Abstract
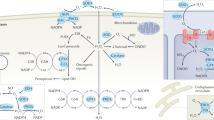
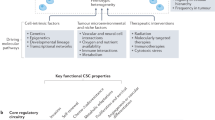
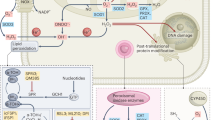
The metabolism of oxygen, although central to life, produces reactive oxygen species (ROS) that have been implicated in processes as diverse as cancer, cardiovascular disease and ageing. It has recently been shown that central nervous system stem cells1,2 and haematopoietic stem cells and early progenitors3,4,5,6 contain lower levels of ROS than their more mature progeny, and that these differences are critical for maintaining stem cell function. We proposed that epithelial tissue stem cells and their cancer stem cell (CSC) counterparts may also share this property. Here we show that normal mammary epithelial stem cells contain lower concentrations of ROS than their more mature progeny cells. Notably, subsets of CSCs in some human and murine breast tumours contain lower ROS levels than corresponding non-tumorigenic cells (NTCs). Consistent with ROS being critical mediators of ionizing-radiation-induced cell killing7,8, CSCs in these tumours develop less DNA damage and are preferentially spared after irradiation compared to NTCs. Lower ROS levels in CSCs are associated with increased expression of free radical scavenging systems. Pharmacological depletion of ROS scavengers in CSCs markedly decreases their clonogenicity and results in radiosensitization. These results indicate that, similar to normal tissue stem cells, subsets of CSCs in some tumours contain lower ROS levels and enhanced ROS defences compared to their non-tumorigenic progeny, which may contribute to tumour radioresistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
09 April 2009
The name of the coauthor Frederick M. Dirbas was corrected on 9 April 2009.
References
Smith, J., Ladi, E., Mayer-Proschel, M. & Noble, M. Redox state is a central modulator of the balance between self-renewal and differentiation in a dividing glial precursor cell. Proc. Natl Acad. Sci. USA 97, 10032–10037 (2000)
Tsatmali, M., Walcott, E. C. & Crossin, K. L. Newborn neurons acquire high levels of reactive oxygen species and increased mitochondrial proteins upon differentiation from progenitors. Brain Res. 1040, 137–150 (2005)
Ito, K. et al. Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature 431, 997–1002 (2004)
Ito, K. et al. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nature Med. 12, 446–451 (2006)
Tothova, Z. et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell 128, 325–339 (2007)
Miyamoto, K. et al. Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell 1, 101–112 (2007)
Powell, S. & McMillan, T. J. DNA damage and repair following treatment with ionizing radiation. Radiother. Oncol. 19, 95–108 (1990)
Ward, J. F. Biochemistry of DNA lesions. Radiat. Res. Suppl. 8, S103–S111 (1985)
Shackleton, M. et al. Generation of a functional mammary gland from a single stem cell. Nature 439, 84–88 (2006)
Stingl, J. et al. Purification and unique properties of mammary epithelial stem cells. Nature 439, 993–997 (2006)
Jang, Y. Y. & Sharkis, S. J. A low level of reactive oxygen species selects for primitive hematopoietic stem cells that may reside in the low-oxygenic niche. Blood 110, 3056–3063 (2007)
Hosokawa, K. et al. Function of oxidative stress in the regulation of hematopoietic stem cell-niche interaction. Biochem. Biophys. Res. Commun. 363, 578–583 (2007)
Liu, R. et al. The prognostic role of a gene signature from tumorigenic breast-cancer cells. N. Engl. J. Med. 356, 217–226 (2007)
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005)
Cho, R. W. et al. Isolation and molecular characterization of cancer stem cells in MMTV-Wnt-1 murine breast tumors. Stem Cells 26, 364–371 (2008)
Riley, P. A. Free radicals in biology: oxidative stress and the effects of ionizing radiation. Int. J. Radiat. Biol. 65, 27–33 (1994)
Dorie, M. J. et al. DNA damage measured by the comet assay in head and neck cancer patients treated with tirapazamine. Neoplasia 1, 461–467 (1999)
Cohen-Jonathan, E., Bernhard, E. J. & McKenna, W. G. How does radiation kill cells? Curr. Opin. Chem. Biol. 3, 77–83 (1999)
