human genetics of meningococcal infections
Human Genetics (2020)
-
A homopolymeric adenosine tract in the promoter region of nspA influences factor H-mediated serum resistance in Neisseria meningitidis
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.