Bao, S. et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444, 756–760 (2006)
Phillips, T. M., McBride, W. H. & Pajonk, F. The response of CD24-/low/CD44+ breast cancer-initiating cells to radiation. J. Natl. Cancer Inst. 98, 1777–1785 (2006)
Estrela, J. M., Ortega, A. & Obrador, E. Glutathione in cancer biology and therapy. Crit. Rev. Clin. Lab. Sci. 43, 143–181 (2006)
Warren, L., Bryder, D., Weissman, I. L. & Quake, S. R. Transcription factor profiling in individual hematopoietic progenitors by digital RT–PCR. Proc. Natl Acad. Sci. USA 103, 17807–17812 (2006)
Griffith, O. W. Biologic and pharmacologic regulation of mammalian glutathione synthesis. Free Radic. Biol. Med. 27, 922–935 (1999)
Hahn, S. M. et al. Potential use of nitroxides in radiation oncology. Cancer Res. 54, 2006s–2010s (1994)
Bailey, H. H. l-S,R-buthionine sulfoximine: historical development and clinical issues. Chem. Biol. Interact. 111–112, 239–254 (1998)
Jamieson, C. H. et al. Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N. Engl. J. Med. 351, 657–667 (2004)
Akala, O. O. et al. Long-term haematopoietic reconstitution by Trp53-/-p16Ink4a-/-p19Arf-/- multipotent progenitors. Nature 453, 228–232 (2008)
Saretzki, G. et al. Downregulation of multiple stress defense mechanisms during differentiation of human embryonic stem cells. Stem Cells 26, 455–464 (2008)
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J. & Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl Acad. Sci. USA 100, 3983–3988 (2003)
Prince, M. E. et al. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc. Natl Acad. Sci. USA 104, 973–978 (2007)
Liao, M. J. et al. Enrichment of a population of mammary gland cells that form mammospheres and have in vivo repopulating activity. Cancer Res. 67, 8131–8138 (2007)
Acknowledgements
We thank D. Spitz for helpful discussions and D. Menke, D. Rossi, and J. Seita for technical assistance. This work was supported by grants from the National Institutes of Health (M.F.C., G.S. and I.L.W.), the Virginia and D.K. Ludwig Foundation (M.F.C. and I.L.W.), the Breast Cancer Research Foundation (M.F.C.), the Machiah foundation (T.K.), the American Society for Therapeutic Radiology and Oncology (M.D.), and the Radiological Society of North America (M.D.). M.D. is a recipient of the Leonard B. Holman Research Pathway fellowship.
Author Contributions M.D. and R.W.C. contributed equally to this work. M.D., R.W.C., N.A.L., T.K., M.J.D., A.N.K., D.Q., J.S.L., L.A. and M.W. performed the experiments. B.J., M.J.K., I.W., F.D., G.S., C.G., B.P., J.S. and S.K.L. aided in human tumour tissue acquisition. G.S. designed a pre-operative protocol allowing for tissue acquisition. M.D., R.W.C. and M.F.C. designed the experiments and wrote the manuscript. S.R.Q., J.M.B. and I.L.W. provided intellectual input and aided in experimental design.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
[Competing Interests: M.F.C. is a founder and consultant for Oncomed Pharmaceuticals, Inc. in Redwood City, California, USA.]
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S7 with Legends and Supplementary Tables 1-4 (PDF 344 kb)
Rights and permissions
About this article
Cite this article
Diehn, M., Cho, R., Lobo, N. et al. Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature 458, 780–783 (2009). https://doi.org/10.1038/nature07733
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07733
This article is cited by
-
Oxidative stress promotes cytotoxicity in human cancer cell lines exposed to Escallonia spp. extracts
BMC Complementary Medicine and Therapies (2024)
-
POLQ inhibition attenuates the stemness and ferroptosis resistance in gastric cancer cells via downregulation of dihydroorotate dehydrogenase
Cell Death & Disease (2024)
-
Hypoxia, oxidative stress, and the interplay of HIFs and NRF2 signaling in cancer
Experimental & Molecular Medicine (2024)
-
Potential Role of Nrf2, HER2, and ALDH in Cancer Stem Cells: A Narrative Review
The Journal of Membrane Biology (2024)
-
Regulation and signaling pathways in cancer stem cells: implications for targeted therapy for cancer
Molecular Cancer (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.